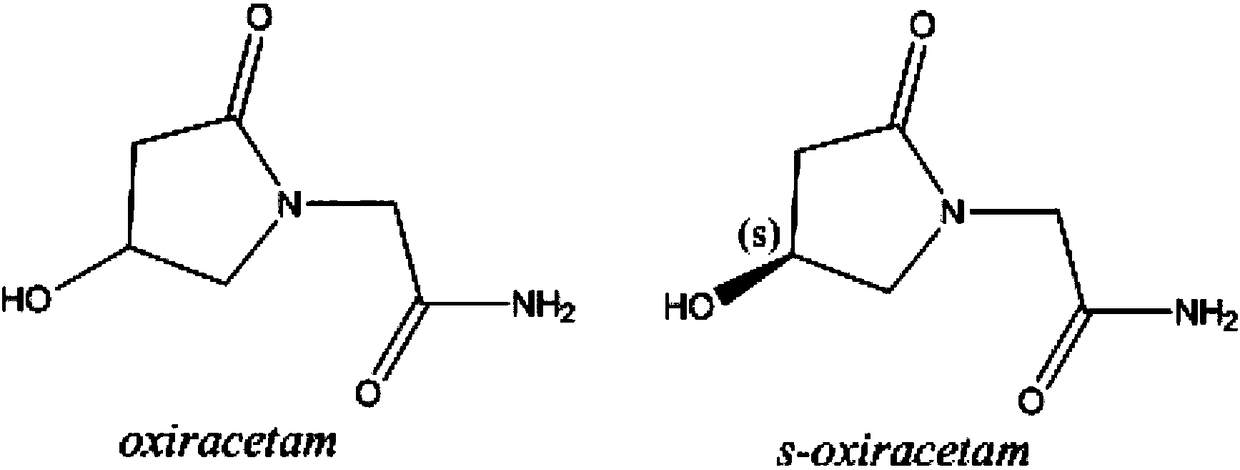
A kind of levoxiracetam sustained-release capsule with good stability and preparation method thereof
A technology for sustained-release capsules and stability, which is applied in the field of levoxiracetam sustained-release capsules and its preparation, and can solve problems such as failure to meet the requirements of sustained-release preparations, poor control of release, and large differences in product quality. , to achieve the effect of simple and feasible preparation process, slow release speed and reduced taking times
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
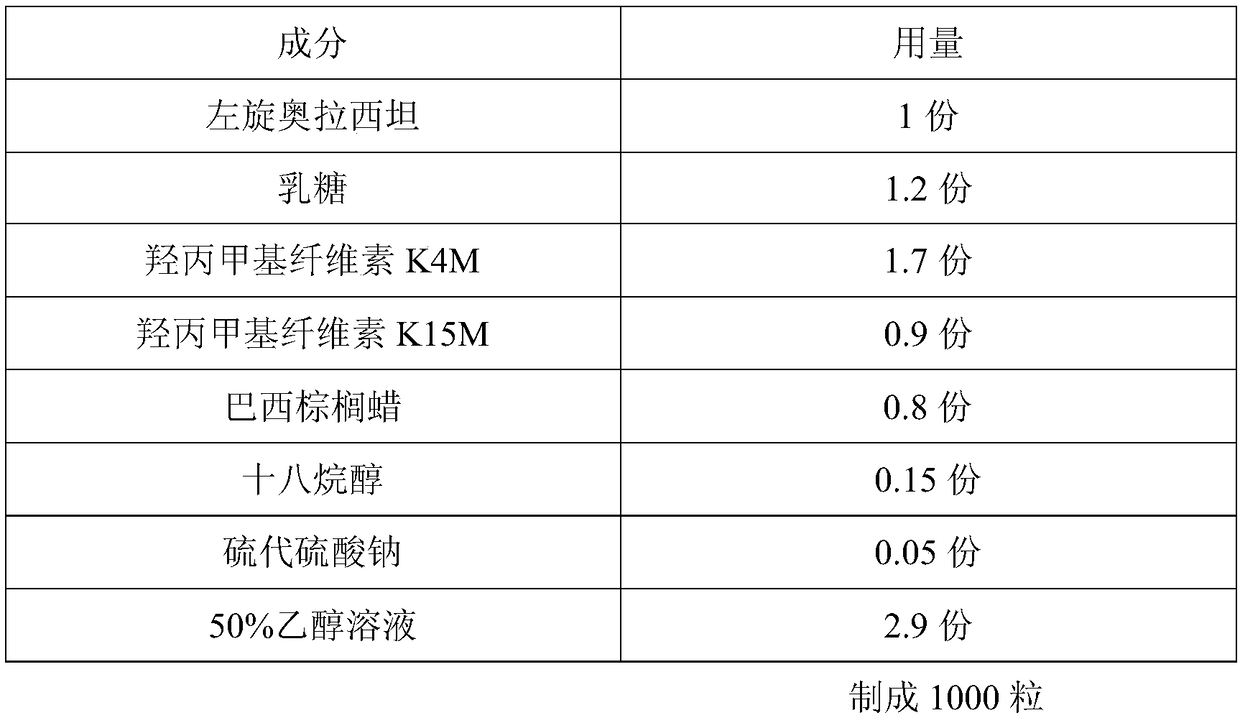
[0023] A kind of levoxiracetam sustained-release capsule with good stability is prepared according to the following steps:
[0024]
[0025] Preparation process:
[0026] 1. Pretreatment of raw and auxiliary materials: take the prescribed amount of levoxiracetam, lactose, hydroxypropylmethylcellulose K4M, hydroxypropylmethylcellulose K15M, carnauba wax, and sodium thiosulfate, mix and pulverize in a mixing grinder Become fine powder (all pass through No. 5 sieve and the amount that can pass through No. 6 sieve must not be less than 95% of total amount), sieve;
[0027] 2. Granulation: Add ethanol solution, mix and granulate with 18-mesh sieve, place the wet granules in a hot air oven, set the temperature at 40-60°C, dry until the moisture content of the granules is ≤3%, and granulate (over 24 Mesh sieve), spare;
[0028] 3. Total blending: crush stearyl alcohol through a 100-mesh sieve, add it to the granulated granules, and mix with a three-dimensional motion mixer for 1...
