A detection, treatment and prognosis target and application of cholangiocarcinoma
A cholangiocarcinoma-specific technology, applied in the field of biomedicine, can solve the problems of insensitivity of cholangiocarcinoma to chemotherapy and difficulty in entering the anatomical location of cholangiocarcinoma
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
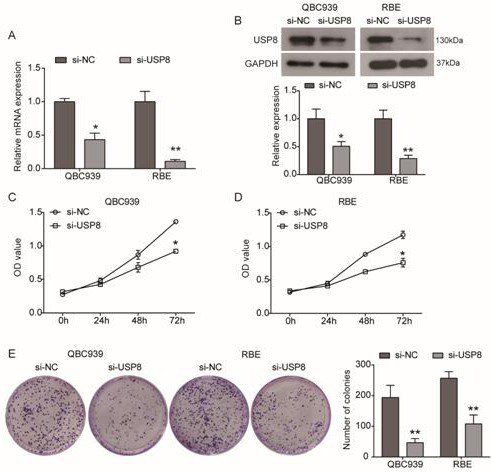
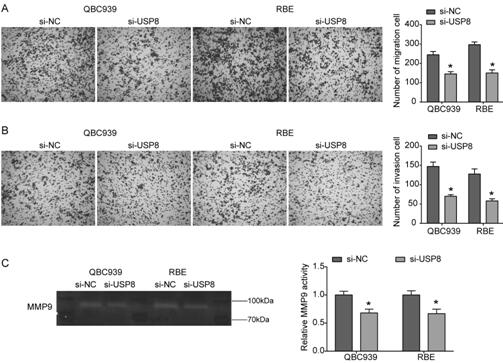
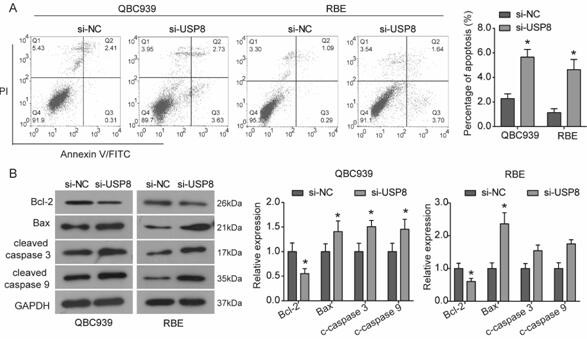
[0022] Cell Culture and Transfection
[0023] Human cholangiocarcinoma cell lines QBC939 and RBE obtained from the Chinese Academy of Sciences (Shanghai) type culture cell bank were used at 37°C, 5% CO 2 1640 medium (Hyclone, Logan, UT, USA) supplemented with 10% fetal bovine serum (Hyclone, Logan, UT, USA) and penicillin / streptomycin. After entering the logarithmic growth phase, the cells were washed 3 times with PBS, digested with trypsin, and placed in 6-well plates. When the cell density of the 6-well plate reached 50-60% confluency, Lipofectamine 2000 was used to transfect siRNA (Invitrogen, USA). siRNA targeting USP8 (si-USP8) was synthesized by OriGene (Beijing, China), and the siRNA negative control (OriGene) was used as a negative control (si-NC).
[0024] qRT-PCR analysis
[0025] 24 hours after transfection with siRNA, total RNA was extracted from the cells with Ultrapure RNA Kit (CWBIO, Beijing, China), and reverse transcribed into cDNA with HiFiScript cDNA Synt...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com