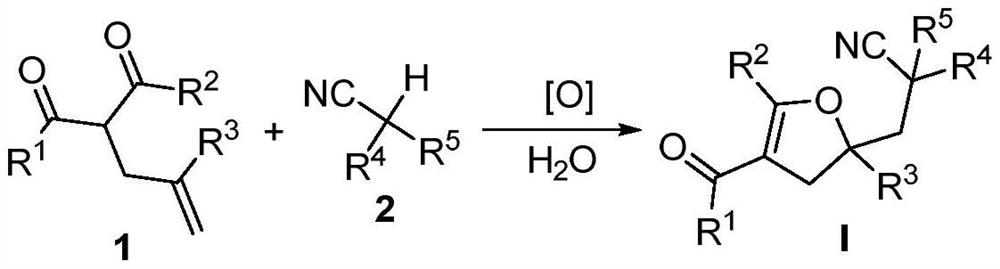
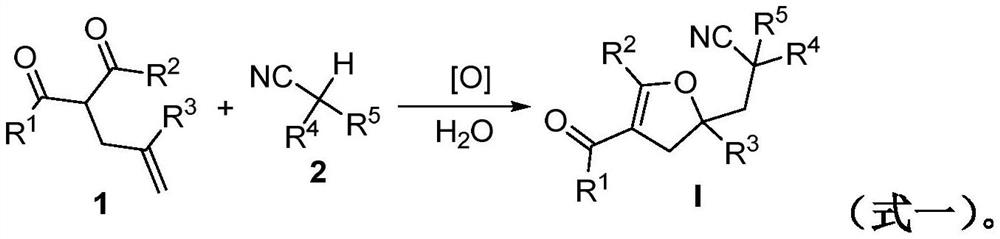
Method for preparing a 2, 3-dihydrofuran derivative
A kind of technology of dihydrofuran and derivatives, applied in the field of organic synthesis
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0030]
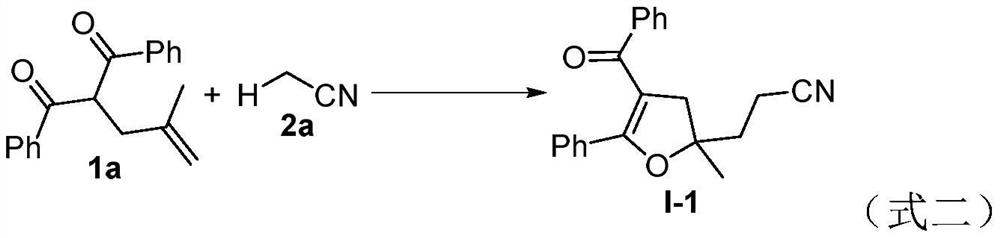
[0031] In the Schlenk bottle, add the compound (55.6mg, 0.2mmol) shown in formula 1a, the nitrile compound (0.2mL) shown in formula 2a, H 2 O (1.8mL), tert-butyl peroxybenzoate (TBPB, 77.7mg, 2.0eq), then the reactor was stirred and reacted at 130°C under an air atmosphere, and the reaction process was monitored by TLC until the raw materials disappeared (the reaction time was 12 hours), after the reaction was completed, the reaction solution was extracted with ethyl acetate, the organic phase was dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to remove the solvent, and the residue was separated by column chromatography (elution solvent: ethyl acetate / n-hexane) to obtain target product I-1 (83%yield); 1 H NMR (500MHz, CDCl 3 )δ:7.43-7.41(m,2H),7.24-7.21(m,1H),7.19-7.17(m,3H),7.09-7.04(m,4H),3.19-3.10(m,2H),2.60- 2.56(m,2H),2.26-2.16(m,2H),1.59(s,3H); 13 C NMR (125MHz, CDCl 3 )δ: 193.4, 164.3, 138.8, 131.3, 130.9, 130.2, 1...
Embodiment 2
[0033] The reaction temperature was lowered to 120° C. for the reaction, and the remaining conditions were the same as in Example 1, and the yield of the target product I-1 was 65%.
Embodiment 3
[0035] The oxidizing agent used dibenzoyl peroxide (BPO) instead of tert-butyl peroxybenzoate (TBPB), and the remaining conditions were the same as in Example 1, and the yield of the target product I-1 was 61%.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com