Slow releasing micropill containing active component of indapamide and method for preparing the same
A technology of sustained-release pellets and indapamide, which is used in pharmaceutical formulations, bulk delivery, blood diseases, etc., can solve the problems of sudden release of sustained-release tablets and difficult quality control, and achieve increased safety, high yield and Reproducible, easy-to-industrial effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
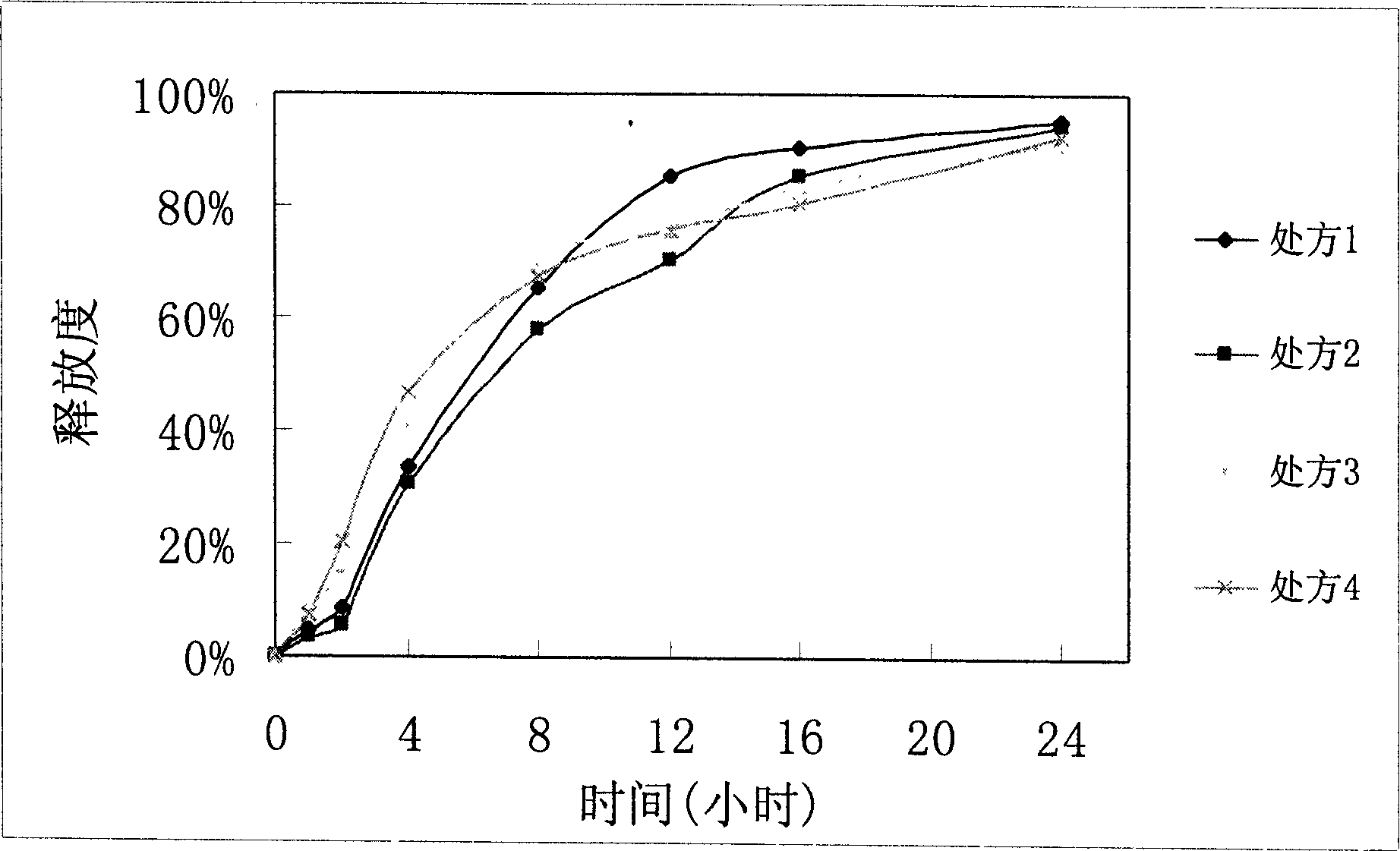
Image
Examples
Embodiment 1
[0039] Embodiment 1, the preparation of blank pellet
[0040] 1
2
100g
30g
40g
20g
10g
silica
80g
60g
20g
20g
20g
[0041] B. Preparation process
[0042] Mix the prescription amount of lactose (or microcrystalline cellulose) and starch evenly, add the binder prepared by sucrose or hypromellose, use a granulator (or coating pan) to start and roll the pellets to the required The mesh number is 15-30 mesh (lubricant and glidant are added while rolling the pellets), dried, and the best 18-25 mesh is screened out.
Embodiment 2
[0043] Embodiment 2, the preparation of quick-release pellets
[0044] 1
2
Blank pellets
Indapamide
polyethylene glycol
water
120g
1.5g
2g
10g
0.4g
100ml
Blank pellets
Indapamide
polyethylene glycol
70% ethanol
120g
1.5g
5g
15g
0.3g
100ml
[0045] B. Preparation process
[0046] Add indapamide, hypromellose (or polyvinylpyrrolidone) and talcum powder described in the prescription into water or 70% ethanol respectively, and stir for 20 minutes to form a uniform mixed solution. In the fluidized bed (or coating pan), the temperature of the pellets was kept at 30° C., and the mixed solution was evenly sprayed on the outer layer of the blank pellets obtained in Example 1 to obtain immediate-release pellets.
Embodiment 3
[0047] Embodiment 3, the preparation of sustained-release pellets
[0048] 1
2
medicated pellets
Acrylic resin NE30D
Talc powder
water
150g
20g
3.5g
5g
150ml
medicated pellets
Acrylic RS30D
Acrylic resin RL30D
Talc powder
water
150g
15g
1.5g
3.8g
11.3g
135ml
3
4
medicated pellets
0.1% diethyl phthalate
75% ethanol
150g
10g
2g
5g
100ml
medicated pellets
Aqueous dispersion of ethyl cellulose
Talc powder
water
150g
15g
10g
100ml
[0049] B. Preparation process
[0050] Put the quick-release pellets obtained in Example 2 into a fluidized bed (or coating pan), and keep the temperature of the pellets at 2...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com