Compounds and methods for treating aberrant adrenocartical cell disorders
a technology of aberrant adrenal cortex and compounds, applied in the direction of biocide, animal repellents, drug compositions, etc., can solve the problems of dismal prognosis of patients with advanced disease, affecting the treatment effect, so as to achieve the effect of increasing hormone production
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
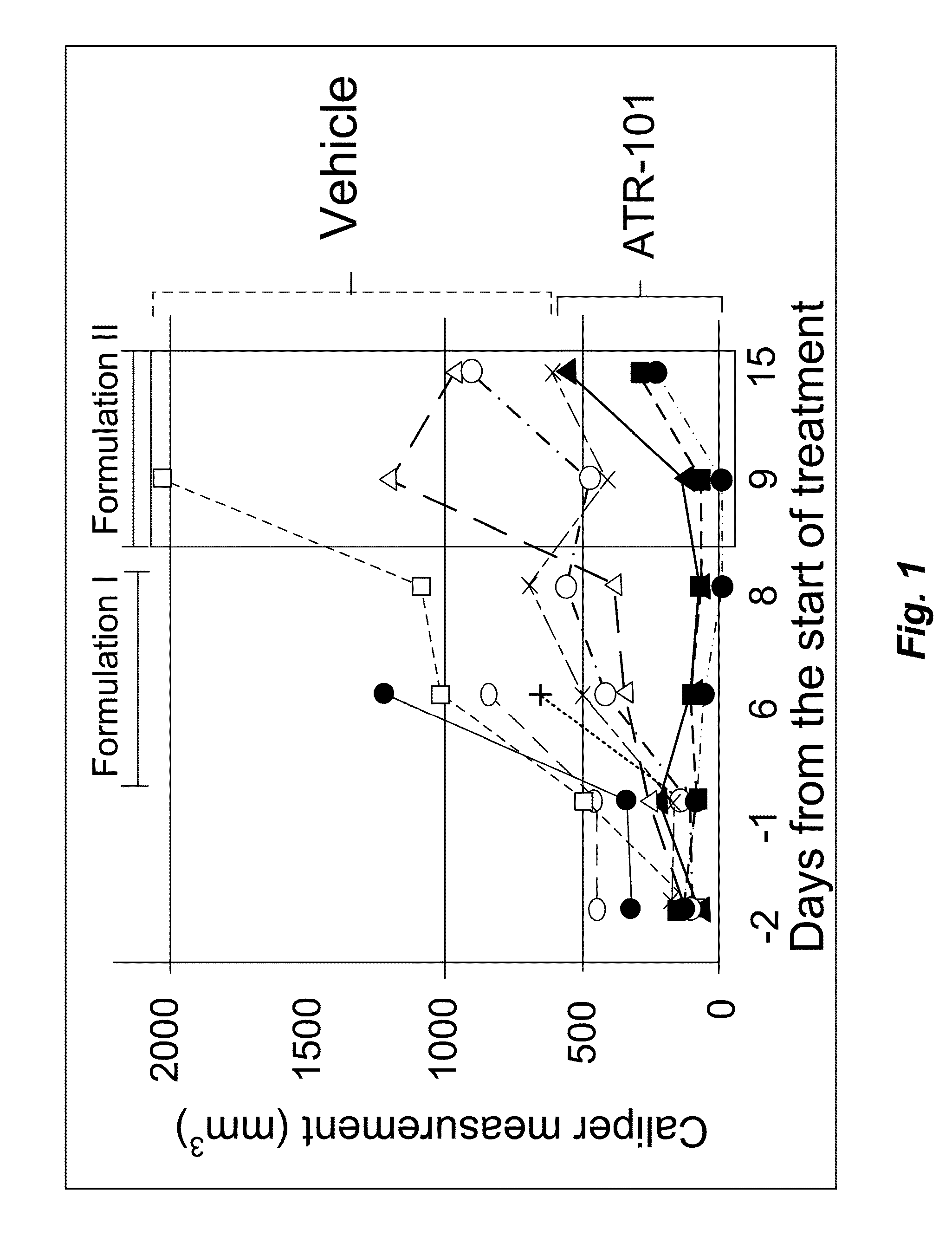
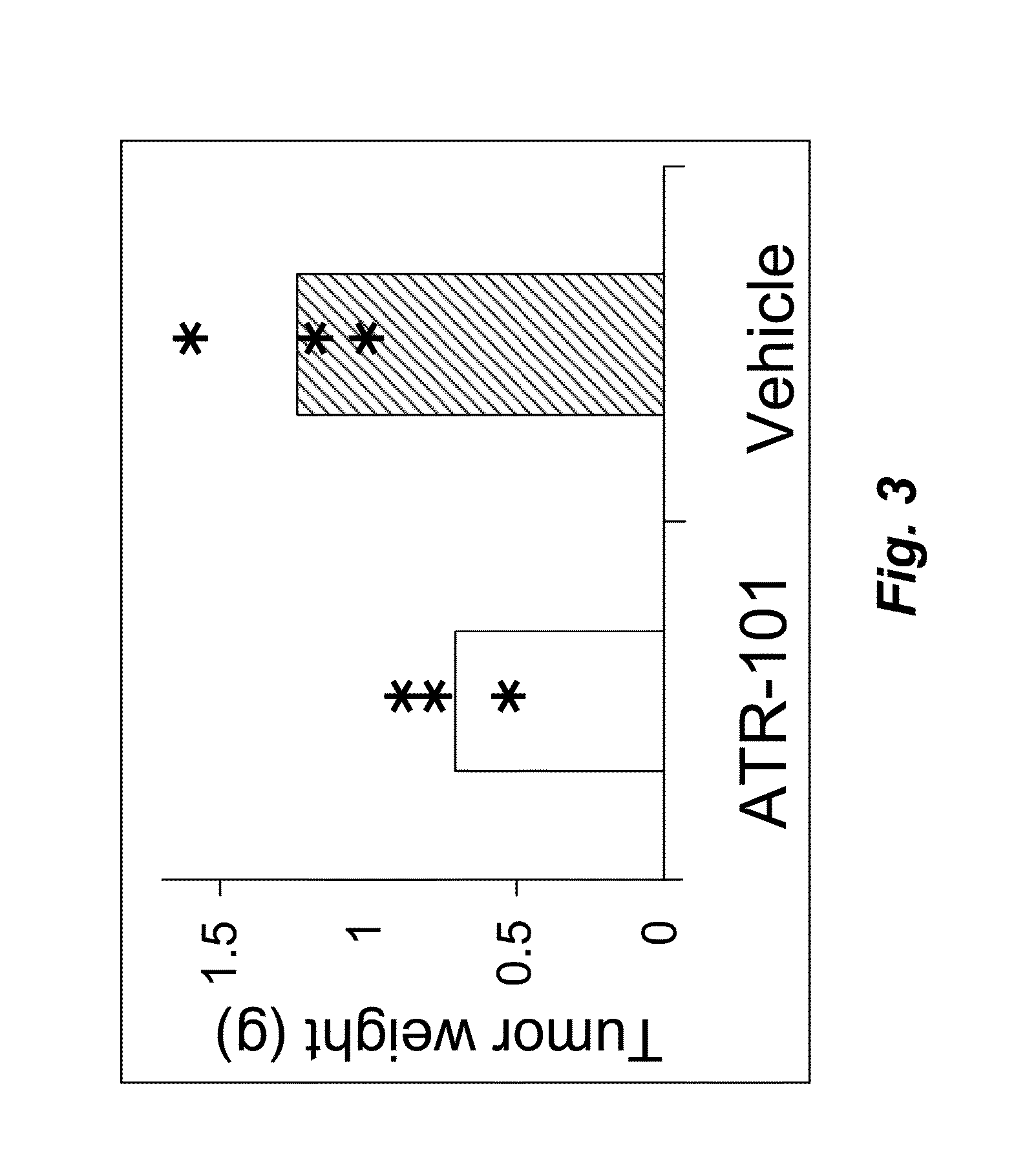
[0168]To investigate the potential therapeutic effect of ATR-101 in a mouse model of adrenocortical carcinoma (ACC), xenografts of the human ACC-derived cell line H295R in SCID mice (6-7 week males) were established. After the xenografts had grown to measurable size, the mice were randomized in two groups. The treatment group was administered 300 mg / kg / day ATR-101 by oral gavage. The control group was administered the vehicle without ATR-101. The tumor size was measured using calipers before and on the indicated days after starting administration of ATR-101.
[0169]Four of the mice in the treatment group and two of the mice in the control group died within 2 days of transferring the mice to the biohazardous agent containment facility and initiating administration. The cause(s) of these deaths are unknown, but unrelated to ATR-101 treatment. These mice were excluded from the analysis.
[0170]One of the mice in the treatment group and two of the mice in the control group died six days aft...
