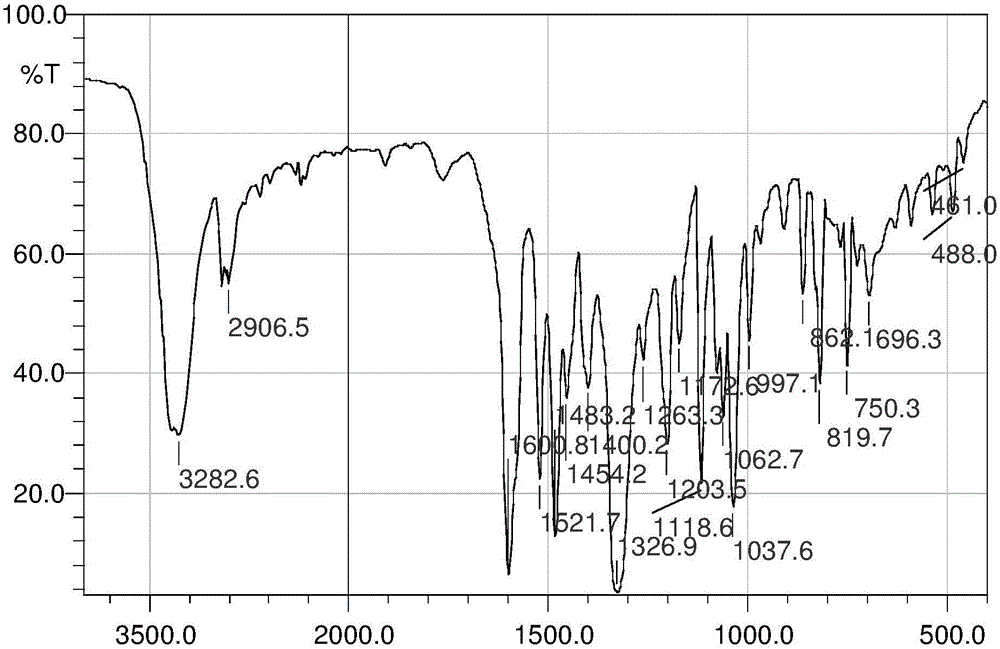
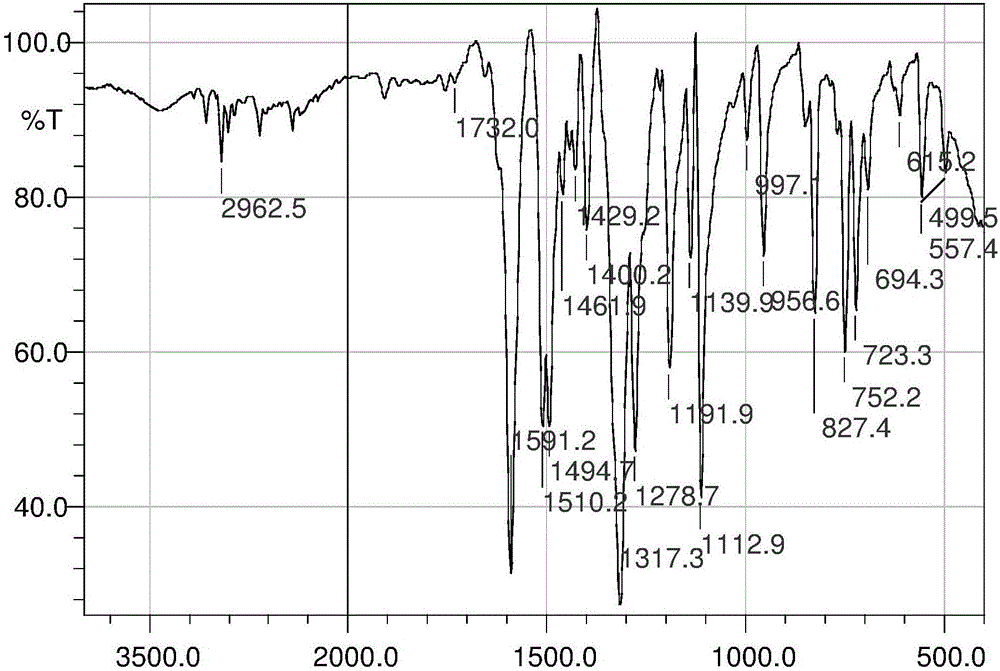
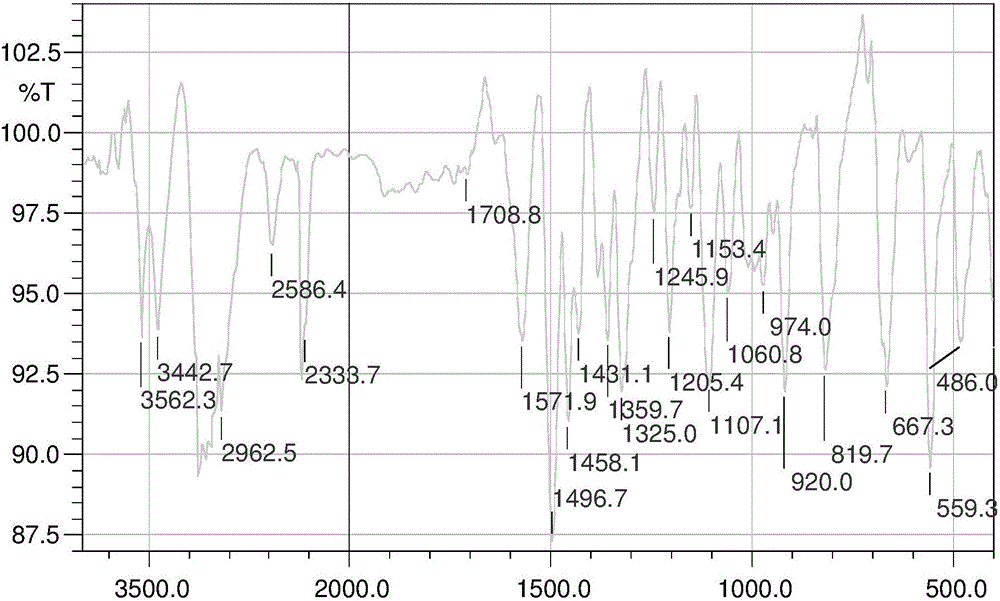
Nitrogen mustard derivative N,N-di(2-chloroethyl)-N'-hexadecanoyl-1,4-phenylenediamine and preparation method thereof
A technology of chloroethyl and derivatives, which is applied in the preparation of carboxylic acid amides, amino compounds, and organic compounds. It can solve the problems of blocking and killing, reducing the defense function of the body, and achieves enhanced therapeutic index and reduced complications. The risk of disease and the effect of reducing toxic side effects
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
[0038] The preparation method of the above-mentioned nitrogen mustard derivative N,N-bis(2-chloroethyl)-N'-hexadecanoyl-1,4-phenylenediamine comprises the following steps:
[0039] Step 1, preparing N,N-bis(2-chloroethyl)-1,4-phenylenediamine;
[0040] Substep 1a, first prepare N,N-bis(2-hydroxyethyl)-4-nitroaniline according to the following steps: 4-chloronitrobenzene, diethanolamine, potassium carbonate and 1% CuSO 4 The solution is added to the reactor in turn, and stirred evenly, the CuSO 4For the solution, when the temperature is raised to 82°C, add toluene into the reactor, continue to heat up to 115-120°C, and stir at a constant temperature for 6-9 hours. Use thin-layer chromatography to track the reaction process. After the reaction is complete, distill to remove the toluene. Pour the remaining liquid after distillation into hot water at 50°C-70°C, stir, and after standing overnight, filter the water layer to obtain the crude product. Use absolute ethanol to recrysta...
Embodiment 1
[0045] 1.1 Synthesis of N,N-bis(2-hydroxyethyl)-4-nitroaniline
[0046]
