Recombinant protein Tumstatin-CD137L4 with Tumstatin activities as well as preparation method and application thereof
A recombinant protein and active technology, applied in recombinant DNA technology, medical preparations containing active ingredients, peptide/protein components, etc., can solve problems such as lack of research, achieve preparation avoidance, good application prospects and market value, and simple preparation Effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
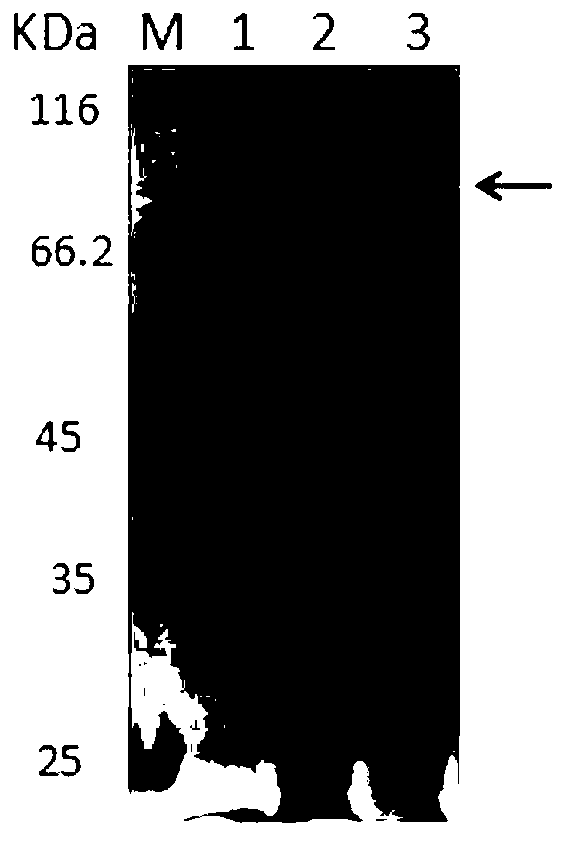
[0066] Example 1: Expression and purification of recombinant protein Tumstatin-CD137L4 with Tumstatin activity in yeast expression system
[0067] (1) Construction of recombinant protein Tumstatin-CD137L4 yeast expression vector with Tumstatin activity
[0068] The present invention designs 6 kinds of recombinant protein Tumstatin-CD137L4 expression vectors with Tumstatin activity, its nucleotide sequence is the sequence shown in SEQ ID NO.1 to SEQ ID NO.6, and the amino acid sequence is SEQ ID NO.7 to SEQ ID The sequence shown in NO.12. The histone or polypeptide consists of three groups of tumstatins with different active sites (three groups have amino acid sequences of 45-98, 60-132, and 60-98 positions of full-length tumstatin) and CD137L (with full-length CD137L extracellular domain protein) 50-240 amino acid sequence) are respectively connected and fused by Linker connecting peptides. Wherein the linker amino acid sequence is: GGGGSGGGGSGGGGS or AEAAAKEAAAKEAAAAKEAAAKA...
Embodiment 2
[0088] Example 2: Expression and purification of recombinant protein Tumstatin-CD137L4 with Tumstatin activity in Bacillus subtilis
[0089] (1) Construction of recombinant protein Tumstatin-CD137L4 Bacillus subtilis expression vector with Tumstatin activity
[0090] The present invention designs 6 kinds of recombinant protein Tumstatin-CD137L4 expression vectors with Tumstatin activity, its nucleotide sequence is the sequence shown in SEQ ID NO.1 to SEQ ID NO.6, and the amino acid sequence is SEQ ID NO.7 to SEQ ID NO The sequence shown in .12. The histone or polypeptide consists of three groups of tumstatins with different active sites (three groups have amino acid sequences of 45-98, 60-132, and 60-98 positions of full-length tumstatin) and CD137L (with full-length CD137L extracellular domain protein) 50-240 amino acid sequence) are respectively connected and fused by Linker connecting peptides. Wherein the linker amino acid sequence is: GGGGSGGGGSGGGGS or AEAAAKEAAAKEAAAA...
Embodiment 3
[0113] Example 3: Expression of recombinant protein Tumstatin-CD137L4 with Tumstatin activity in Escherichia coli expression vector and inclusion body protein renaturation and purification
[0114] (1) Construction of recombinant protein Tumstatin-CD137L4 with Tumstatin activity in Escherichia coli expression vector
[0115] The present invention designs 6 kinds of recombinant protein Tumstatin-CD137L4 expression vectors with Tumstatin activity, its nucleotide sequence is the sequence shown in SEQ ID NO.1 to SEQ ID NO.6, and the amino acid sequence is SEQ ID NO.7 to SEQ ID The sequence shown in NO.12. The histone or polypeptide consists of three groups of tumstatins with different active sites (three groups have amino acid sequences of 45-98, 60-132, and 60-98 positions of full-length tumstatin) and CD137L (with full-length CD137L extracellular domain protein) 50-240 amino acid sequence) are respectively connected and fused by Linker connecting peptides. Wherein the linker a...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap