Intraocular Lens Cell Migration Inhibition System
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
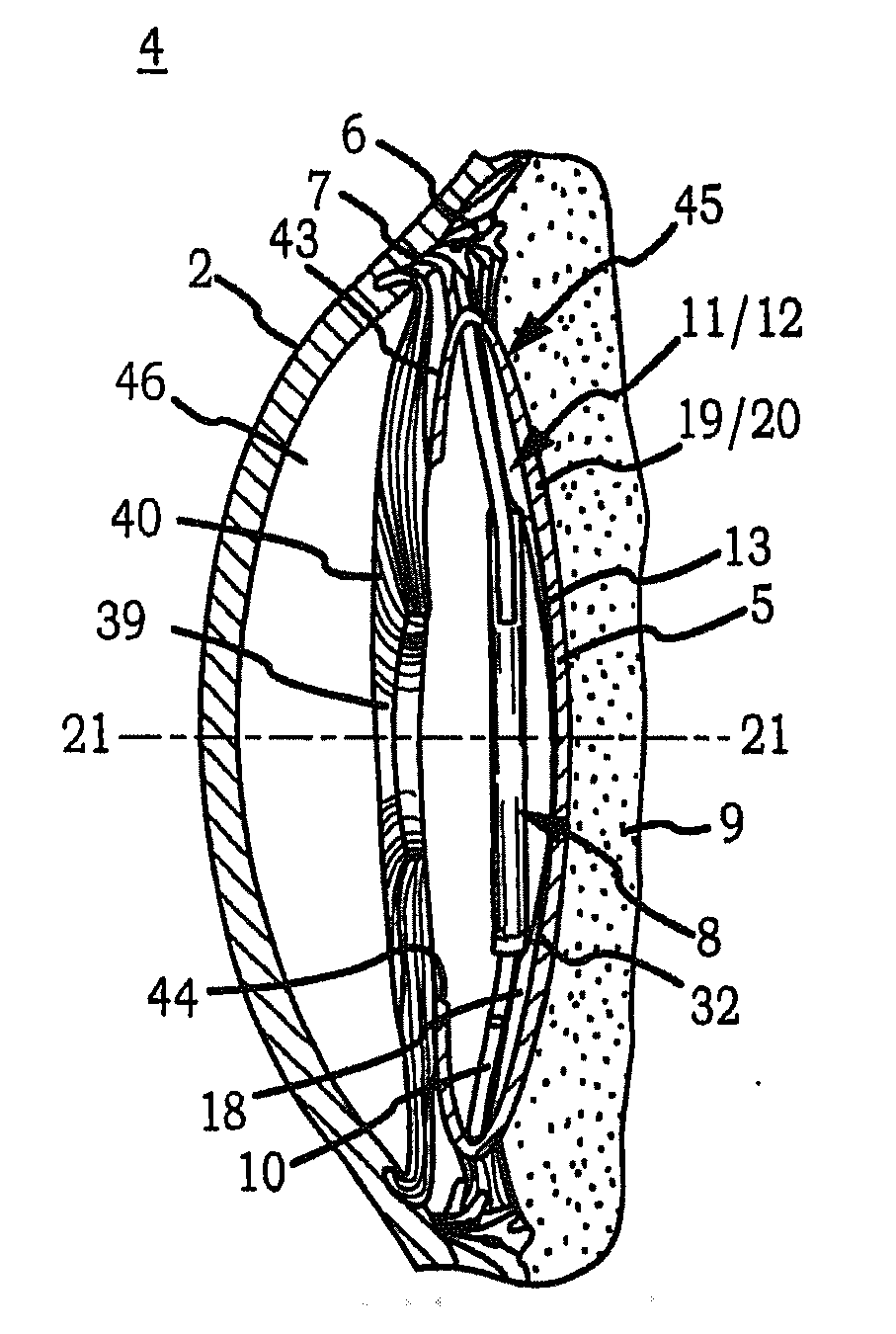
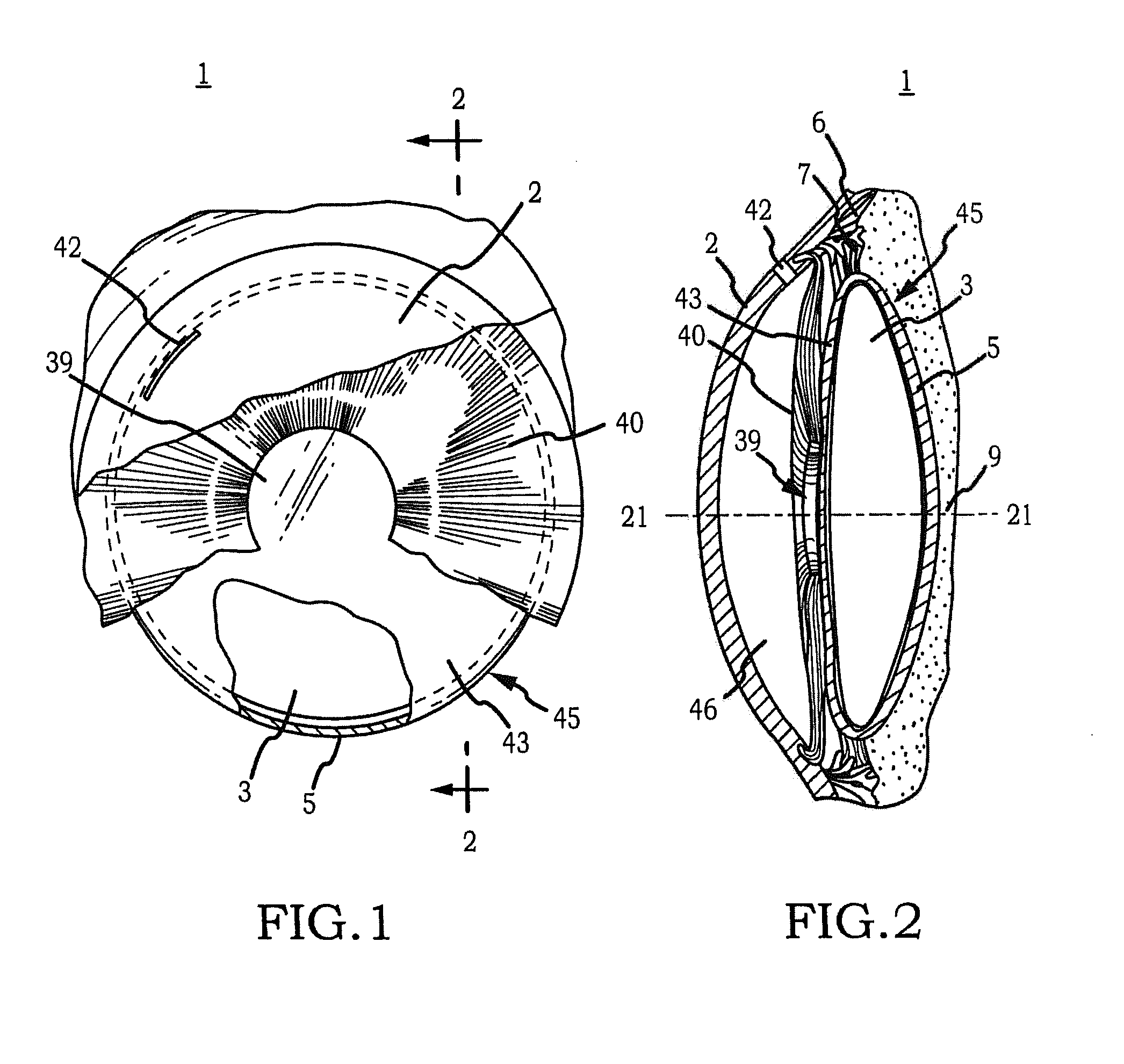
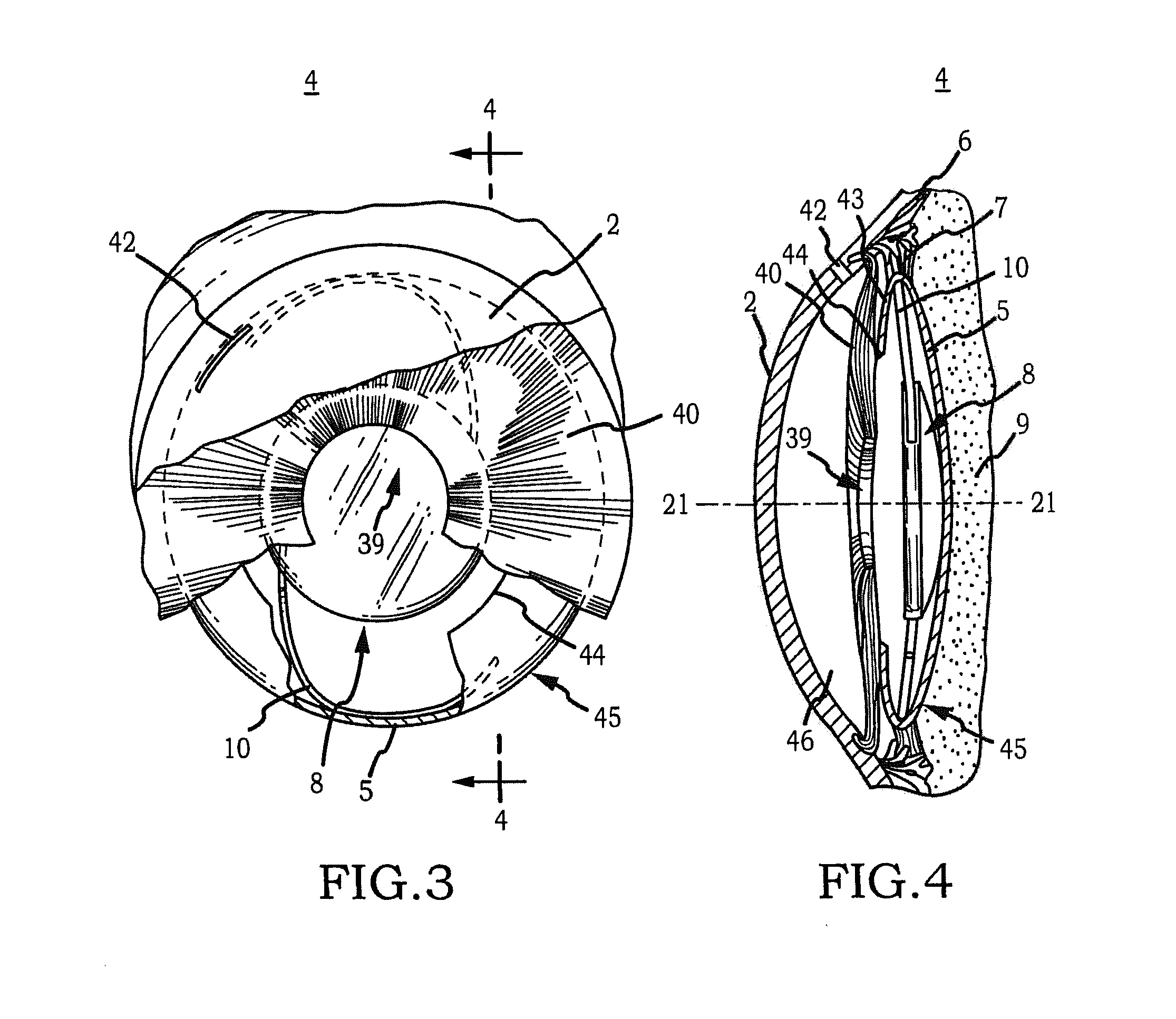
[0070]Generally, an intraocular implant and methods for treating an ocular condition. In particular, an intraocular implant which implanted between an intraocular lens and the surface of the posterior capsule of the eye inhibits migration of residual lens epithelial cells after cataract surgery by providing structural barriers to reduce posterior capsule opacification of the eye.
Definitions
[0071]“A” or “an” entity refers to one or more of that entity; for example, “a polymer” refers to one or more of those compositions or at least one composition. As such, the terms “a” or “an”, “one or more” and “at least one” can be used interchangeably herein. Furthermore, the language “selected from the group consisting of” refers to one or more of the elements in the list that follows, including combinations of two or more of the elements.
[0072]“About” for the purposes of the present invention means that ranges may be expressed as from “about” one particular value to “about” another particular ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com