Embodiment 2
[0060] A kind of levoxiracetam sustained-release capsule with good stability is prepared according to the following steps:
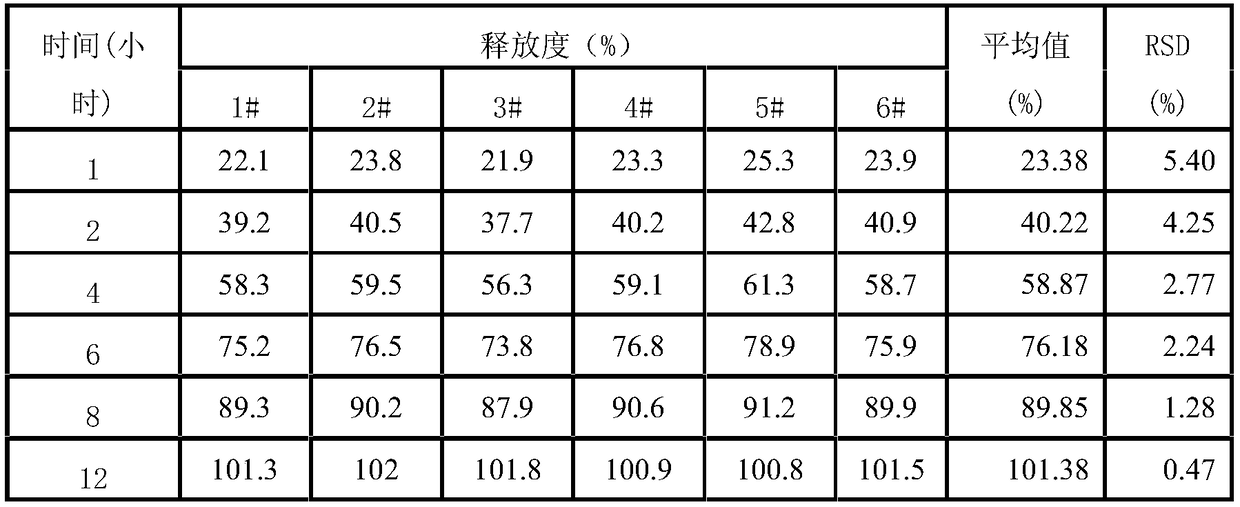
[0061] Preparation process: prepared according to the preparation process of Example 1. Carry out the test by the test method of embodiment 1, and release degree test result shows that levoxiracetam is slow release, and release time is up to 12 hours, and the release degree RSD of each sample at different time points is all less than 6%, and stability test result It shows that the sample quality is stable in June, and the impurity increase is only 0.05%, and the quality is stable for 24 months, and the impurity increase is only 0.05%, so the validity period of this product is at least 24 months.
Embodiment 3
[0063] A kind of levoxiracetam sustained-release capsule with good stability is prepared according to the following steps:
[0064] Preparation process: prepared according to the preparation process of Example 1. Carry out the test by the test method of embodiment 1, the release measurement test result shows that levoxiracetam is slow release, and the release time is up to 12 hours, and the release RSD of each sample at different time points is all less than 5%, and the stability test result It shows that the sample quality is stable in June, and the impurity increase is only 0.06%, and the quality is stable for 24 months, and the impurity increase is only 0.05%, so the validity period of this product is at least 24 months.
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap