Aminomethylbenzoic acid sodium chloride injection and preparation method thereof
A technology of aminotoluic acid sodium chloride and aminotoluic acid, applied in the field of injection, can solve problems such as lack of application of aminotoluic acid
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
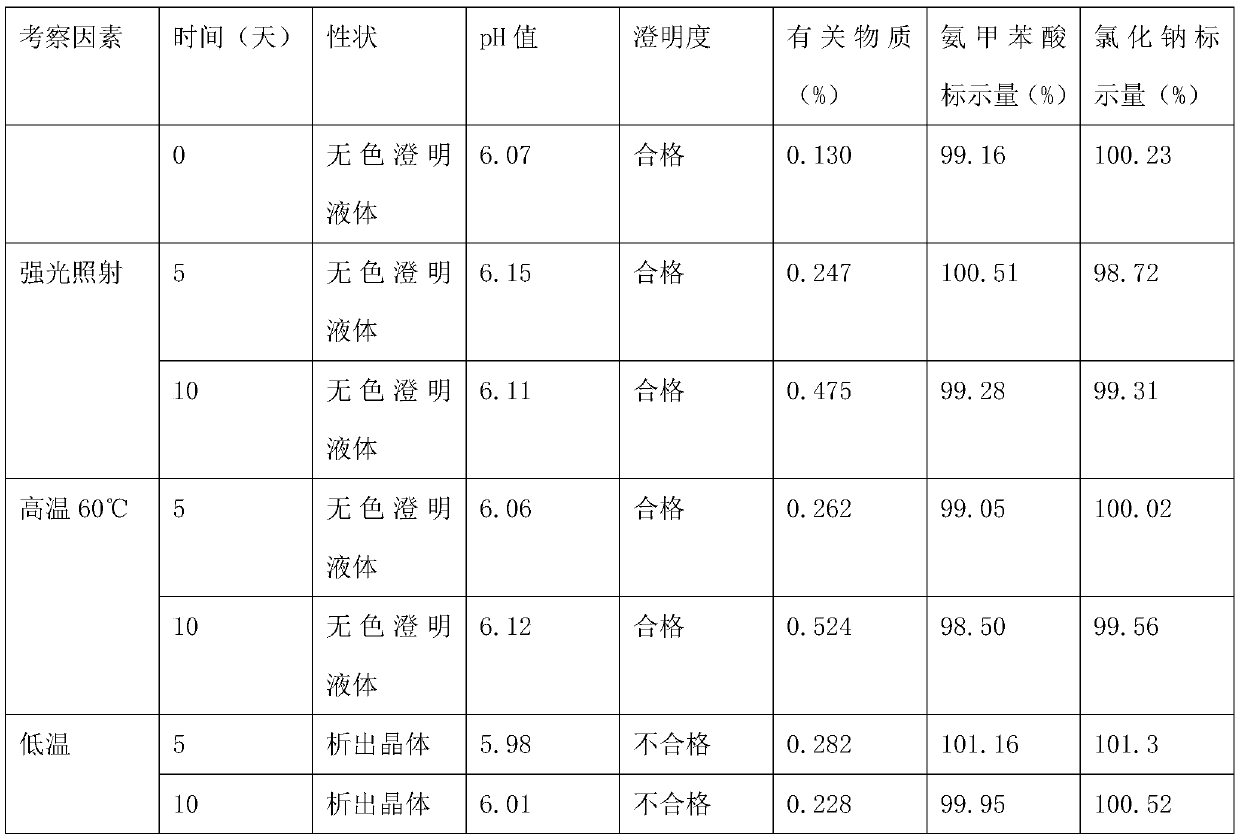
[0016] An aminotoluic acid sodium chloride injection, the raw material of this injection consists of: aminotoluic acid 5kg, sodium chloride 26kg, activated carbon 1.1kg, water for injection added to 3000000ml;
[0017] The preparation method of described aminomethylbenzoic acid sodium chloride injection is:
[0018] (1) Weighing: select raw materials according to the formula;
[0019] (2) Concentrated formulation: take an appropriate amount of water for injection, heat and stir the prescribed amount of sodium chloride to dissolve, add 0.1-0.3% (w / v) activated carbon (for injection), and boil for 15 minutes;
[0020] (3) Filtration: Cool the medicinal liquid prepared in step (2) to 55±5° C., decarbonize and filter it into the dilution tank;
[0021] (3) Dilute formulation: dilute the prescribed amount of aminomethylbenzoic acid with water for injection, add it to the sodium chloride solution in the dilute formulation tank, stir well to dissolve, and then add water for injectio...