example 2
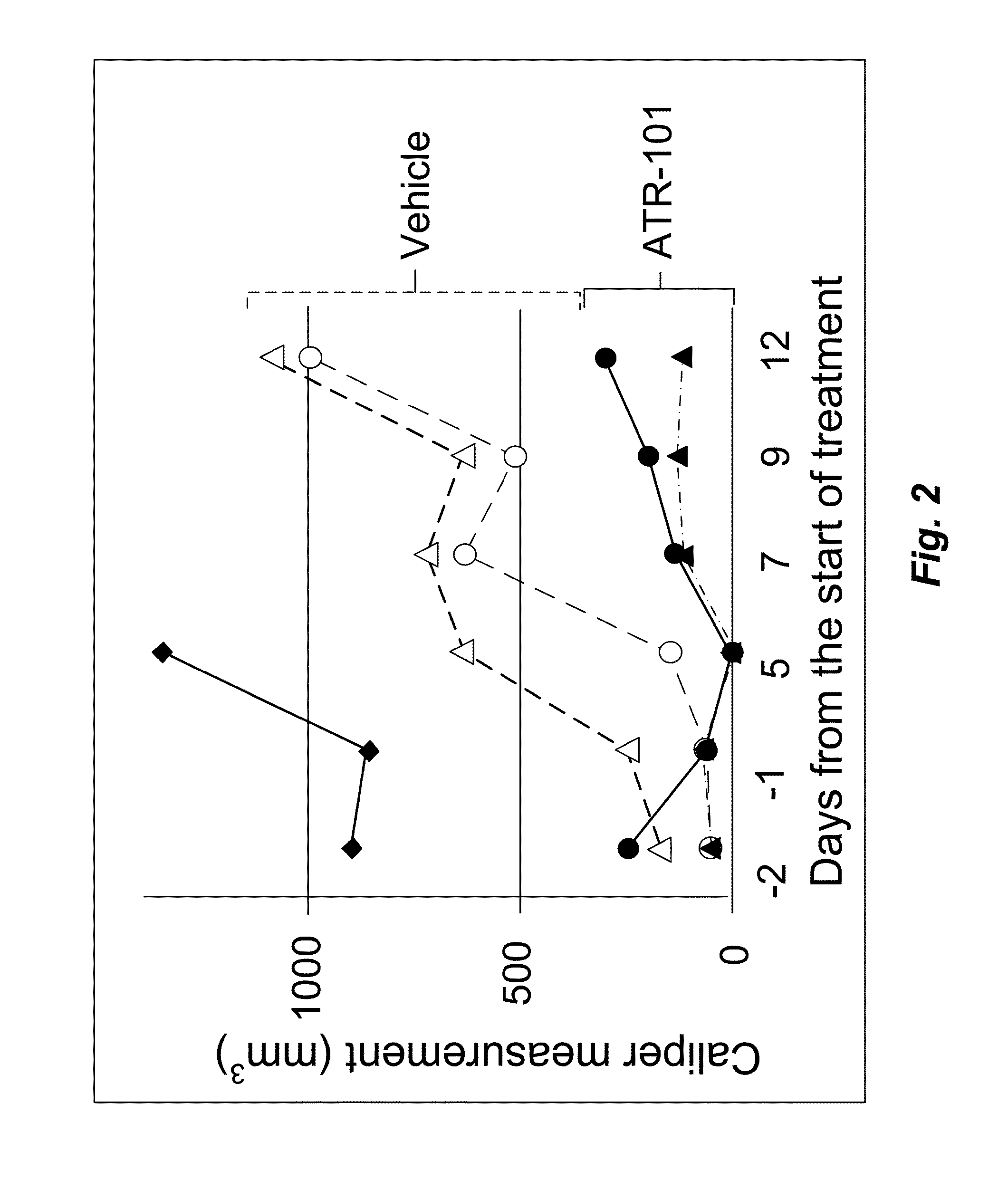
[0175]To determine if the effects of ATR-101 on ACC-derived cell line xenotransplants were reproducible, the experiment using xenotransplants of the same H295R cell line in SCID mice (10-11 week males) were repeated.
[0176]One of the mice in the treatment group had a large tumor produced by H295R cells implanted 4 weeks earlier. This mouse died 6 days after ATR-101 administration and is not included in the analysis. The tumors of the remaining mice in the treatment group (n=2) decreased to undetectable size at six days after the start of ATR-101 administration (FIG. 2). Meanwhile, the tumors of the mice in the control group (n=2) more than doubled in size. The tumors of the mice in the treatment group resumed growth within eight days after the start of ATR-101 administration. The tumors of the treated mice were one-eighth to one-third of the size of the tumors in the control mice 13 days after the start of ATR-101 administration. The resumption of tumor growth coincided with the swit...
example 3
[0179]Xenograft size was measured using calipers as a function of time after the start of oral administration of 300 mg / kg / day ATR-101 (triangles) versus control mice treated with vehicle (circles) (FIG. 4). The data show the average and standard deviation of 9 and 10 mice in each group. One mouse included in the treatment group was eliminated because the abdominal location of the xenograft prevented accurate measurement of its size. The 20 mice were randomized between the treatment and control arms of the study. The xenografts were produced by subcutaneous injection of 100,000,000 H295R cells in each mouse.
PUM
| Property | Measurement | Unit |
|---|---|---|
| CMC pH | aaaaa | aaaaa |
| CMC pH | aaaaa | aaaaa |
| time | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com