Monoclonal cell strain of human embryonic kidney epithelial cells 293T-Clone3 and application of monoclonal cell strain of human embryonic kidney epithelial cells 293T-Clone3
A 293t-clone3, epithelial cell technology, applied to embryonic cells, urinary tract/kidney cells, artificial cell constructs, etc., can solve the problem of low AAV production efficiency
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
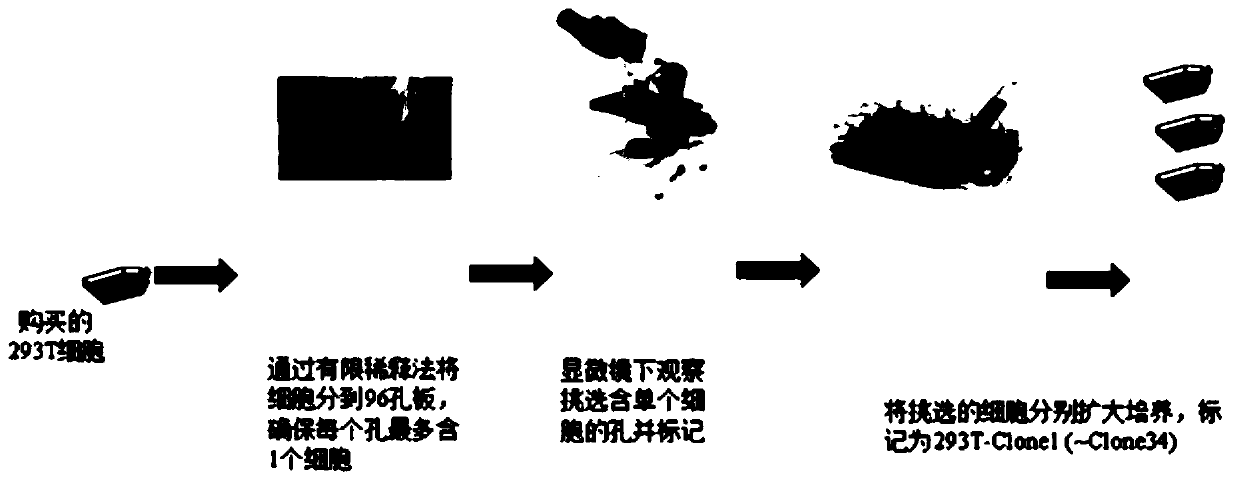
[0030] Obtaining 293T-Clone3 Monoclonal Cell Line of Human Embryonic Kidney Epithelial Cells
[0031] (1) Culture the 293T cells purchased from ATCC, and divide them into multiple cell culture flasks for culture when they are full.
[0032] (2) When the cell density of one of the bottles is about 70-80%, discard the supernatant, wash it once with PBS, and then add an appropriate amount of trypsin digestion solution to digest in an incubator to disperse them into single cells.
[0033] (3) After the digestion to a single cell was observed under a microscope, complete medium was added to terminate the digestion, and then centrifuged at 1000 rpm for 5 min.
[0034] (4) Discard the supernatant, resuspend the cells with complete medium and count with trypan blue.
[0035] (5) Dilute the cells with cell culture medium (the dilution ratio is based on the fact that each well of the 96-well plate contains at most 1 cell) and add them to the 96-well plate for culture;
[0036] (6) On ...