Embodiment 2
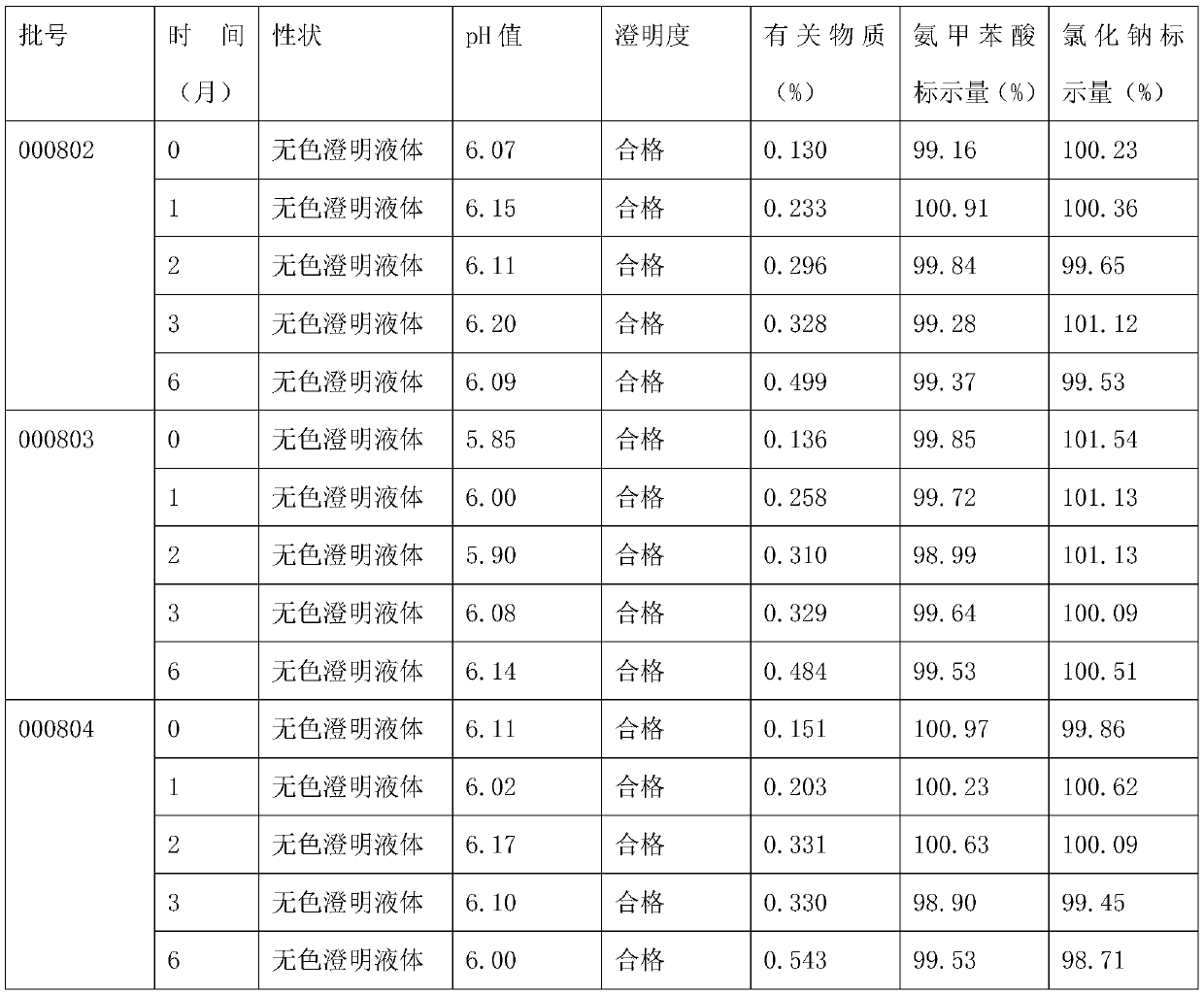
[0026] A kind of aminotoluic acid sodium chloride injection, the raw material of this injection consists of: aminotoluic acid 7kg, sodium chloride 28kg, activated carbon 1.3kg, water for injection added to 3000000ml;
[0027] The preparation method of described aminomethylbenzoic acid sodium chloride injection is:
[0028] (1) Weighing: select raw materials according to the formula;
[0029] (2) Concentrated formulation: take an appropriate amount of water for injection, heat and stir the prescribed amount of sodium chloride to dissolve, add 0.1-0.3% (w / v) activated carbon (for injection), and boil for 15 minutes;
[0030] (3) Filtration: Cool the medicinal liquid prepared in step (2) to 55±5° C., decarbonize and filter it into the dilution tank;
[0031] (3) Dilute formulation: dilute the prescribed amount of aminomethylbenzoic acid with water for injection, add it to the sodium chloride solution in the dilute formulation tank, stir well to dissolve, and then add water for i...
