Sulodexide for use in the treatment of pathologies wherein metalloproteinases are involved
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
[0139]Process to Obtain Dermatan Sulfate Fraction from Sulodexide
[0140]A quantity of sulodexide extracted from intestinal animal mucosa, corresponding to 8 mg was dissolved in a binding buffer constituted by TRIS 50 mM and NaCl 50 mM at pH 7.4, and loaded on an affinity column prepared by coupling 7.5 mg of ATIII / ml of CNBr-activated Sepharose gel (GE).
[0141]The column was washed with the same buffer solution and the absorbed material was eluted with a buffer solution consisting of TRIS 50 mM, NaCl 3M at pH 7.4.
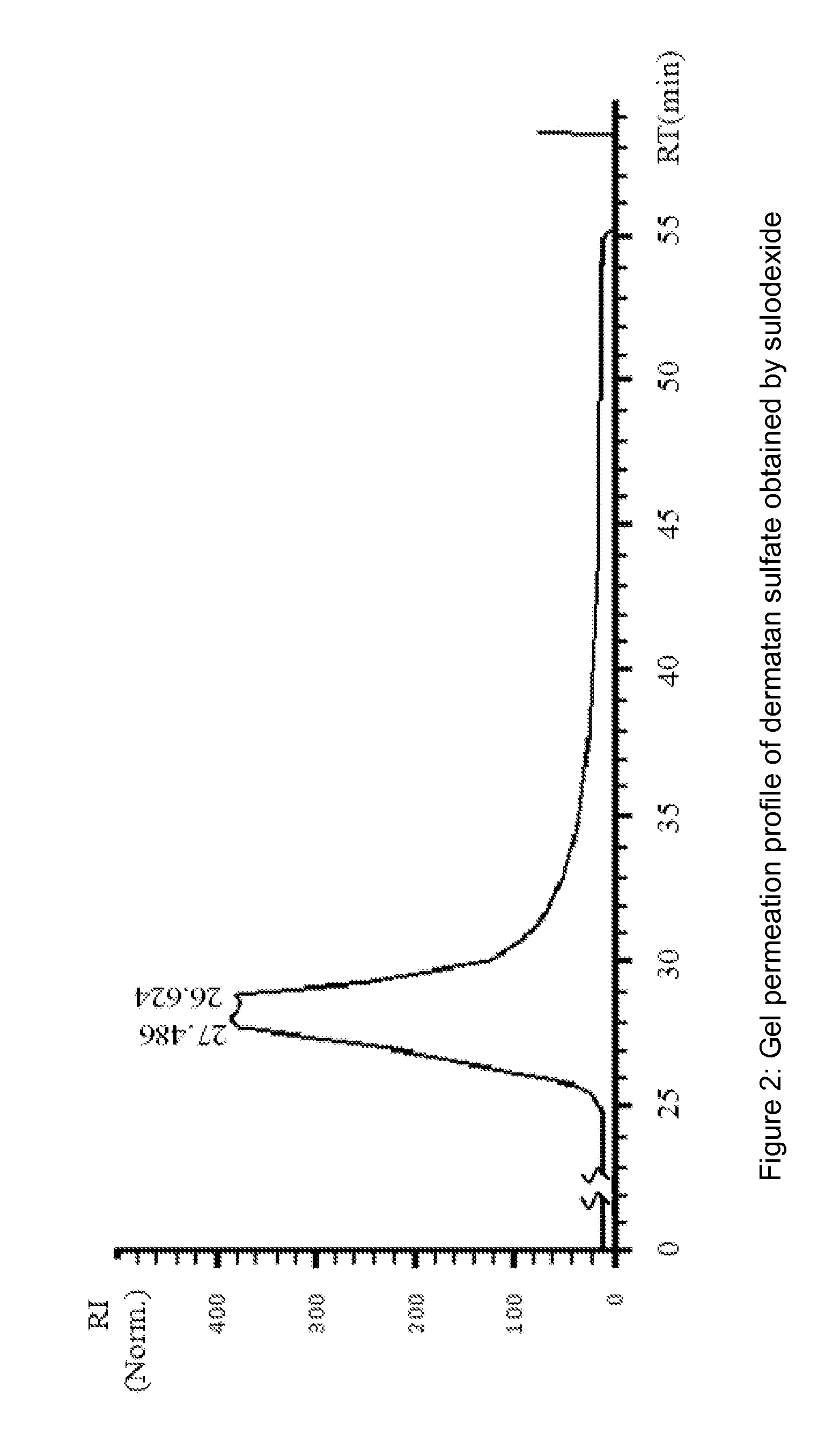
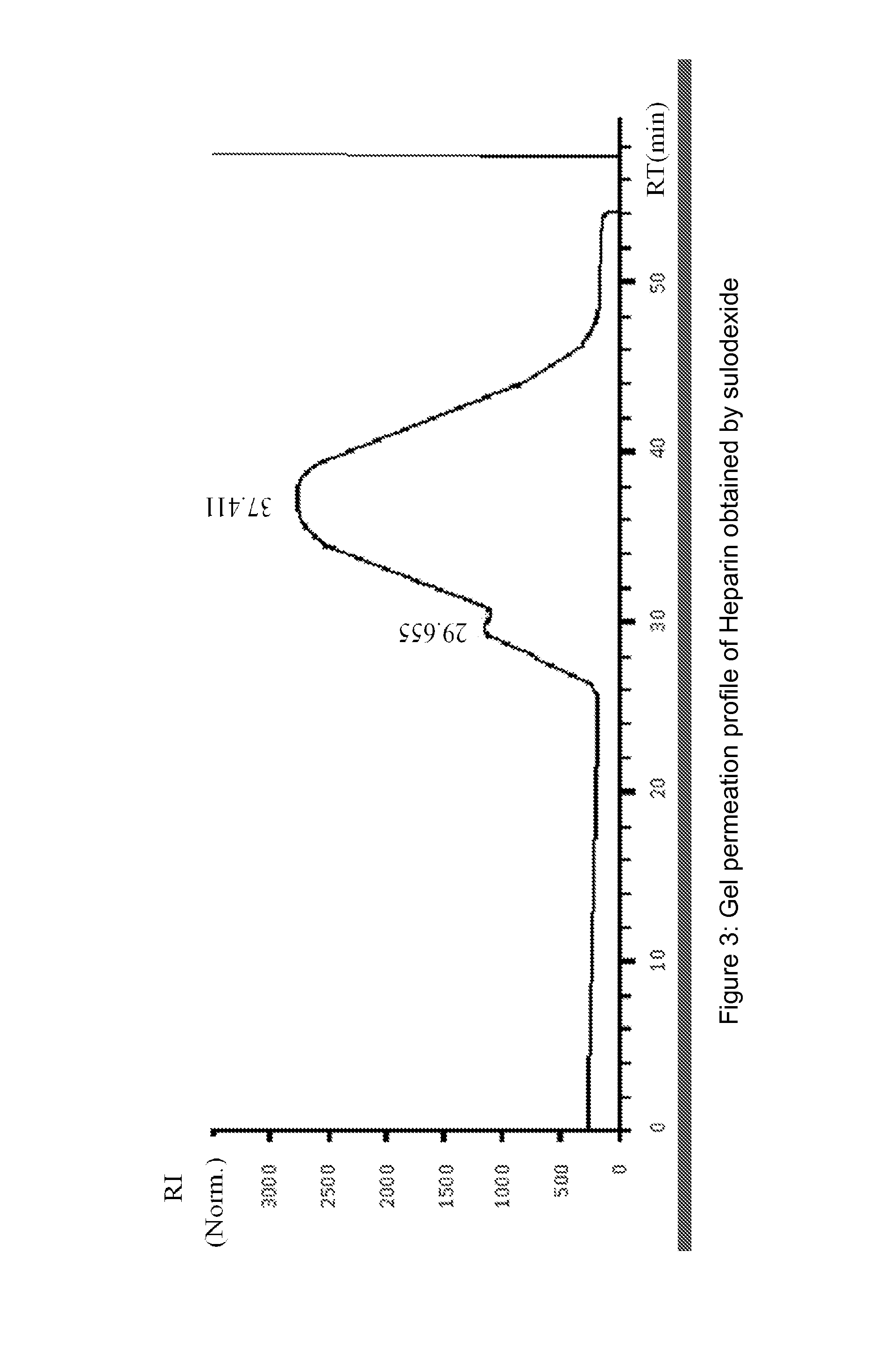
[0142]Dermatan sulfate fraction was collected in the non absorbed material present in the flow-through, while heparin fraction, is collected in the eluted fractions.
[0143]Dermatan sulfate and heparin fraction could be stored at −20° or optionally lyophilized.
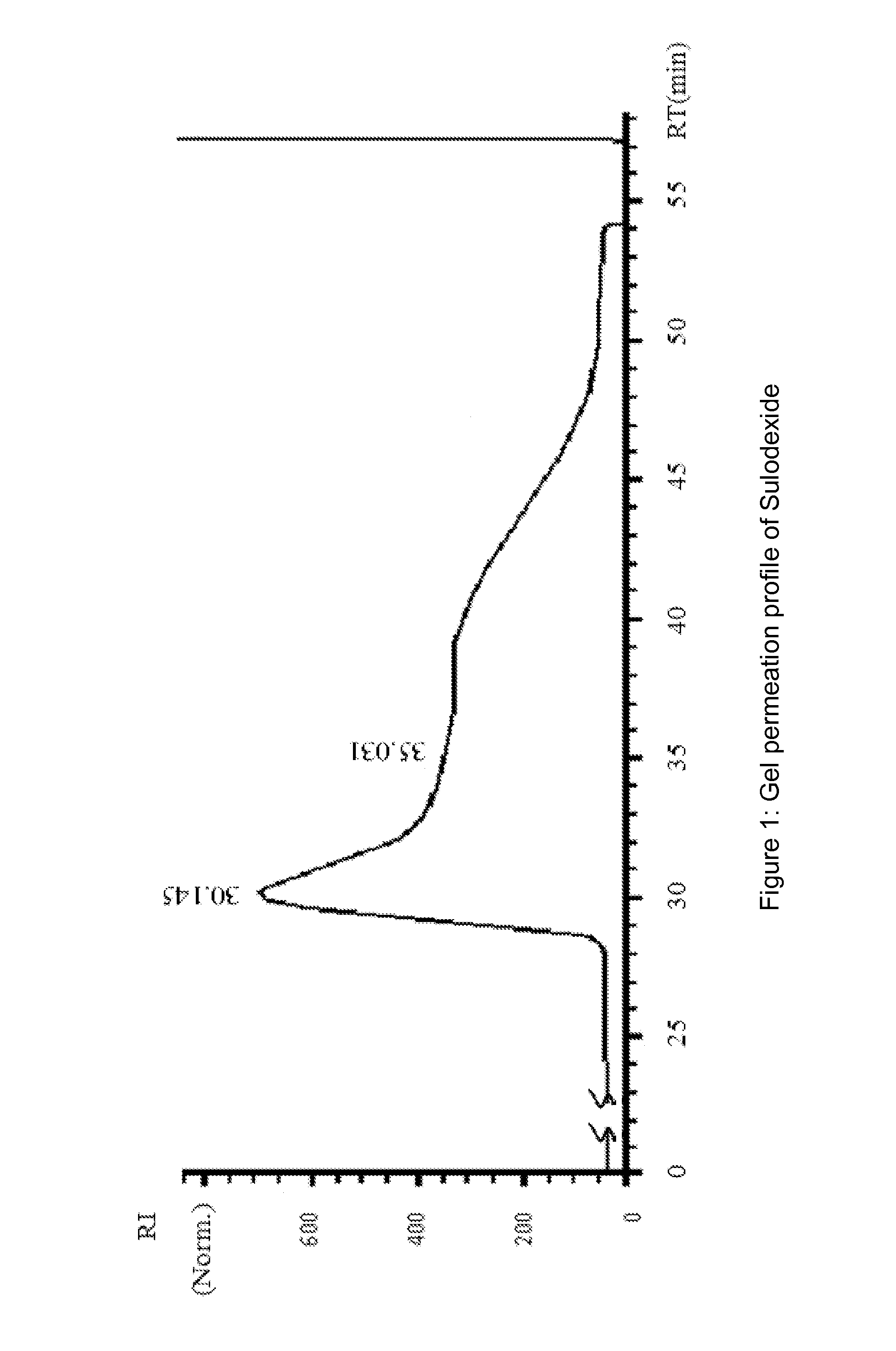
[0144]Heparin pooled fractions and dermatan sulfate pooled fractions were analyzed in gel permeation chromatography (GPC) and in agarose gel.
[0145]Chromatographic profiles were obtained in HPLC system HP 1090, equipped wi...
example 2
[0151]Effect of sulodexide on circulating MMPs in blood samples
[0152]Samples of blood collected from 50 healthy volunteers, 25 female and 25 male of age between 25 and 58, average age of 37 years, were placed in test tubes and two sets of samples were prepared; in the first set were added variable amount of sulodexide until to obtain final concentration corresponding to 0, 12, 24, 48 μg / ml, in the second set were added variable amounts of dermatan obtained as in Example 1, up to the final concentration corresponding to 0, 2.5, 5, and 10 μg / ml. The samples without the addition of sulodexide and dermatan were used as control and the bands of MMPs, of these samples were attributed the arbitrary value of intensity of 100%. The intensity of the MMPs bands of the treated samples were reported as percentage in respect to the untreated samples.
[0153]Overall samples were centrifuged at 1550 g for 10 minutes at 4° C., an amount of supernatants of serum samples (S) corresponding to 150 μg of t...
example 3
[0158]Effect of Sulodexide on the Amounts of Circulating MMPs in the Plasma
[0159]Samples of blood are collected from 50 healthy volunteers, 25 female and 25 male of age between 25 and 58, average age of 37, and 1 ml of each sample is added to 38 μl of a solution of sodium citrate 0.13 M and centrifuged at 1550 g for 10 minutes at 4° C.
[0160]Increased amounts of sulodexide are added to the samples up to obtain final concentration corresponding respectively to 0, 12, 24 and 48 μg / ml.
[0161]An amount corresponding to 150 μg of total proteins of supernatants of plasma samples (P) obtained by centrifugation, measured through Bradford method, is analyzed by zymography-gelatin. The standard of gelatinase, prepared diluting the capillary blood from healthy volunteers by 15 volumes of Laemmli buffer, in non reducing conditions, are loaded in the gel.
[0162]The gelatin-zymograms and the electrophoresis rune are executed as described in Example 2.
[0163]The zymograms areas of the band are determi...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Fraction | aaaaa | aaaaa |
| Fraction | aaaaa | aaaaa |
| Mass | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com