Application of PLpro protein inhibitor in medicine for treating or preventing novel coronavirus infection
A new type of coronavirus technology, applied in antiviral agents, drug combinations, pharmaceutical formulations, etc., can solve problems such as high risk of side effects
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0052] Example 1: Induced expression and purification of PLpro protein
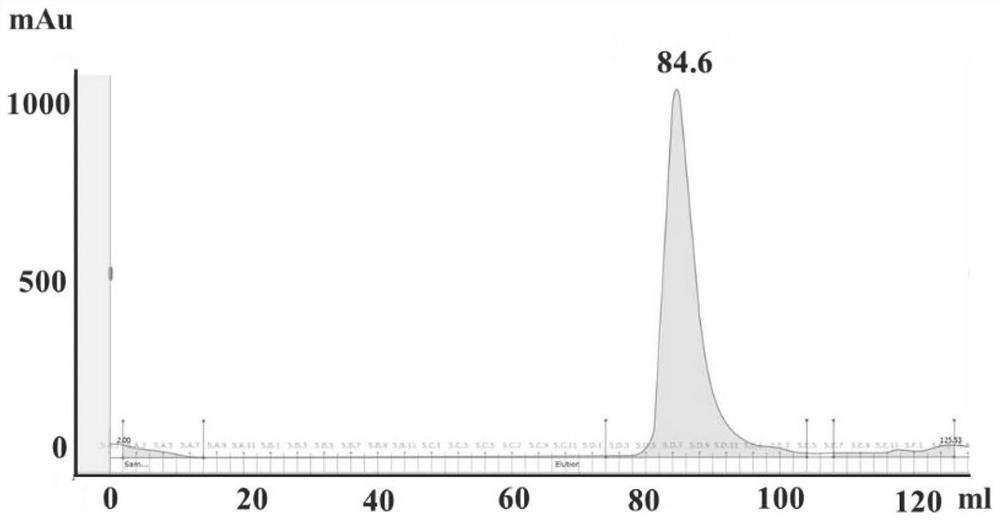
[0053] The PLpro prokaryotic expression plasmid (see SEQ ID NO.1 for the expressed PLpro protein amino acid sequence) was transformed into BL21 (DE3) E. coli competent cells, and grown in LB medium at 37°C until OD600 was 0.6-0.8 Time. Protein expression was induced by the addition of 0.5 mM IPTG and 1 mM zinc chloride (ZnCl 2 ), and then E. coli was grown overnight at 18°C. After collection by centrifugation, the cell pellet was resuspended with 50 mM Tris-HCl, 150 mM NaCl, 10 mM Imidazole, 2 mM DTT, and pH=8.5 buffer, disrupted by sonication, and centrifuged at 18,000 rpm to take the supernatant. Purified on a His-TRAPTM column and finally eluted with buffer (50 mM Tris-HCl, 150 mM NaCl, 250 mM Imidazole, 1 mM DTT, pH 7.4) and further purified on a gel chromatography column (Superdex 20016 / 60, GE) , the SEC buffer was 20 mM Tris-HCl, 100 mM NaCl, 1 mM DTT, pH 7.4. The purified PLpro protein was conc...
Embodiment 2
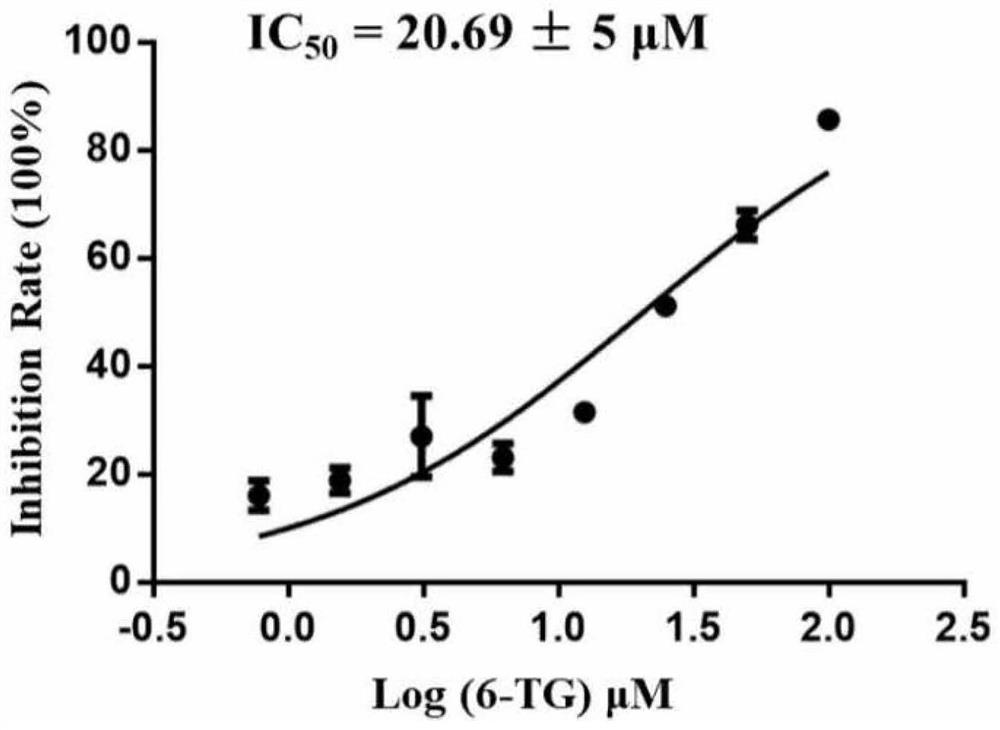
[0054] Example 2: In vitro assay to measure the activity of thioguanine (6-TG) in inhibiting PLpro protein (IC50)
[0055]The reaction system was 50 μL, wherein the concentration of PLpro was 30 nM, the concentration of substrate Ub-AMC (Boston Biochem) was 250 nM, and the reaction buffer was 50 mM Hepes (pH=7.5), 0.01% TrixtonX-100, 0.1 mg / ml BSA and 2 mM DTT. A gradient concentration (100, 50, 25, 12.5, 6.25, 3.125, 1.56, 0.78 μM) of inhibitor thioguanine (6-TG) was added, and then the fluorescence emission intensity was measured using a microplate reader (excitation: 340 nm; emission: 430 nm) ), the activity of thioguanine (6-TG) to inhibit PLpro enzyme (IC50) was calculated according to the fluorescence intensity, and the inhibition curve was fitted using GraphPad Prism. For fitting results, see figure 2 .
Embodiment 3
[0056] Example 3: P3 laboratory tested the inhibitory activity of thioguanine (6-TG) on the new coronavirus
[0057] (1) Compound cytotoxicity test:
[0058] Will 1x10 4 Vero cells (from: VERO cells from the Cell Resource Center, Shanghai Academy of Biological Sciences, Chinese Academy of Sciences) were seeded in a 96-well plate, set up three duplicate wells, grown at 37 degrees for 20-24 hours, removed the medium, and added 100 μL containing different concentrations. Compounds (100, 50, 25, 12.5, 6.25, 3.125, 1.56, 0 μM) were grown in new medium for 72 hours, with DMSO as a control, using MTS / CCK8 reagent (the above reagents were purchased from: Beijing Soleibao Technology Co., Ltd., Cat. No. CK04-500T), cell viability was measured, and compound cytotoxicity (CC50) was calculated.
[0059] (2) Compound virus inhibition activity test:
[0060] Will 1x10 4 Vero cells were seeded in 96-well plates, three replicate wells were set up, and they were grown at 37°C for 20-24 hour...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com