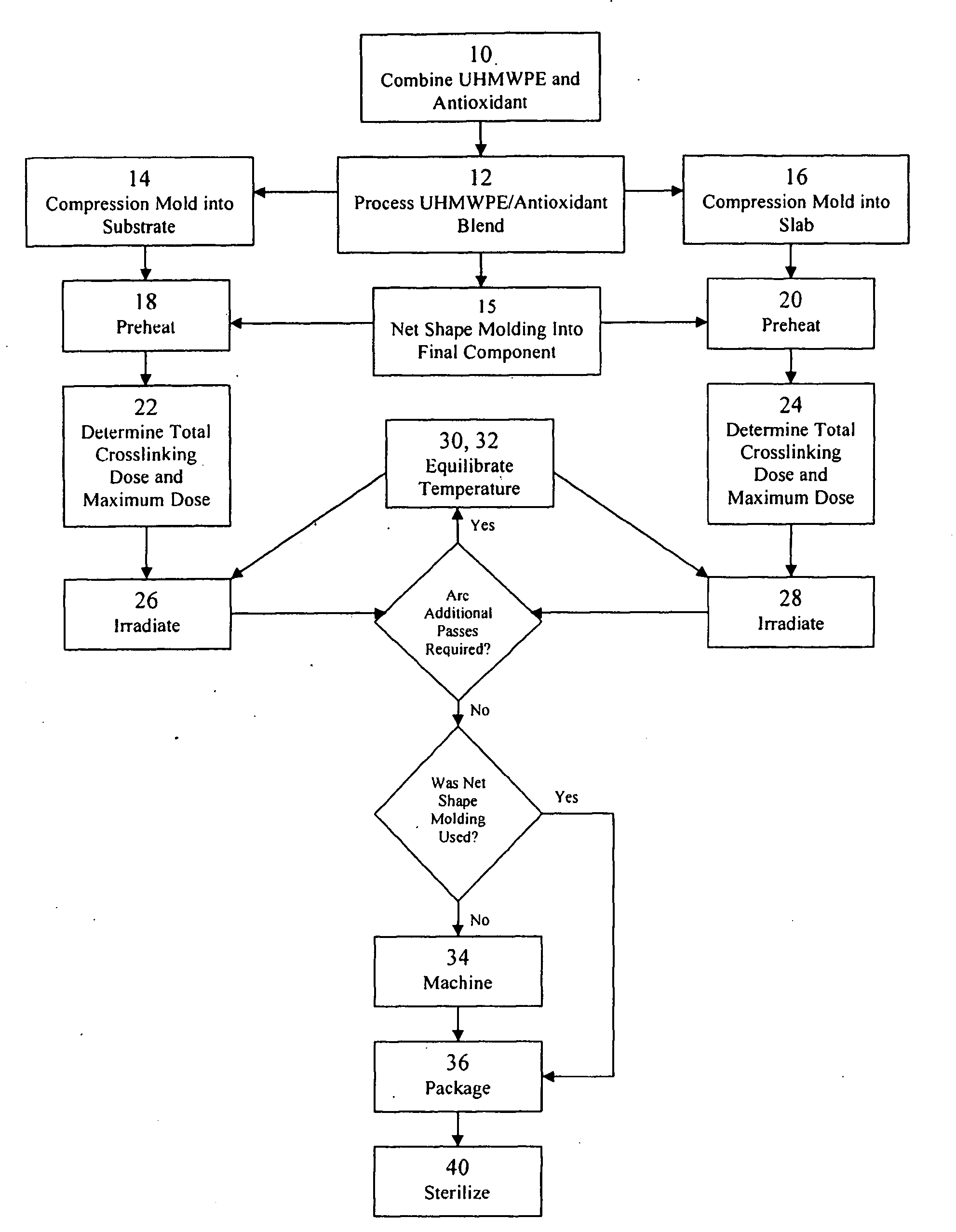
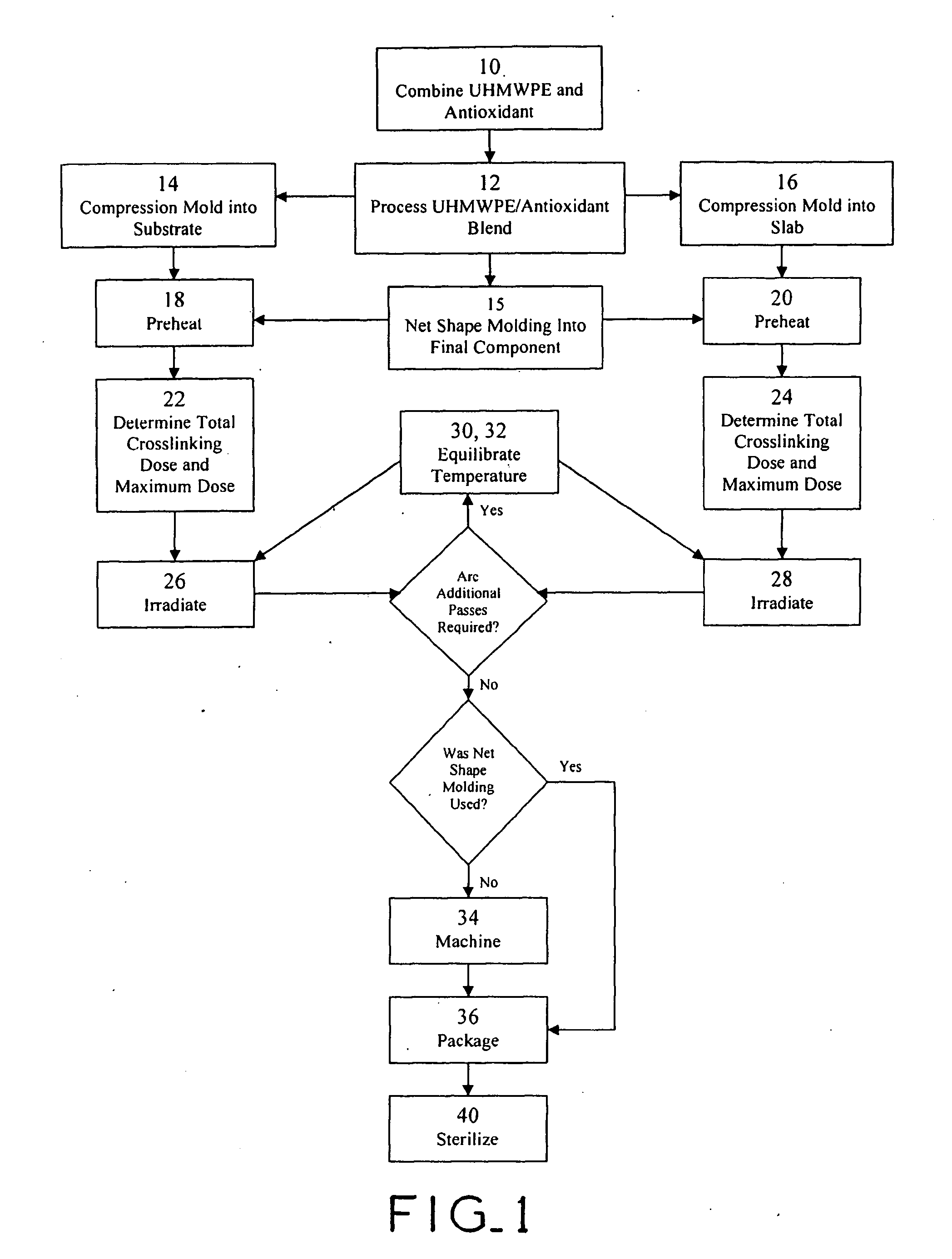
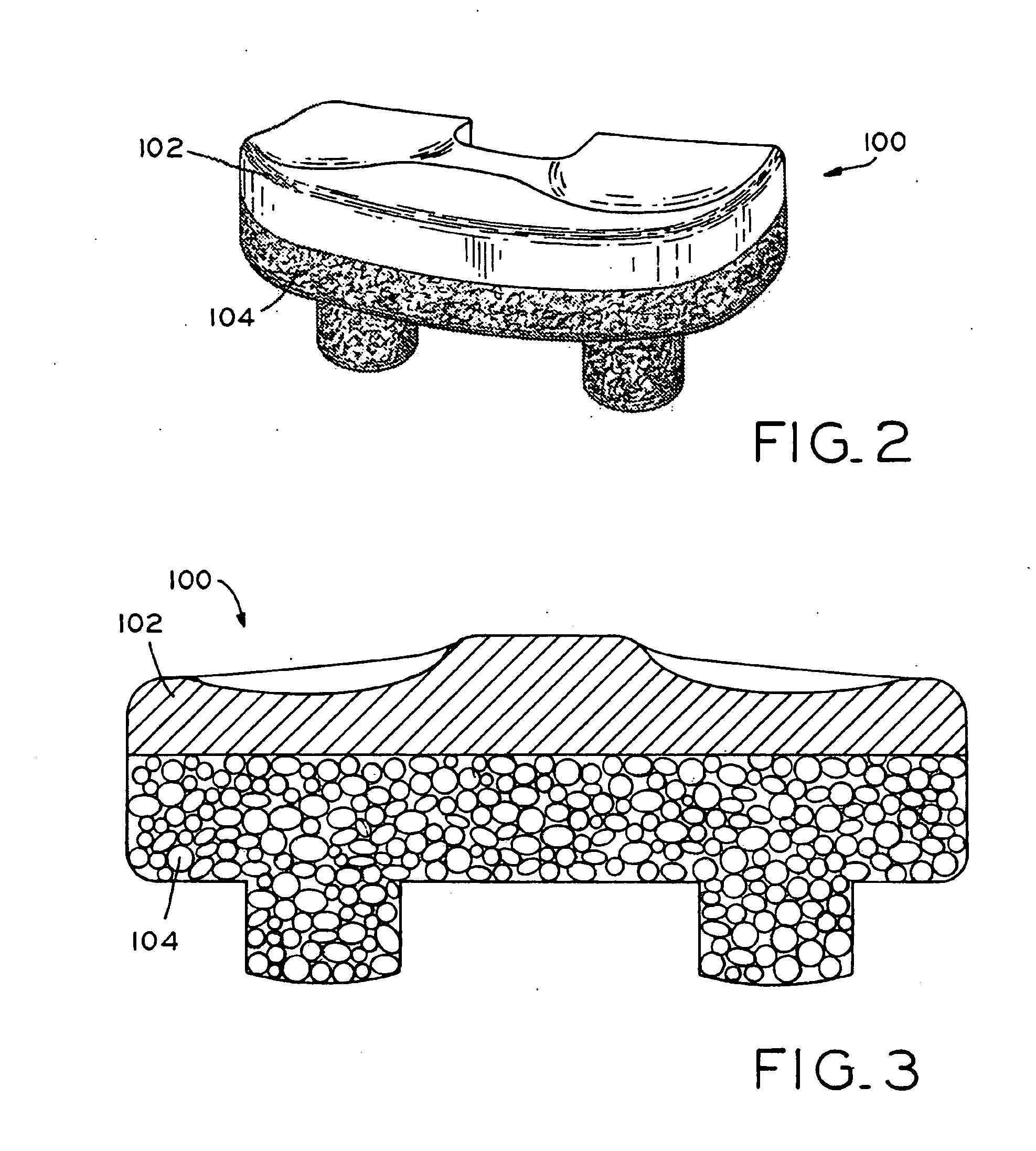
Antioxidant stabilized crosslinked ultra-high molecular weight polyethylene for medical device applications
a technology of polyethylene and antioxidant, applied in the field of crosslinked ultra-high molecular weight polyethylene, can solve the problems of increasing the wear rate, reducing the likelihood of additional oxidation, and unlikely to substantially alter any desired material properties, so as to achieve the desired mechanical properties, prevent substantial melting of the uhmwpe, and encourage the mobility of individual polyethylene molecules
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Feasibility Study of α-Tocopherol Acetate
[0046]The feasibility of blending α-tocopherol acetate with UHMWPE was investigated. α-tocopherol acetate was obtained from DSM Nutritional Products, Ltd. of Geleen, Netherlands and medical grade UHMWPE powder GUR 1050 was obtained from Ticona, having North American headquarters located in Florence, Ky. Isopropanol was then added to the α-tocopherol acetate as a diluent and the α-tocopherol acetate was solvent blended with the UHMWPE powder. The blending continued until two different UHMWPE / α-tocopherol acetate blends were obtained, one