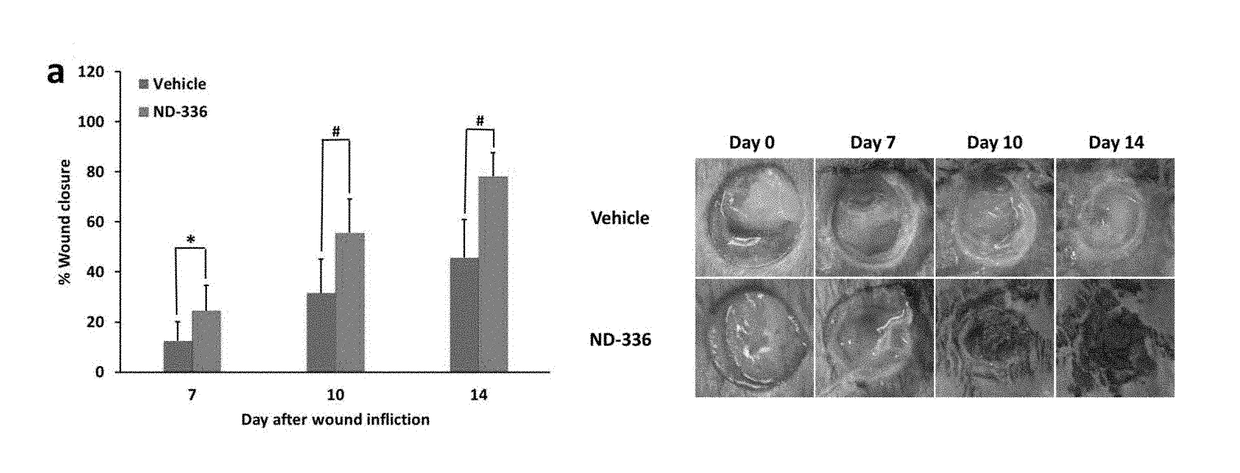
Acceleration of diabetic wound healing
a wound healing and acceleration technology, applied in the field of acceleration of diabetic wound healing, can solve the problems of amputation of lower limbs, and inability to accelerate wound healing, so as to accelerate the healing process of wounds
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
and Analysis of MMP Inhibitors
[0125]Synthesis of ND-336. The synthesis of ND-336 is shown in Scheme 1. The reaction of 4-mercaptophenol (2) with allyl bromide (1) produced compound 3, which was allowed to react with 4-fluorobenzonitrile to afford diphenyl ether 4 in good yields (94% and 82% for the first and second steps, respectively). Subsequent reduction of the nitrile with LiAlH4, followed by Boc-protection of the resultant amine yielded compound 5, which was oxidized to the corresponding oxirane 6. The reaction of 6 with thiourea produced the Boc-protected thiirane 7. After the final acid Boc-deprotection, the desired ND-336 was in hand as the HCl salt.
Other MMP-9 inhibitors can be prepared by the methods described in U.S. Pat. No. 6,703,415 (Mobashery et al.); U.S. Pat. No. 7,114,917 (Mobashery et al.); U.S. Pat. No. 7,928,127 (Lee et al.), U.S. Pat. No. 8,093,287 (Lee et al.); and U.S. Pat. No. 8,937,151 (Chang et al.); U.S. Patent Publication No. 2013 / 0064878 (Chang et al.);...
example 2
ion of Diabetic Wound Healing Using a Novel Protease-Anti-Protease Combination Therapy
[0138]Enzyme Inhibition Studies.
[0139]Human recombinant active MMP-2 and MMP-7, and the catalytic domains of MMP-3 and MMP-14 / MT1-MMP were purchased from EMD Chemicals, Inc. (San Diego, Calif., USA); human recombinant catalytic domains of MMP-1, MMP-8, and MMP-9 were purchased from Enzo Life Sciences, Inc. (Farmingdale, N.Y., USA); human recombinant active ADAM9 and ADAM10 were purchased from R&D Systems (Minneapolis, Minn., USA). Fluorogenic substrates MOCAc-Pro-Leu-Gly-Leu-A2pr(Dnp)-Ala-Arg-NH2 (for MMP-2, MMP-7, MMP-9 and MMP-14) and MOCAc-Arg-Pro-Lys-Pro-Val-Glu-Nva-Trp-Arg-Lys(Dnp)-NH2 (for MMP-3) were purchased from Peptides International (Louisville, Ky., USA); Mca-KPLGL-Dpa-AR-NH2 (for MMP-1, MMP-8 and ADAM10) and Mca-PLAQAV-Dpa-RSSSR-NH2 (for ADAM9) were purchased from R&D Systems (Minneapolis, Minn., USA). The Km values for MMP-2, MMP-9 and MMP-14 were the same as previously reported by G...
example 3
tical Dosage Forms
[0165]The following formulations illustrate representative pharmaceutical dosage forms that may be used for the therapeutic or prophylactic administration of a compound, enzyme, or composition described herein, or a pharmaceutically acceptable salt, solvate, or composition thereof (hereinafter referred to as ‘Compound X’):
(i) Tablet 1mg / tablet‘Compound X’100.0Lactose77.5Povidone15.0Croscarmellose sodium12.0Microcrystalline cellulose92.5Magnesium stearate3.0300.0
(ii) Tablet 2mg / tablet‘Compound X’20.0Microcrystalline cellulose410.0Starch50.0Sodium starch glycolate15.0Magnesium stearate5.0500.0
(iii) Capsulemg / capsule‘Compound X’10.0Colloidal silicon dioxide1.5Lactose465.5Pregelatinized starch120.0Magnesium stearate3.0600.0
(iv) Injection 1 (1 mg / mL)mg / mL‘Compound X’ (free acid form)1.0Dibasic sodium phosphate12.0Monobasic sodium phosphate0.7Sodium chloride4.51.0N Sodium hydroxide solutionq.s.(pH adjustment to 7.0-7.5)Water for injectionq.s. ad 1 mL
(v) Injection 2 (10 m...
PUM
| Property | Measurement | Unit |
|---|---|---|
| residence times | aaaaa | aaaaa |
| residence times | aaaaa | aaaaa |
| residence times | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com