[0047] Add 12.64g (0.080mol) of 4-chloronitrobenzene, 10.99g (0.104mol) of diethanolamine and 7.18g (0.052mol) of potassium carbonate to a 250mL three-necked flask, and finally add 1.2mL of CuSO with a mass concentration of 1%. 4 solution, mix well. Heat to raise the temperature. When the temperature reaches 82°C, add 5.0mL of toluene to the reaction liquid, continue to raise the temperature to control the reaction temperature to 117°C, use thin-layer chromatography to track the reaction progress, and stir at constant temperature for 6h. After the reaction is complete, toluene is removed by distillation, and the remaining liquid after distillation is poured into hot water at 50°C, stirred vigorously, left to stand overnight, and the water layer is removed by suction filtration to obtain a crude product, which is recrystallized with absolute ethanol to obtain a yellow needle 11.39 g of N,N-bis(2-hydro...
Embodiment 2
[0058] 1.1 Synthesis of N,N-bis(2-hydroxyethyl)-4-nitroaniline
[0059]
[0060] Add 4-chloronitrobenzene, diethanolamine and potassium carbonate at a molar ratio of 1:1.2:0.6 to a 250mL three-necked flask, and finally add CuSO with a mass concentration of 1%. 4 solution, and the mass concentration is 1% CuSO 4 The dosage ratio of the solution to 4-chloronitrobenzene is 0.5mL:3g, mix well; heat up the temperature, when the temperature reaches 82°C, add toluene to the reaction solution, and the dosage of toluene and 4-chloronitrobenzene The ratio was 1mL:2g; continue to increase the temperature to control the reaction temperature to 120°C, use thin-layer chromatography to track the reaction progress, and stir the reaction at constant temperature for 9h. After the reaction is complete, toluene is removed by distillation, and the mother liquor after distillation is poured into hot water at 60°C, stirred vigorously, left to stand overnight, and the water layer is removed by su...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Boiling point | aaaaa | aaaaa |
| Density | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - Generate Ideas
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com