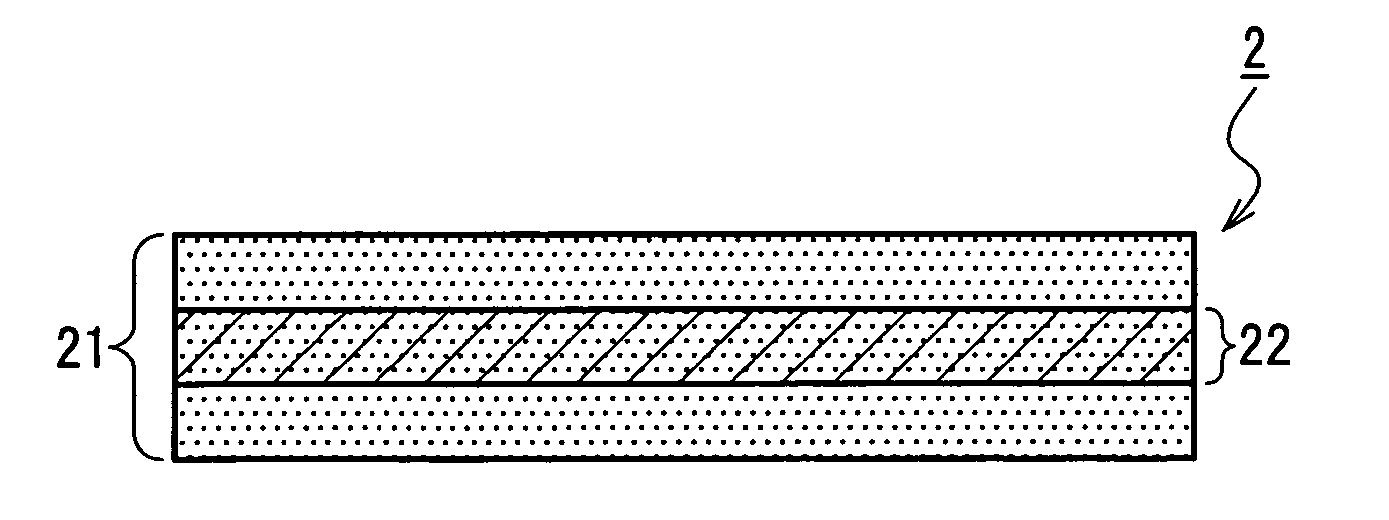
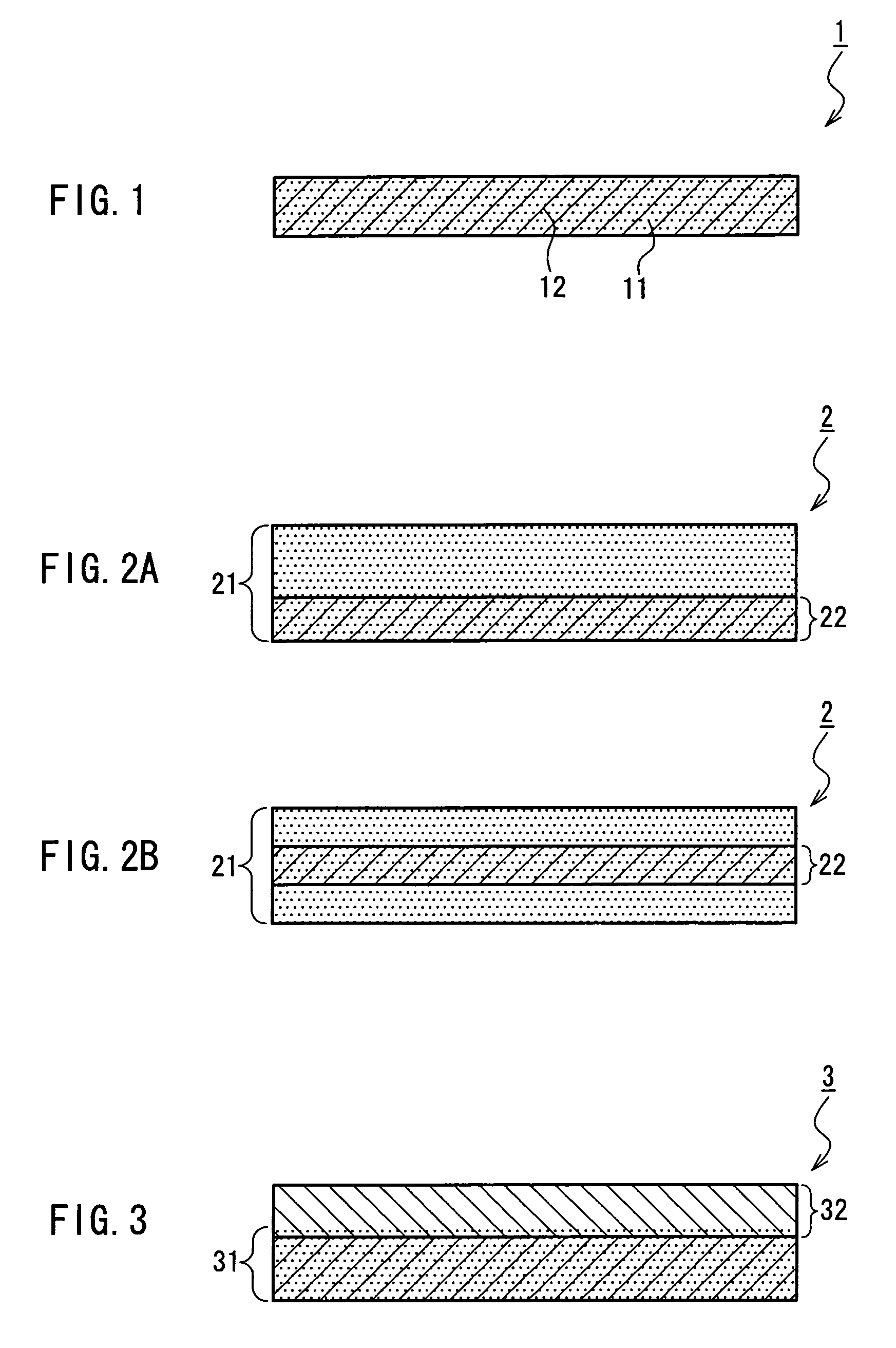
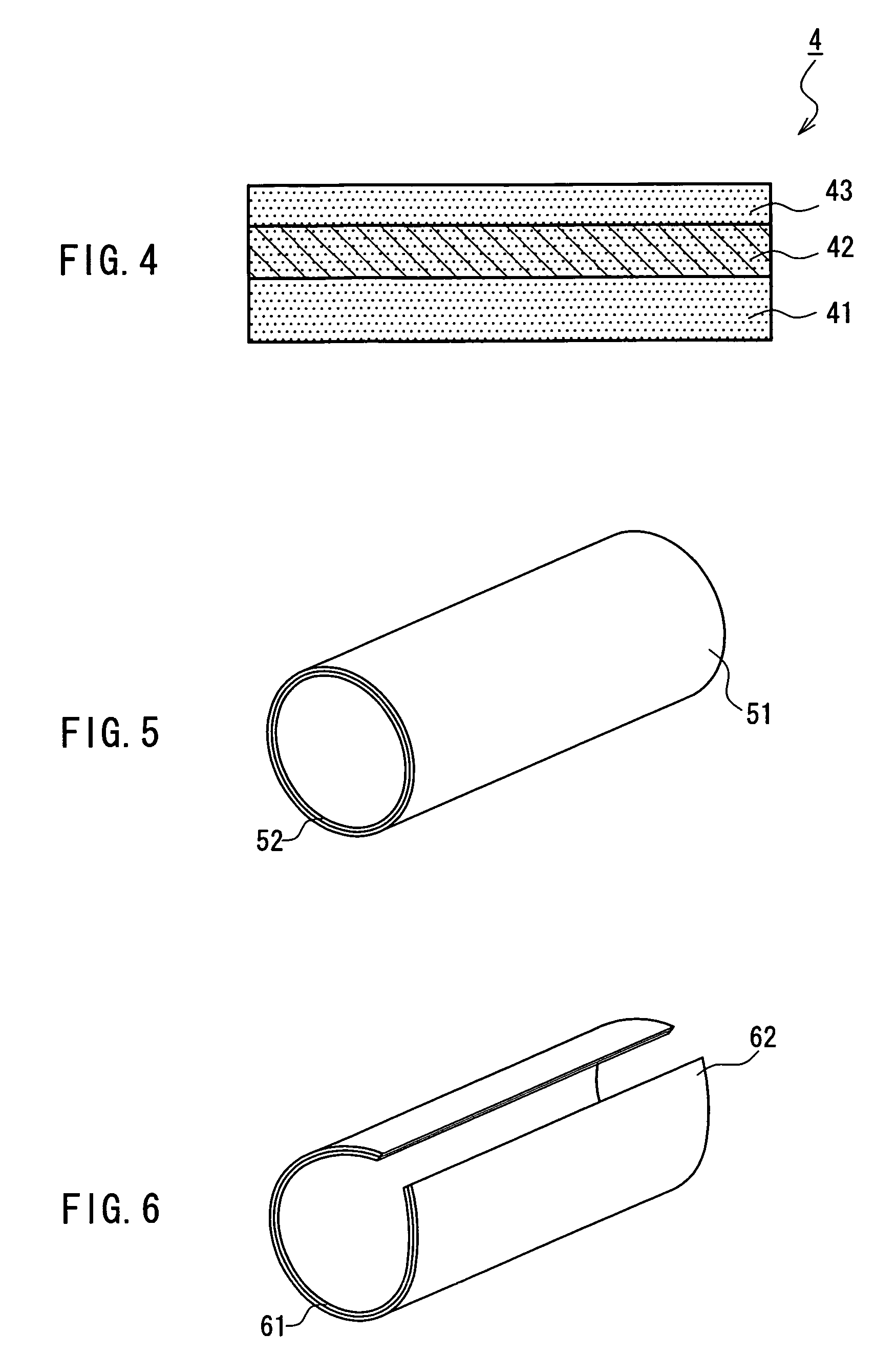
Medical film
a medical film and film technology, applied in the field of medical film, can solve the problems of pain or impair function, difficult follow-up operation with respect to primary disease, and the living body adhesion to one, and achieve the effects of excellent biocompatibility, excellent strength in suturing and bonding
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Manufacture of Fabric Body
[0106]Using a lactic acid-caprolactone copolymer multifilament yarn (thickness: 42 decitex (dtex)), a twin loop knit (thickness: 200 μm) in which a vertical length and a horizontal length of a unit of stitches were both 3.5 mm and a twin loop knit (thickness: 200 μm) in which a vertical length and a horizontal length of a unit of stitches were both 1.5 mm were prepared. The “dtex” (decitex: 1 dtex=1.111×1 denier) is a thickness unit according to the International System of Units (SI). As described above, FIG. 7 illustrates a schematic diagram of a knit stitch structure of a twin loop knit. Each of the twin loop knit fabric bodies was held between two glass plates and subjected to a vacuum heat treatment at 120° C. for 3 hours. Subsequently, each of the heat-treated twin loop knit fabric bodies was subjected to a plasma treatment at room temperature, in oxygen gas at 67 Pa (0.5 torr), with 50 W, for 30 seconds.
Integration with Gelatin Film
[0107]Each of the f...
example 2
[0121]As fabric bodies, complex films were prepared using a twin knit and a warp knitted fabric shown below, respectively, and the strength of the thus-obtained films were determined. Note here that a lactic acid-caprolactone copolymer multifilament yarn used for the preparation of the films was prepared using lactic acid-caprolactone copolymer containing lactide (a dimer of lactic acid) and caprolactone at a composition ratio (a molar ratio) of 75:25 by a known method (see JP 8(1996)-317968 A, for example).
Reinforcing Material
[0122](Twin Knit 2-1)
[0123]Using a lactic acid-caprolactone copolymer multifilament yarn (thickness: 75 decitex (dtex)), a twin loop knit in which a vertical length of a unit of stitches was 2.7 mm and a horizontal length of the same was 3.1 mm was prepared. FIG. 10A is a photograph showing a knit stitch structure of the twin knit 2-1 (25× magnification).
[0124](Twin Knit 2-2)
[0125]Using a lactic acid-caprolactone copolymer multifilament yarn (thickness: 67 dec...
PUM
| Property | Measurement | Unit |
|---|---|---|
| horizontal length | aaaaa | aaaaa |
| thickness | aaaaa | aaaaa |
| yarn threading tension | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com