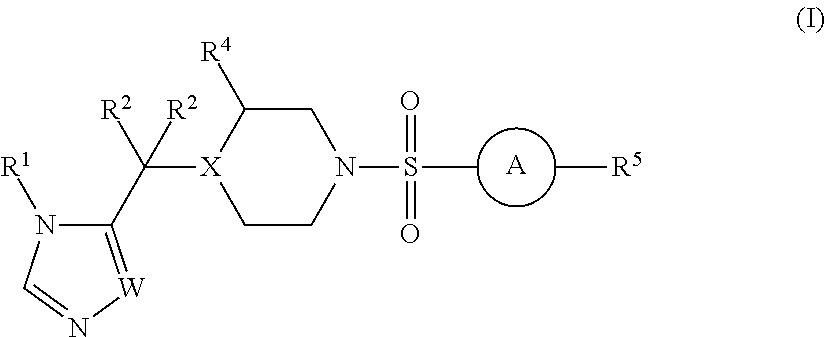
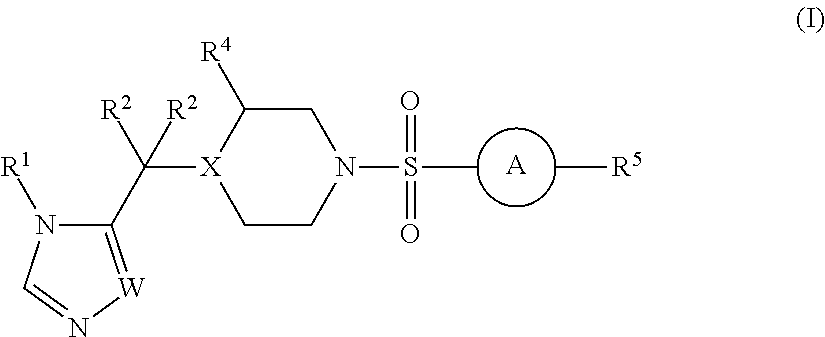
Sulfonamide derivatives and methods of use thereof for improving the pharmacokinetics of a drug
a technology of sulfonamide and derivatives, which is applied in the field of sulfonamide derivatives, can solve the problems of unfavorable pharmacokinetics, sub-therapeutic plasma levels of those drugs over time, and resistance of the virus to the drug, and achieve the effect of improving the pharmacokinetics
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Preparation of Compound 1
[0238]
Step A—Preparation of Int 1-1
[0239]
[0240]To a solution of 1-(4-fluoro-phenyl)-ethanone (400 g, 2.9 mol) and 2-methylpropane-2-sulfinic acid amide (350 g, 2.9 mol) in THF (4 L) was added Ti(OiPr)4 (824 g, 2.9 mol). The reaction mixture was allowed to stir at 90° C. for 48 hours when TLC (PE:EA=10:1) showed the reaction was complete. The mixture was cooled to 0° C. and NaBH4 (110 g, 209 mol) was added in portions. The reaction was allowed to stir at 0° C. until TLC (petroleum ether / EtOAc=1:1) showed the reaction was complete (˜1 hour). The reaction mixture was poured into ice water and the formed precipitate was filtered and washed with ethyl acetate. The filtrate was concentrated in vacuo dissolved into water and the aqueous layer was extracted with ethyl acetate (800 mL×3), dried over anhydrous Na2SO4 and concentrated in vacuo to provide the resulting residue Int 1-1, which was purified using silica gel column. (350 g, 50 MS (ESI) m / z (M+1): 244.
Step B...
example 2
Preparation of Compound 28
[0260]
Step A Preparation of Int 2-1
[0261]
[0262]Potassium tert-butoxide (2.8 g, 24.84 mmol) was dissolved in DMF (10 mL) and benzylthiol (2.6 g, 20.70 mmol) was added dropwise at 0° C. The mixture was allowed to stir at room temperature for 15 minutes and then cooled to 0° C., a solution of 5-bromo-2-chloropyridine (4.0 g, 20.70 mmol) in DMF (4 mL) was added dropwise at 0° C. and the mixture was heated at 80° C. for 1.5 hours. The mixture was poured into water (100 mL) and extracted with ethyl estate (3×100 mL). The combined organic phases were washed with brine (100 mL), water (100 mL) and dried (Na2SO4). Concentrated in vacuo to provide the residue, which was purified with column chromatography (petroleum ether:EtOAc=10:1) to provide Int 2-1 (4.0 g, 69%). MS (ESI): m / z (M+H)+ 280.
Step B Preparation of Int 2-2
[0263]
[0264]To a solution of Int 2-1 (1.5 g, 5.40 mmol) in DCM (100 mL) was added water (100 mL) and HCOOH (100 mL). The heterogeneous mixture was coo...
example 3
Preparation of Compound 31
[0270]
Step A Preparation of Int 3-1
[0271]
[0272]To a stirred solution of 5-bromo-2-cyanopyridine (30.0 g, 0.165 mol) and Ti(O-iPr)4 (51.5 g, 0.181 mol) in 900 mL THF was added EtMgBr (330 mL, 0.045 mmol) under nitrogen at 0° C. The reaction mixture was allowed to stir at room temperature for 5 hours. The reaction was quenched by water and extracted by EtOAc, filtrated and the organic layers were dried over Na2SO4, filtrated and concentrated in vacuo. The resulting residue was purified using flash column chromatography on silica gel eluted with petroleum ether:EtAOc=30:1 to provide product (7.2 g, 21%). MS-ESI (m / z): 213, 215 (M+H)+
Step B Preparation of Int 3-3
[0273]
[0274]To a solution of compound Int 3-1 (7.1 g, 0.033 mol) in the 80 mL of mixed solvent (MeCN / H2O=1:1) was added 2,5-bis(hydroxymethyl)-1,4-dioxane-2,5-diol (7.81 g, 0.043 mol), KSCN (4.18 g, 0.043 mol) and acetic acid (4 mL). The mixture was allowed to stir at 90° C. for 6 hrs. then cooled to 0°...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Structure | aaaaa | aaaaa |
| Therapeutic | aaaaa | aaaaa |
| Pharmacokinetics | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - Generate Ideas
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com