UHMWPE blend having 0.05 wt. % α-tocopherol acetate and the other UHMWPE blend having 0.5 wt. % α-tocopherol acetate. Each of the UHMWPE blends were then compression molded to form four one-inch-thick pucks. Two pucks of each UHMWPE blend, i.e., two pucks of the UHMWPE blend having 0.05 wt. % α-tocopherol acetate and two pucks of the UHMWPE blend having 0.5 wt. % α-tocopherol acetate, were preheated to 120° C....
example 2
Chemical Properties of UHMWPE Blended with Tocopherol
[0050]The chemical properties of d / 1-α-tocopherol mechanically blended with a UHMWPE powder which was slab molded into bars and electron beam irradiated were investigated. To perform this investigation, Design Expert 6.0.10 software, obtained from Stat-Ease, Inc. Minneapolis, Minn., was utilized to setup a modified fractional factorial Design of Experiment (DOE). The DOE evaluated five different variables: UHMWPE resin type, wt. % of d / 1-α-tocopherol, preheat temperature, dose rate, and irradiation dose.
[0051]GUR 1050 and GUR 1020 medical grade UHMWPE powders were obtained from Ticona, having North American headquarters in Florence, Ky. d / 1-α-tocopherol was obtained from DSM Nutritional Products, Ltd. of Geleen, Netherlands. The GUR 1050 and GUR 1020 were separately mechanically blended with the d / 1-α-tocopherol by low intensity blending using a Diosna P100 Granulator, available from Diosna GmbH of Osnabrück, Germany, a subsidiary...
example 3
Free Radical Concentrations in UHMWPE Blended with d / 1-α-Tocopherol
[0054]The impact of mechanically blending d / 1-α-tocopherol with UHMWPE powder on free radical concentration of electron beam irradiated UHMWPE blend molded pucks was investigated. To perform this investigation, Design Expert 6.0.10 software, obtained from Stat-Ease, Inc. Minneapolis, Minn., was utilized to setup a modified central composite Design of Experiment (DOE). The DOE evaluated five factors: preheat temperature, dose rate, irradiation dose, d / 1-α-tocopherol concentration, and predetermined hold time, i.e., the time elapsed between removal of the UHMWPE blend from the oven until the initiation of electron beam irradiation.
[0055]GUR 1050 medical grade UHMWPE powder was obtained from Ticona, having North American headquarters in Florence, Ky. d / 1-α-tocopherol was obtained from DSM Nutritional Products, Ltd. of Geleen, Netherlands. The GUR 1050 UHMWPE power was mechanically blended with the d / 1-α-tocopherol by hi...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Temperature | aaaaa | aaaaa |
| Temperature | aaaaa | aaaaa |
| Time | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com