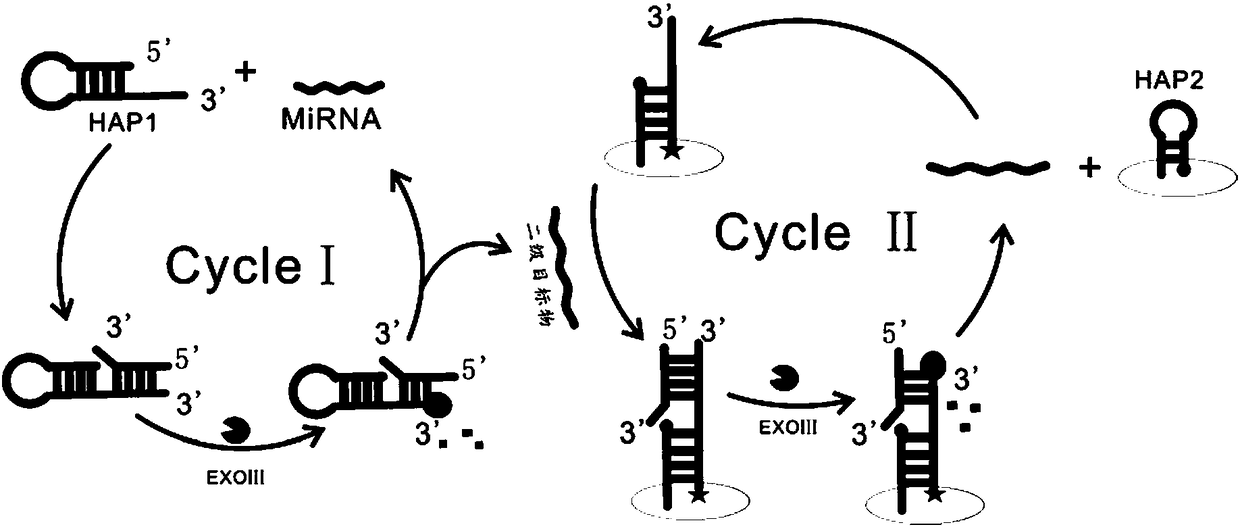
Electrochemical biosensor for detecting miRNA-122
A miRNA-122, biosensor technology, applied in the field of biosensors, can solve the problems of long time consumption, complicated instrument operation, etc., and achieve the effects of simple preparation method, good repeatability and mild reaction conditions
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
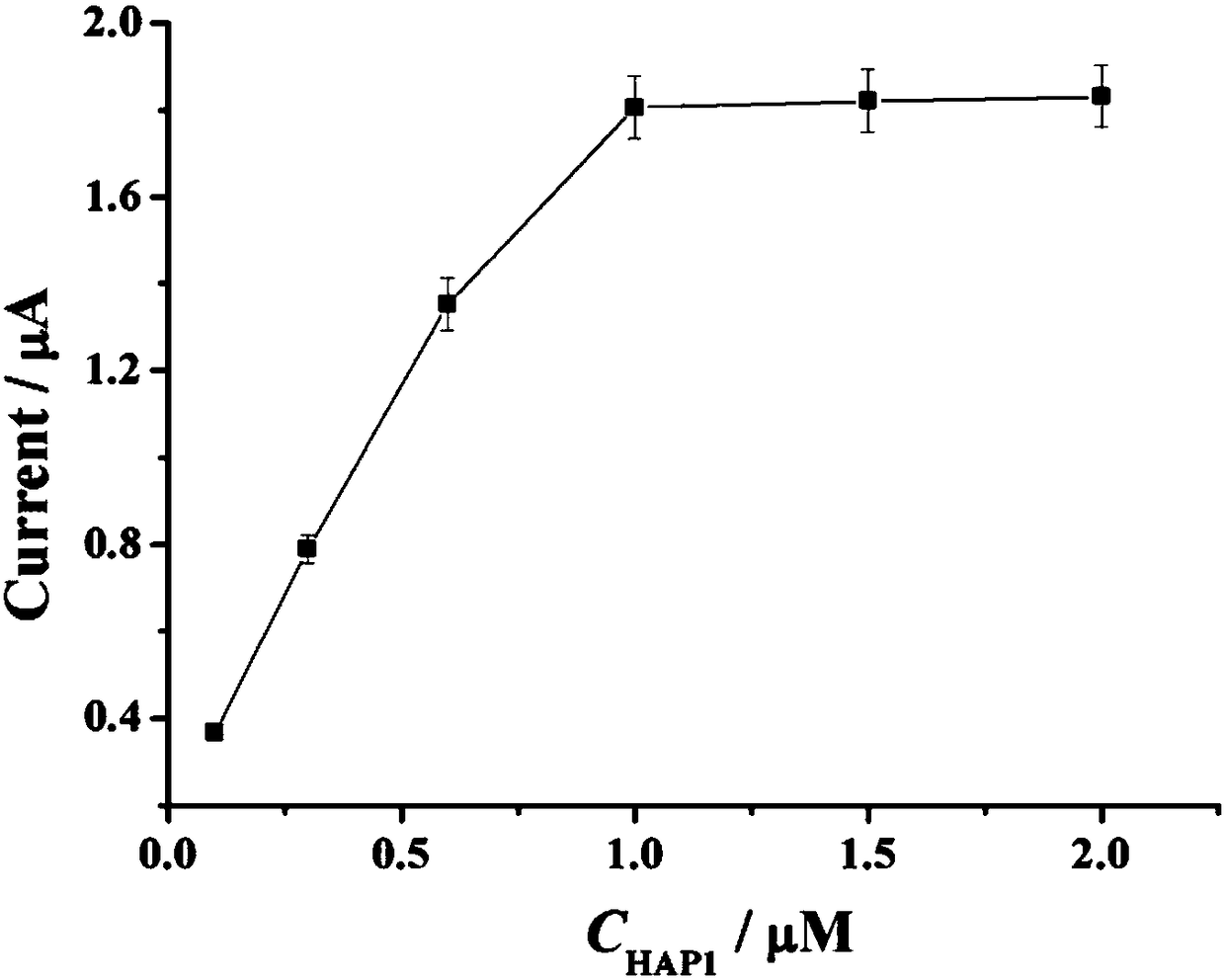
[0043] Example 1 The change of the current signal with the concentration of HAP1.
