Protein-based streptococcus pneumoniae vaccines
a streptococcus pneumoniae and vaccine technology, applied in the field of protein-based streptococcus pneumoniae vaccines, can solve the problems of increasing the /i>protein of pneumococcal infections, affecting the survival rate of infants under two years of age who are at most risk of pneumococcal infections, and not responding efficiently to the currently available polysaccharide-based vaccines
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
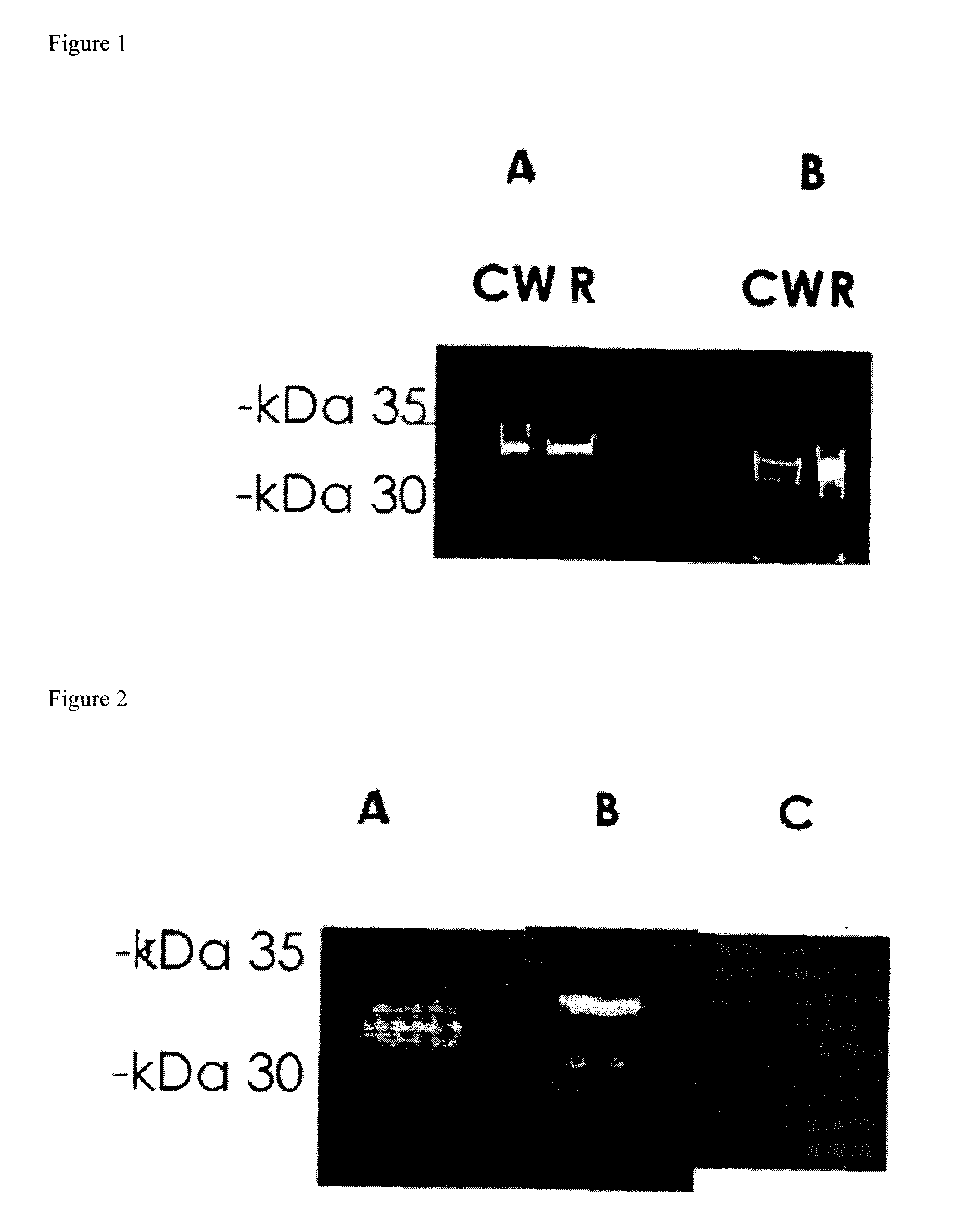
[0127]Prevention of S. pneumoniae Infection in Mice by Inoculation with S. pneumoniae Cell Wall Protein Fractions
Methods:
[0128]Bacterial Cells: The bacterial strain used in this study was an S. pneumoniae serotype 3 strain and R6. The bacteria were plated onto tryptic soy agar supplemented with 5% sheep erythrocytes and incubated for 17-18 hours at 37° C. under anaerobic conditions. The bacterial cells were then transferred to Todd-Hewitt broth supplemented with 0-5% yeast extract and grown to mid-late log phase. Bacteria were harvested and the pellets were stored at −70° C.
