Combination of sedative and a neurotransmitter modulator, and methods for improving sleep quality and treating depression
a neurotransmitter and sleep quality technology, applied in the field of combination of sedatives and neurotransmitter modulators, and methods for improving sleep quality and treating depression, can solve the problems of limiting the usefulness of certain patient populations, high potential for addiction involving both physical and psychological dependence, and development of tolerance, so as to increase the effect of antidepressant therapy
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
example 1
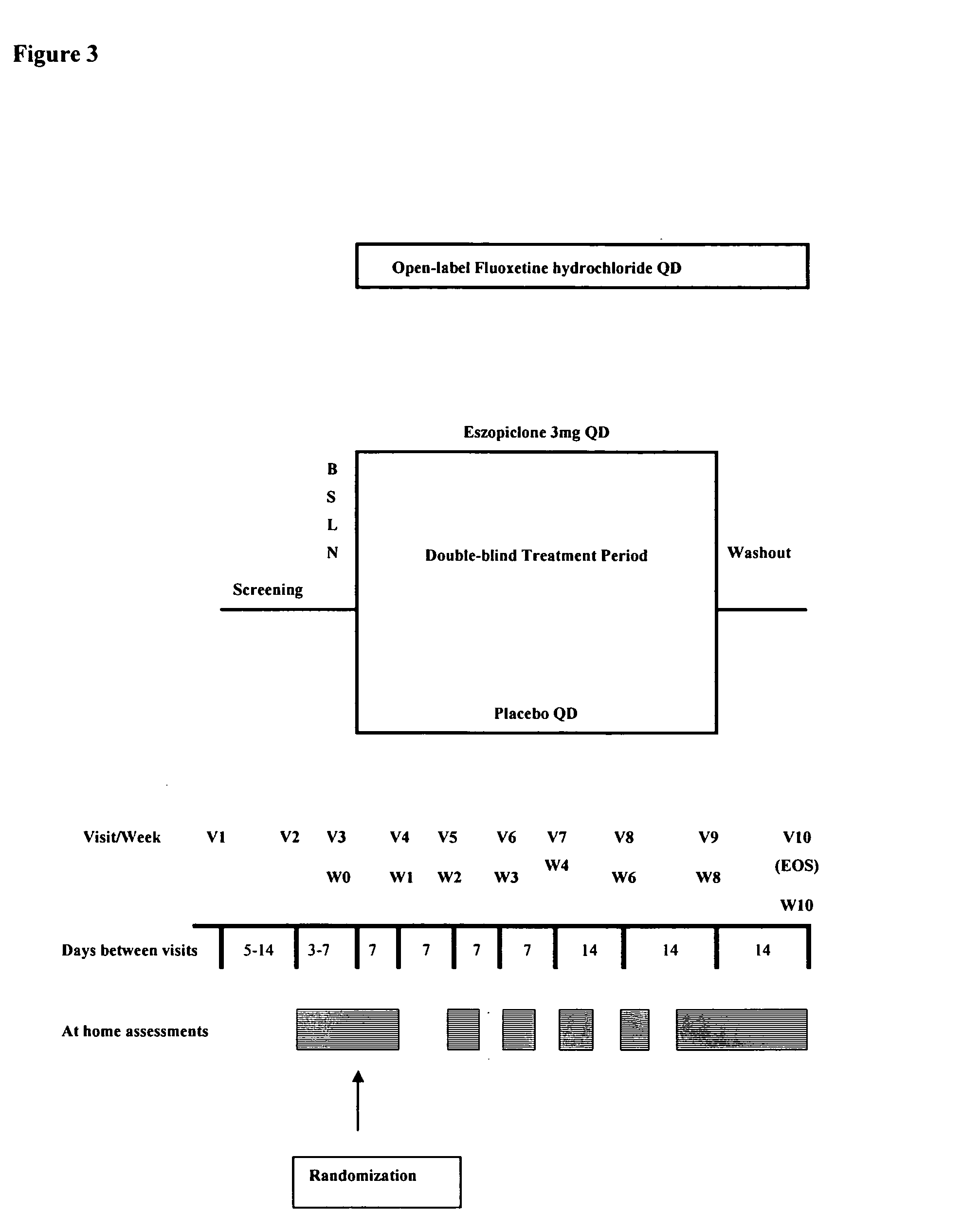
[0852] Depression Response to Eszopiclone in Adults With Major Depressive Disorder (DREAMDD): A Randomized, Double-Blind, Placebo-Controlled, Parallel-Group, 8-Week, Safety and Efficacy Study of Eszopiclone 3 mg Compared to Placebo in Subjects with Insomnia Related to Major Depressive Disorder
Rationale & Objectives
[0853] Insomnia often occurs simultaneously with the onset of major depression, and may negatively impact the depressed subject's quality of life (QoL) and functional abilities. In fact, in some patients, insomnia is the most disabling of the depressive symptom complex. In addition, many subjects are treated with selective serotonin re-uptake inhibitors (SSRIs) that may accentuate insomnia initially following treatment with these agents. This study aims to establish that the early addition of continuous eszopiclone 3 mg at bedtime to a standard antidepressant treatment with fluoxetine hydrochloride (20 or 40 mg) daily in the morning for 8 weeks is safe and provides impr...
example 2
Adjunctive Eszopiclone with Fluoxetine for MDD and Insomnia: Sleep Effects
[1159] Insomnia and depression often co-exist. This study evaluated the efficacy of eszopiclone for insomnia associated with MDD during concurrent fluoxetine treatment.
[1160] Methods: Patients (n=545) met DSM-IV criteria for MDD and insomnia, including reported sleep latency (SL)≧30 min (median 73 min), wake time after sleep onset (WASO)≧45 min (median 90 min), and total sleep time (TST)≦6.5 h (median 294 min). All patients received fluoxetine QAM, and were randomly assigned to double-blind treatment with eszopiclone 3 mg or placebo QHS for 8 weeks. Subjective sleep and daytime function were assessed weekly.
[1161] Results: Compared to placebo, eszopiclone was associated with significantly lower SL and greater TST at each treatment week (p<0.03); significantly lower WASO at Weeks 1, 3-5, and 7-8 (p<0.04); higher ratings across the treatment period in sleep quality and depth (p<0.005); and higher ratings of ...
example 3
Adjunctive Eszopiclone and Fluoxetine in MDD & Insomnia: Depression Effects
[1163] Insomnia frequently co-exists with depression. This study evaluated eszopiclone and fluoxetine co-administration in depressed patients with co-morbid insomnia.
[1164] Methods: Patients who met DSM-IV criteria for new MDD and insomnia received fluoxetine 20 mg QAM plus either eszopiclone 3 mg (n=275) or placebo (n=270) nightly for 8 weeks. Efficacy was assessed using HAMD17 and Clinical Global Impression Improvement (CGI-I) and Severity (CGI-S). Response=50% decrease from baseline HAMD17; remission=HAMD17≦7.
[1165] Results: Eszopiclone co-administration resulted in significantly greater changes in HAMD17 scores at Week 4 (−9.9 vs −8.5 for placebo, p=0.02) with progressive improvement at Week 8 (−13.8 vs −11.8, p<0.001). At Week 8, significantly more eszopiclone patients were responders (74% vs 61%, p<0.009) and remitters (54% vs 41%, p<0.02). Even with removal of insomnia items, significant difference...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com