[0044] (1) The gold electrode was polished to a mirror surface in 0.3 µm and 0.05 µm alumina slurry in turn, and rinsed with PBS and sterile water;
[0045] (2) Add 10 μL of HAP2 (10 μM) dropwise to the pretreated electrode surface and incubate at 37 °C for 2 h; Incubate for 1 h;
[0046](3) Mix 12 μL sterilized water, 2 μL 10× NEBuffer, 2 μL HAP1 (final concentration 0.1 μM, 0.3 μM, 0.6 μM, 1 μM, 1.5 μM, 2 μM), 2 μL Exo Ⅲ (20 U / μL) and 2 μL miRNA-122 solution (1 pM) were added to the centrifuge tube, shaken for 30 s, and incubated in a 37 °C incubator for 2 h;
[0047] (4) Add the mixed solution in (3) dropwise onto the gold electrode modified with the HAP2-assisted probe, incubate at a constant temperature of 37 °C for 2 hours, and wash;
[0048] (5) Ag / AgCl was used as the reference electrode, the Pt electrode was used as the counter electrode, and the gold electrode obtained in step (4)...
Embodiment 2
[0050] Example 2 The change of the current signal with the concentration of HAP2.
[0051] (1) The gold electrode was polished to a mirror surface in 0.3 µm and 0.05 µm alumina slurry in turn, and rinsed with PBS and sterile water;
[0052] (2) Add 10 μL of HAP2 (1 μM, 3 μM, 6 μM, 10 μM, 15 μM, 20 μM) onto the pretreated electrode surface and incubate at 37 °C for 2 h; then add 10 μL Auxiliary probes were added dropwise to the electrode surface, and incubated at 37 °C for 1 h;
[0053] (3) Add 12 μL sterilized water, 2 μL 10× NEBuffer, 2 μL HAP1 (1 μM), 2 μL Exo III (20 U / μL) and 2 μL miRNA-122 solution (1 pM) into the centrifuge tube , shaken for 30 s, and incubated in a 37°C incubator for 2 h;
[0054] (4) Add the mixed solution in (3) dropwise onto the gold electrode modified with the HAP2-assisted probe, incubate at a constant temperature of 37 °C for 2 hours, and wash;
[0055] (5) Ag / AgCl was used as the reference electrode, the Pt electrode was used as the counter el...
Embodiment 3
[0057] Example 3 The change of current signal with the concentration of ExoIII.
[0058] (1) The gold electrode was polished to a mirror surface in 0.3 µm and 0.05 µm alumina slurry in turn, and rinsed with PBS and sterile water;
[0059] (2) Add 10 μL of HAP2 (10 μM) dropwise to the pretreated electrode surface and incubate at 37 °C for 2 h; Incubate for 1 h;
[0060] (3) Mix 12 μL sterile water, 2 μL 10× NEBuffer, 2 μL HAP1 (1 μM), 2 μL Exo III (5 U / μL, 10 U / μL, 15 U / μL, 20 U / μL, 25 U / μL, 30 U / μL) and 2 μL miRNA-122 solution (1 pM) were added to the centrifuge tube, shaken for 30 s, and incubated in a 37 °C incubator for 2 h;
[0061] (4) Add the mixed solution in (3) dropwise onto the gold electrode modified with the HAP2-assisted probe, incubate at a constant temperature of 37 °C for 2 hours, and wash;
[0062] (5) Ag / AgCl was used as the reference electrode, the Pt electrode was used as the counter electrode, and the gold electrode obtained in step (4) was used as the ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - Generate Ideas
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com