Purification of Cell Wall Proteins: Bacterial pellets were resuspended in phosphate buffered saline (PBS). The resulting pellets were then treated with mutanolysin to release cell wall components. Supernatants containing the CW proteins were then harvested. Subsequently, the bacteria were sonicated, centrifuged and the resulting pellet containing the bacteria membranes (m) were lysed with 0.5% triton X-100.
Fracti...
example 2
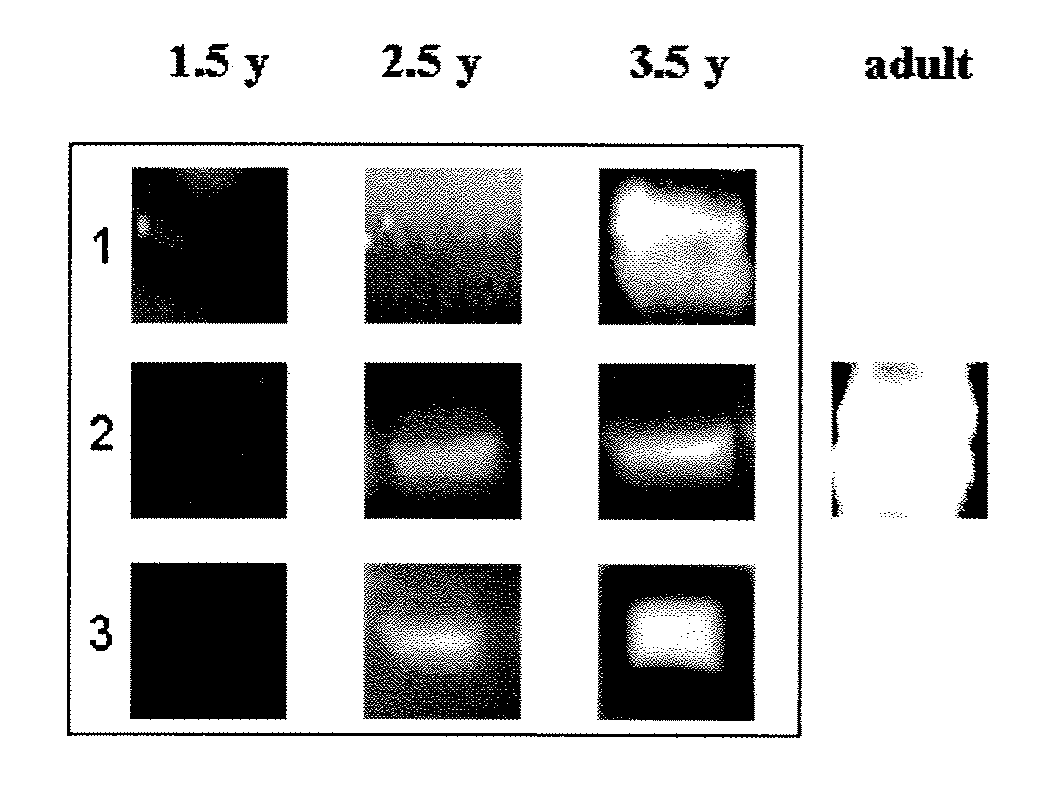
[0129]Determination of Age-Related Immunoreactivity to S. pneumoniae Surface Proteins
[0130]The following study was carried out in order to investigate the age-related development of immunoreactivity to S. pneumoniae cell wall and cell membrane proteins.
[0131]Operating as described hereinabove in Example 1, a fraction containing cell wall proteins was obtained from a clinical isolate of S. pneumoniae. In addition, cell membrane proteins were recovered by solubilizing the membrane pellet in 0.5% Triton X-100. The cell wall and cell membrane proteins were separated by means of two-dimensional gel electrophoresis, wherein the proteins were separated using polyacrylamide gel isoelectric focusing in one dimension, and sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) in the other dimension. The separated proteins were either transferred to a nitrocellulose membrane or directly stained with Coomassie Brriliant blue.
[0132]Sera were collected longitudinally from healthy ch...
example 3
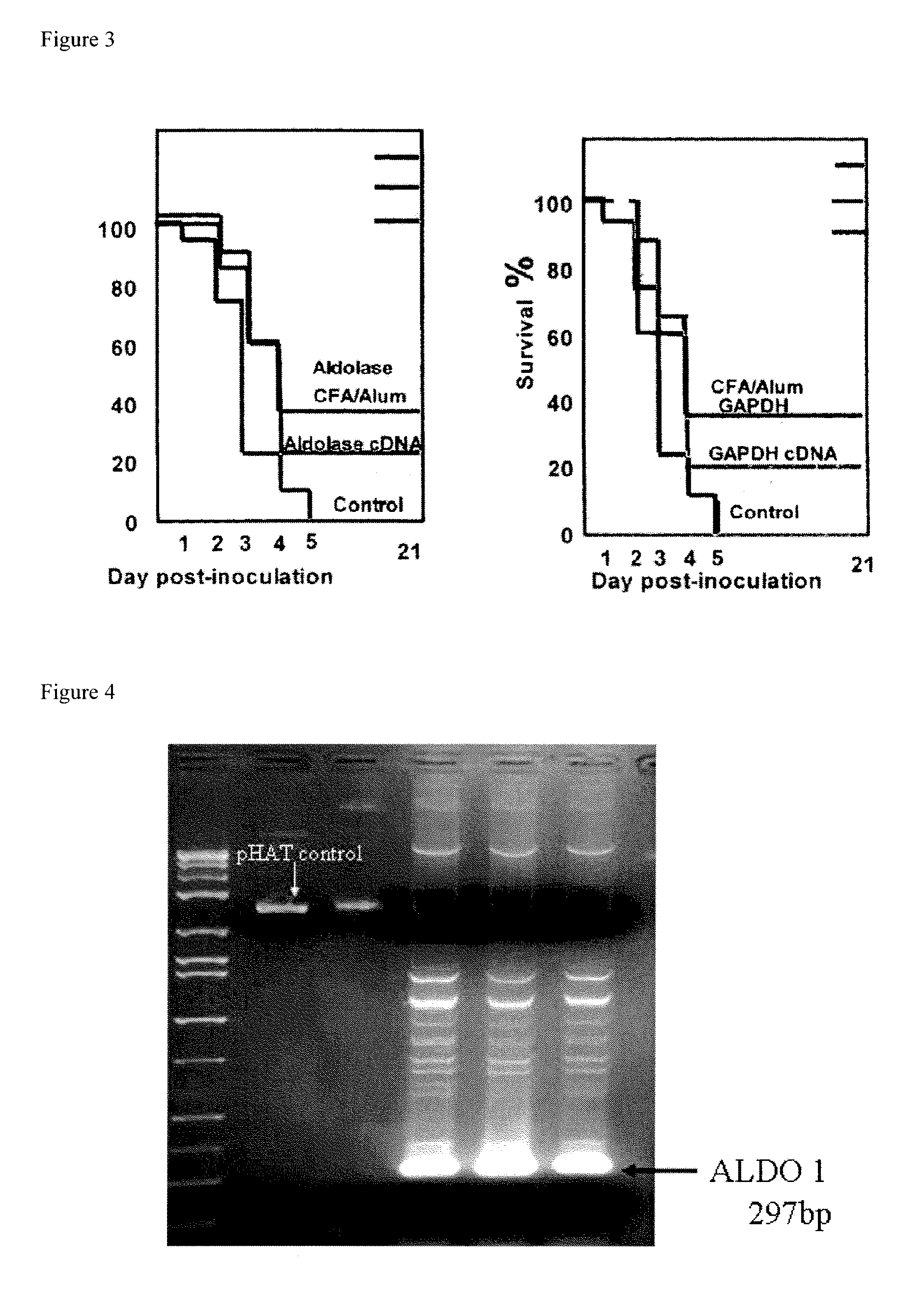
[0137]Prevention of S. pneumoniae Infection in Mice with Recombinantly-expressed S. pneumoniae Cell Surface Proteins
[0138]Glycolytic enzymes associated with the cell surface of Streptococcus pneumoniae are antigenic in humans and elicit protective immune responses in the mouse.
[0139]The glycolytic enzymes fructose-bisphosphate aldolase (FBA, NP—345117, SEQ ID NO:13), and Glyceraldehide 3 phosphate dehydrogenase (GAPDH, NP 346439, SEQ ID NO:12), which are associated with the cell surface of S. pneumoniae, were used to immunize mice against S. pneumonia as described in Ling et al., Clin. Exp. Immunol. 138:290-298. 2004. It was shown that both proteins, which are antigenic in humans, elicit cross-strain protective immunity in mice.
[0140]Cloning of Immunogenic S. pneumoniae Surface Proteins: S. pneumoniae fructose-bisphosphate aldolase (hereinafter referred to as “aldolase”) and GAPDH proteins were cloned into the pHAT expression vector (BD Biosciences Clontech, Palo Alto, Calif., USA; ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Time | aaaaa | aaaaa |
| Fraction | aaaaa | aaaaa |
| Time | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com