Embodiment 2
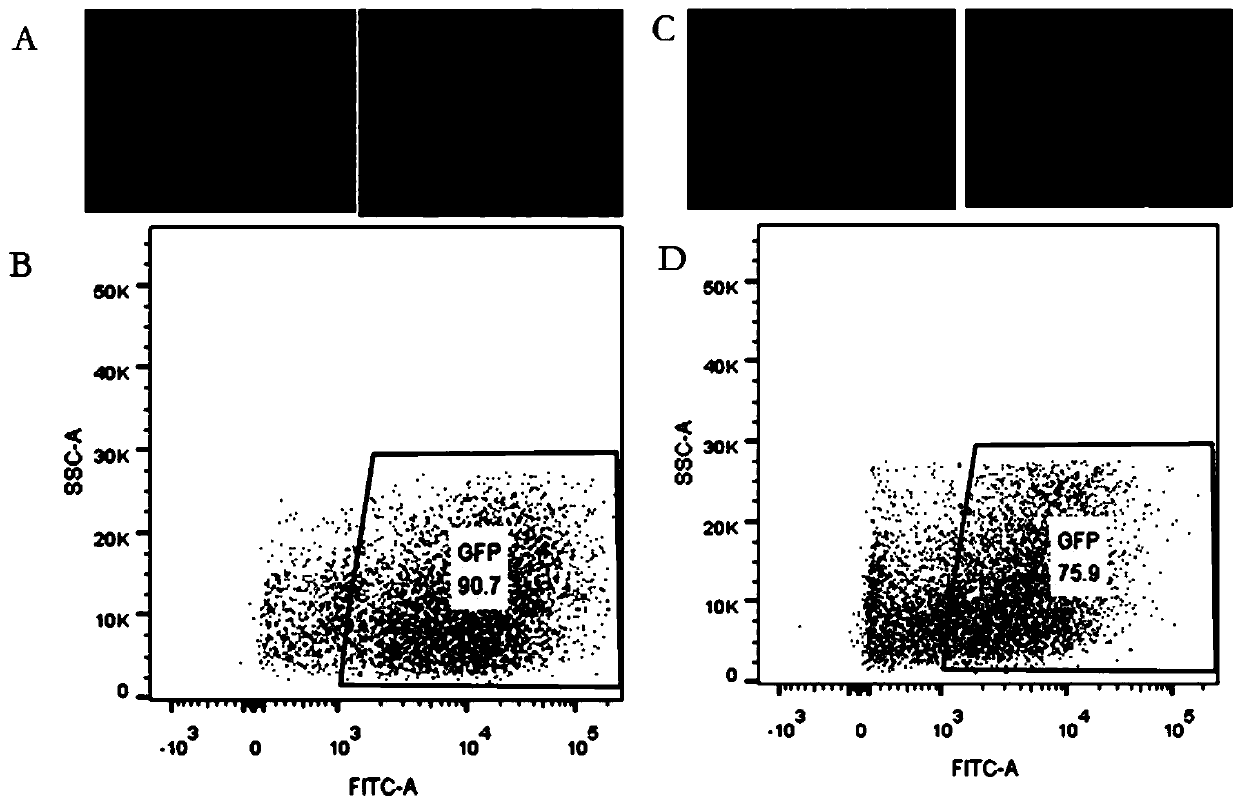
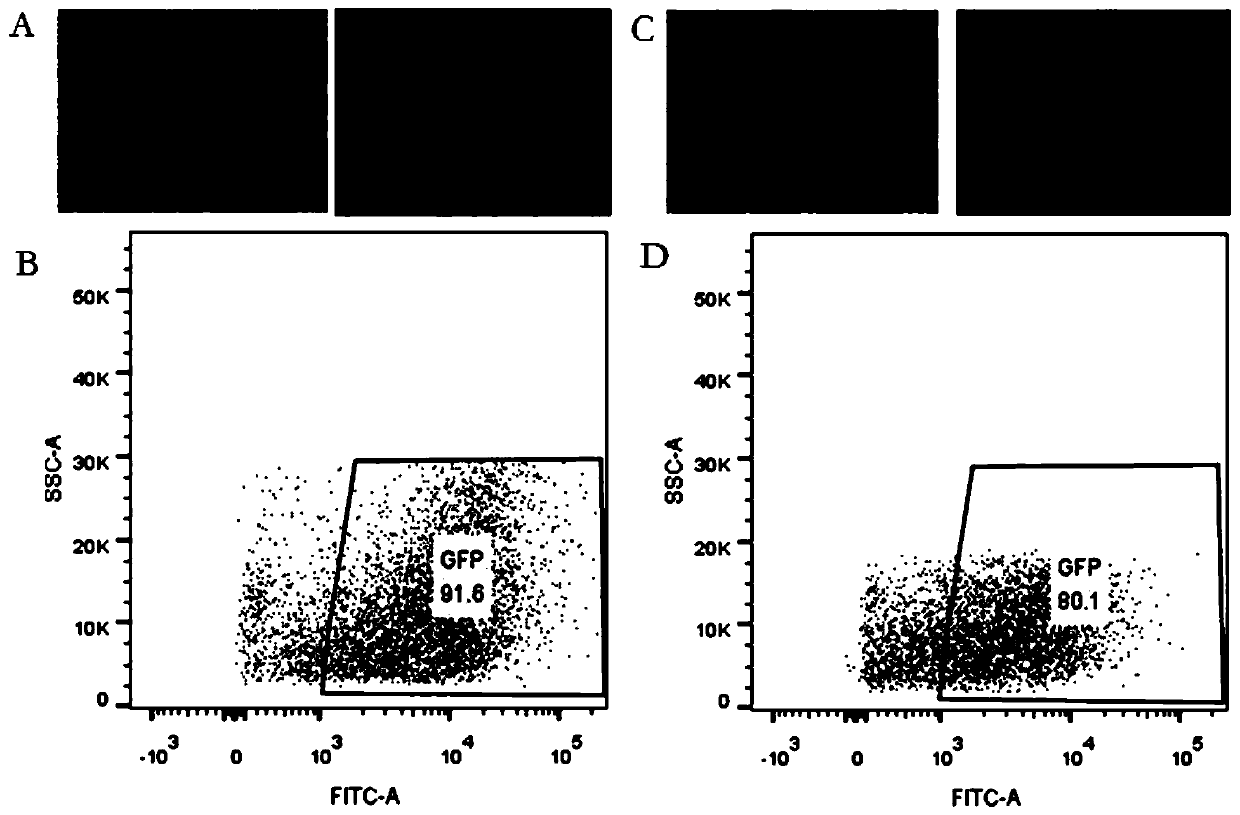
[0039] 1. Virus packaging test of human embryonic kidney epithelial cell 293T-Clone3 monoclonal cell line
[0040] A human embryonic kidney epithelial cell 293T-C3 monoclonal cell strain (hereinafter referred to as 293T-C3 cell) obtained in Example 1 and AAV293 are used to package serotypes AAV1, AAV2, AAV3, AAV5, AAV6, and pDG8 respectively, and measure the packaging. Viral titers of AAV. Specific steps are as follows:
[0041] (1) Plate 293T-C3 in five 150mm dishes, and then put AAV packaging plasmids (pAAV1, pAAV2, pAAV3, pAAV5, pAAV6 and pDG8 plasmids), helper plasmids (pHelper plasmids) and expression plasmids (pAAV-CB-EGFP Plasmids) were mixed at a molar ratio of 1:1:1 to obtain a mixed plasmid, which was then co-transfected into 293T-C3 cells with PEI transfection reagent, and the mass ratio of PEI transfection reagent to mixed plasmid was 1:2, 8- After 10 hours, the cells were replaced with medium, and the culture was continued until 48 hours before taking pictures u...
Embodiment 3293
[0065] Example 3 Establishment of 293T-C3 tertiary cell bank
[0066] 1. Establishment of tertiary cell bank
[0067] (1) if Figure 5 As shown, the 293T-C3 cells screened above were expanded and cultured until 1x10 7 Freeze about 30 tubes per tube, then discard the supernatant of the cells, wash with PBS, add digestion solution and digest in the incubator for 1 minute; add medium to stop digestion, resuspend and count, and centrifuge at 1000rpm for 5 minutes; 1x10 7 The cells were resuspended by adding the corresponding amount of cryopreservation solution, and then 1mL cells were added to each cryopreservation tube, placed in a programmed cooling freezer box, placed at -80°C overnight, then transferred to a gas-phase liquid nitrogen tank as a seed bank and made good record.
[0068] (2) One cell was taken out from the above-mentioned frozen seed bank cells, expanded and cultured, and then expanded and frozen according to the above method, and 50 cells were frozen as the m...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - Generate Ideas
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com