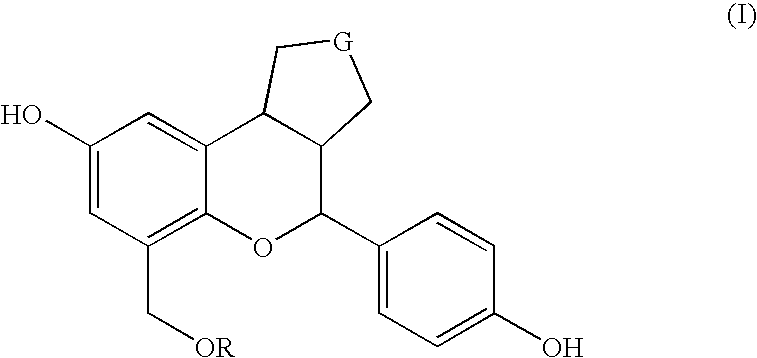
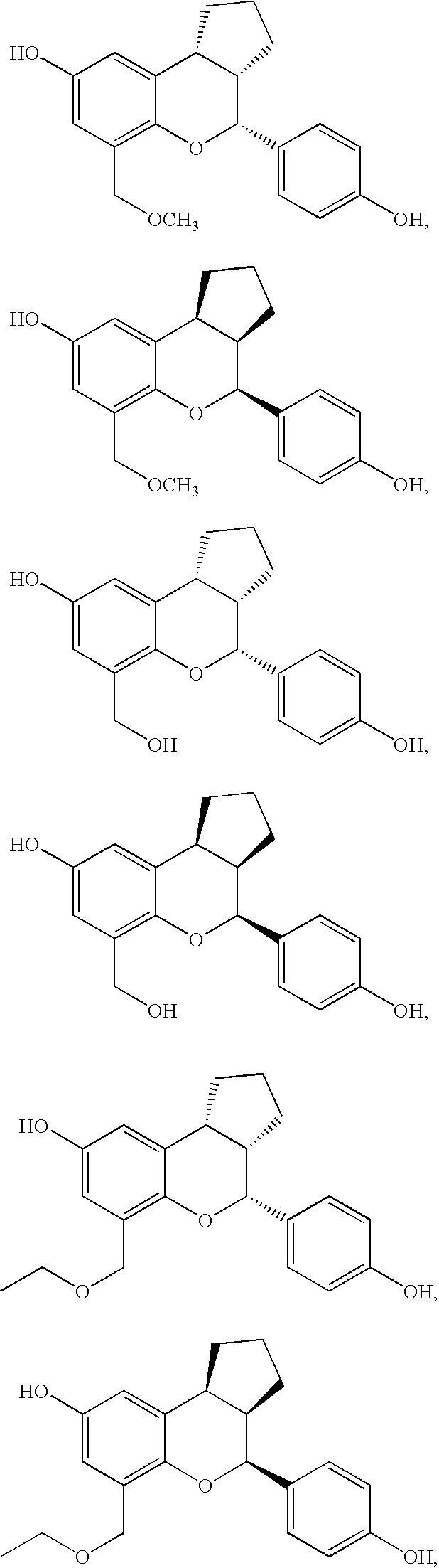
Substituted benzopyrans as selective estrogen receptor-beta agonists
a technology of benzopyran and benzopyran, which is applied in the direction of heterocyclic compound active ingredients, biocide, drug compositions, etc., can solve the problems of increased urination frequency, poor urine stream, hesitation or delay in starting the urine flow,
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
preparation 1
Preparation of 1,3-dibromo-2,5-bis-methoxymethoxy-benzene
[0042]
A. Preparation of 2,6-dibromo-4-methoxy-phenol
[0043]
[0044]To 4-methoxyphenol (10.00 g, 80.6 mmol) in CH2Cl2 (70 mL), add a stirred solution of benzyltrimethylammonium tribromide (69.10 g, 177.2 mmol) in CH2Cl2 (500 mL) and MeOH (200 mL). Stir the reaction for 12 hours and remove the solvent in vacuo. Add methyl tert-butyl ether (MTBE) (600 mL) and collect the precipitate (benzyltrimethylammonium bromide) by filtration and wash with MTBE (200 mL). Filter the organics through Celite® and concentrate to provide the subtitled product (23.0 g, >95%) as an orange solid that may be used without purification. 1H NMR (DMSO-d6) δ 3.69 (s, 3H), 7.14 (s, 2H), 9.32 (s, 1H).
B. Preparation of 2,6-dibromo-benzene-1,4-diol
[0045]
[0046]Dissolve 2,6-dibromo-4-methoxyphenol (23.0 g, 80.6 mmol) in CH3CN (400 mL) and add trimethylsilyl iodide (TMSI) (53 mL, 371 mmol). Reflux the solution for 4 hours and add more TMSI (50 mL, 371 mmol). Heat th...
preparation 2
Preparation of 2-trifluoromethanesulfonyloxy-cyclopent-1-enecarboxylic acid methyl ester
[0049]
[0050]Slowly add a solution of methyl 2-oxocyclopentanecarboxylate (21.0 g, 148 mmol) in CH2Cl2 (30 mL) to a −78° C. solution of N,N,-diisopropylethylamine (DIPEA) (129 mL, 738 mmol) in CH2Cl2 (670 mL). Keep the reaction temperature below −65° C. Then slowly add a solution of trifluoromethanesulfonic anhydride (50.0 g, 177 mmol) in CH2Cl2 (30 mL) to the reaction, keeping the reaction temperature below −65° C. After 1.5 hours, quench the reaction with H2O (500 mL) and warm to room temperature. Separate the layers and extract the aqueous layer with CH2Cl2 (2×100 mL). Combine the organic extracts, dry (Na2SO4), filter and concentrate. Pre-absorb the crude material onto silica gel. Purify using silica gel chromatography, eluting with Hex:EtOAc (90:10) to provide the title product (34.3 g, 85%) as a light yellow oil. 1H NMR (CDCl3) δ 1.97-2.07 (m, 2H), 2.68-2.78 (m, 4H), 3.79 (s, 3H). CIMS (Meth...
preparation 3
Preparation of 1-Bromo-4-methoxymethoxy-benzene
[0051]
[0052]Add sodium hydride (2.78 g of 95% dry, 0.116 mol) in portions to a 500 mL three-necked flask equipped with a nitrogen inlet, magnetic stir bar and DMF (200 mL) at −10 to −15° C. Add a solution of 4-bromophenol (20.00 g, 0.116 mol) in DMF (100 mL) over about 20 minutes, maintaining the internal temperature of the reaction below −5° C. Stir the reaction for 1 hour and then add MOMCl (8.81 mL, 0.116 mol) in portions to maintain the internal temperature below −5° C. Stir the mixture for 3 hours and then add ice until gas evolution ceases. Pour the reaction into ice-H2O (500 mL) and extract with EtOAc (2×300 mL). Wash the combined organic extracts with 1 N NaOH (500 mL), H2O (500 mL) and brine (400 mL). Dry the solution (MgSO4), filter and concentrate to obtain the title product (23.94 g, 93%) as an oil which may be used without further purification. 1H NMR (CDCl3) δ 3.46 (s, 3H), 5.14 (s, 2H), 6.92 (d, J =9.0 Hz, 2H), 7.37 (d, J...
PUM
| Property | Measurement | Unit |
|---|---|---|
| temperature | aaaaa | aaaaa |
| temperature | aaaaa | aaaaa |
| temperature | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com