Embodiment 3
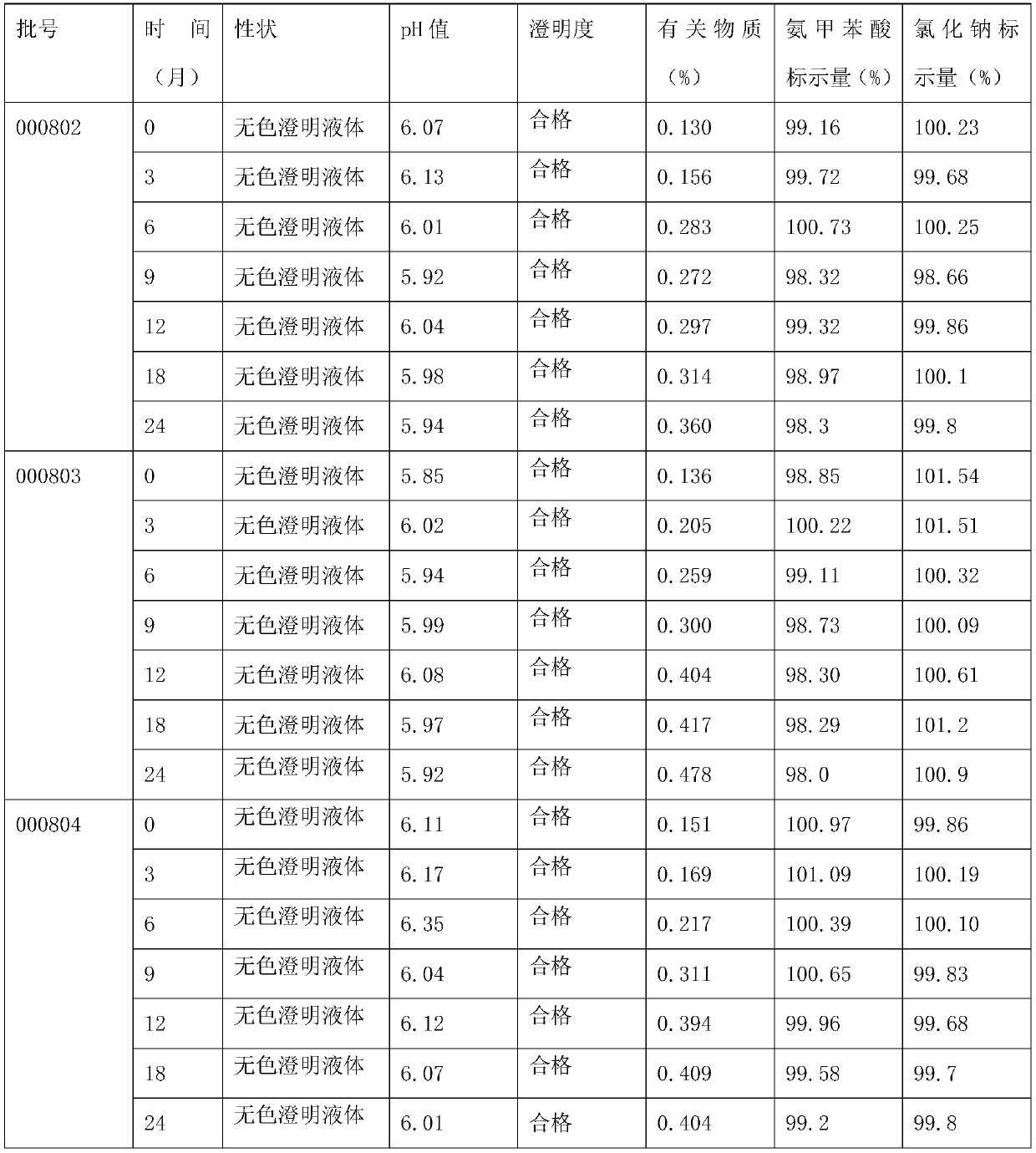
[0036] A kind of aminotoluic acid sodium chloride injection, the raw material of this injection consists of: aminotoluic acid 6kg, sodium chloride 27kg, activated carbon 1.2kg, water for injection added to 3000000ml;
[0037] The preparation method of described aminomethylbenzoic acid sodium chloride injection is:
[0038] (1) Weighing: select raw materials according to the formula;
[0039] (2) Concentrated formulation: take an appropriate amount of water for injection, heat and stir the prescribed amount of sodium chloride to dissolve, add 0.1-0.3% (w / v) activated carbon (for injection), and boil for 15 minutes;
[0040] (3) Filtration: Cool the medicinal liquid prepared in step (2) to 55±5° C., decarbonize and filter it into the dilution tank;
[0041] (3) Dilute formulation: dilute the prescribed amount of aminomethylbenzoic acid with water for injection, add it to the sodium chloride solution in the dilute formulation tank, stir well to dissolve, and then add water for i...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com