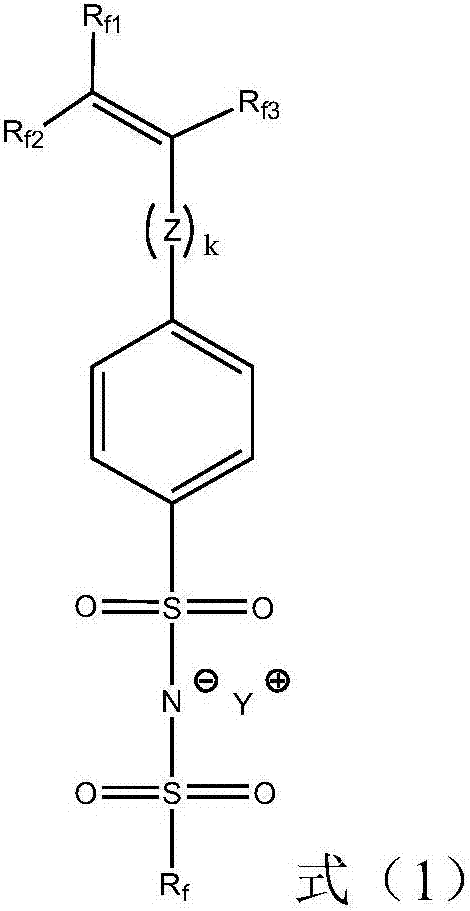
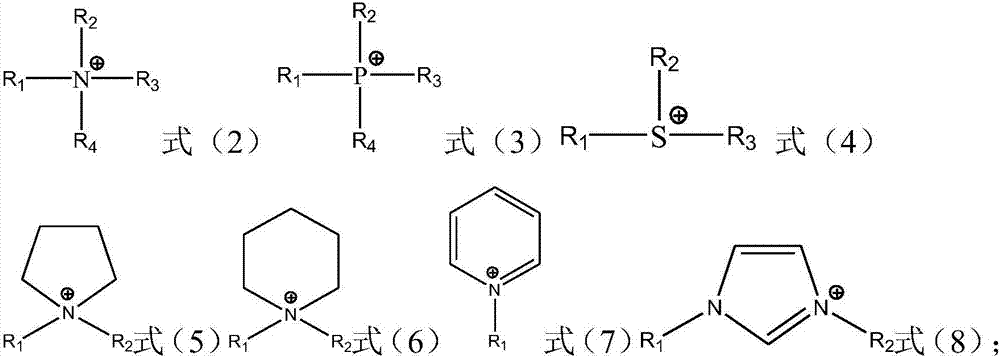
Ionic liquid compound, preparation method thereof, ionic liquid polymer and polymer solid electrolyte containing ionic liquid polymer
A technology of ionic liquids and compounds, applied in circuits, organic chemistry, electrical components, etc., can solve the problems of slow ion migration and low conductivity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
[0062] Optionally, the preparation method of the compound shown in formula (9) comprises: making the compound shown in formula (10) react with the compound shown in formula (11) or the compound shown in formula (12) under Grignard reaction conditions touch:
[0063]
[0064] Among them, Z 1 each independently for C m h 2m 、C m f 2m or (OCH 2 CH 2 ) m ;Z 2 each independently as (CH 2 CH 2 O) m or
[0065] K is each independently an integer of 1-5, and m is each independently an integer of 1-20;
[0066] R f for C h f 2h+1 , h is an integer of 0-10; R f1 , R f2 and R f3 each independently for C i h 2i+1 or C i f 2i+1 , i is an integer of 0-10.
[0067] Optionally, the molar ratio of the compound represented by the formula (10) to the compound represented by the formula (11) or the compound represented by the formula (12) may be 1: (1-1.2).
[0068] Optionally, the conditions of the Grignard reaction may be as follows: the reaction temperature is -20°...
Embodiment 1
[0164] This example is used to illustrate the preparation method of the ionic liquid compound of the present disclosure.
[0165]
[0166]
[0167] Take 1.9164g (10mmol) of p-chlorobenzenesulfonamide, 2.3794g (20mmol) of thionyl chloride and 1.3982g (12mmol) of chlorosulfonic acid at 150°C for 12h to obtain compound 1a (2.6113g, yield 90%) ; 1 H NMR (400MHz, CDCl 3 , ppm), δ=7.87(d,2×1H), 7.55(d,2×1H), 2.0(s,1H);
[0168] Take 2.9014g (10mmol) of compound 1a and 2.1451g (12mmol) of SbF 3 Reacted at 60°C for 12h to obtain compound 1b (2.4632g, yield 90%); 1 H NMR (400MHz, CDCl 3 , ppm), δ=7.87(d,2×1H), 7.55(d,2×1H), 2.0(s,1H);
[0169] Take 2.7369g (10mmol) of compound 1b and 1.3821g (10mmol) K 2 CO 3 Reacted at 25°C for 2h to obtain compound 1c (3.1178g, yield 100%); 1 H NMR (400MHz, CDCl 3 , ppm), δ=7.87(d, 2×1H), 7.55(d, 2×1H);
[0170] 3.1178g (10mmol) of compound 1c was reacted with 0.2917g (12mmol) of metal Mg at 0°C for 4h to obtain compound 1d (3.3609g, ...
Embodiment 2
[0174]
[0175] 1.8718g (10mmol) potassium trifluoromethanesulfonamide monohydrogen was reacted with 2.3218g (11mmol) p-chlorobenzenesulfonyl chloride at 80°C for 12h to obtain compound 2a (2.9133g, yield 90%); 1 H NMR (400MHz, CDCl 3 , ppm), δ=7.87(d,2×1H), 7.55(d,2×1H), 2.0(s,1H);
[0176] Take 3.2370g (10mmol) of compound 2a and 1.3821g (10mmol) K 2 CO 3 Reacted at 25°C for 2h to obtain compound 2b (3.6179g, yield 100%); 1 H NMR (400MHz, CDCl 3 , ppm), δ=δ=7.87(d, 2×1H), 7.55(d, 2×1H);
[0177] 3.6179g (10mmol) of compound 2b was reacted with 0.2917g (12mmol) of metal Mg at 0°C for 4h to obtain compound 2c (3.8610g, yield 100%); 1 H NMR (400MHz, CDCl 3 , ppm), δ=δ=7.9(d, 2×1H), 7.5(d, 2×1H);
[0178] 3.8610g (10mmol) of compound 2c was reacted with 1.0866g (12mmol) of 4-chloro-1-butene at 0°C for 4h to obtain compound 2d (3.4329g, yield 90%); 1 H NMR (400MHz, CDCl 3 , ppm), δ=7.88(d,2×1H), 7.40(d,2×1H), 5.70(m,1H), 5.03(q,1H), 4.97(q,1H), 2.59(t,2H ), 2.29(m,2H...
PUM
| Property | Measurement | Unit |
|---|---|---|
| thickness | aaaaa | aaaaa |
| glass transition temperature | aaaaa | aaaaa |
| decomposition temperature | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com