Capecitabine determination and application thereof
A capecitabine and determination method technology, applied in the directions of measuring devices, instruments, scientific instruments, etc., can solve problems such as interference of n-amyl alcohol content, and achieve the effects of low equipment requirements, simple operation and quality assurance
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0059] Chromatographic conditions for direct injection method:
[0060] Column temperature: programmed temperature rise: the initial temperature is 60°C and kept for 2 minutes, then the temperature is raised to 200°C at a rate of 15°C per minute and kept for 5 minutes.
[0061] Injection port temperature: 70°C;
[0062] Flame ionization detector temperature: 250°C;
[0063] Carrier gas: nitrogen;
[0064] Carrier gas flow rate: 2.7ml / min;
[0065] Split ratio: 10:1.
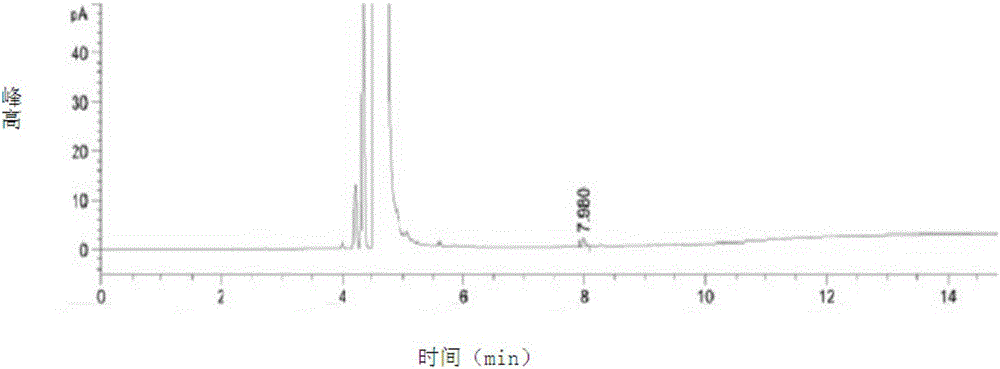
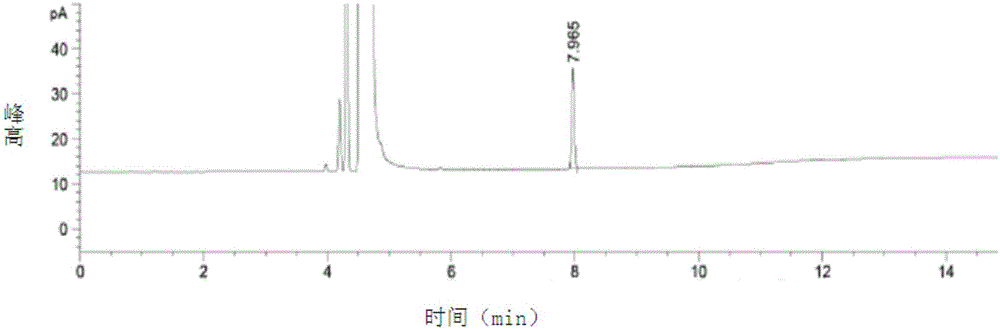
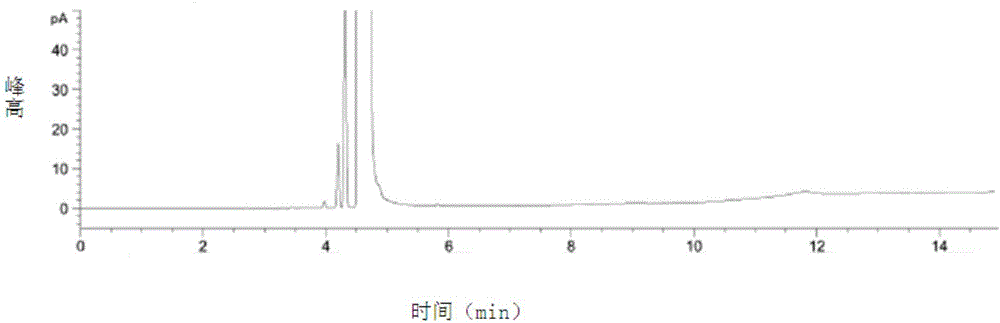
[0066] 1. Detection
[0067] A, preparation of test solution: get capecitabine sample, dissolve and be diluted into the solution of 0.01g / ml with dichloromethane, mix, and set aside;
[0068]B, preparation of reference substance solution: get the residual solvent sample in capecitabine, dilute with dichloromethane solution to make a 500 μg / ml solution, mix well, and set aside;
[0069] C, detection method: get each 1 μ l of need testing solution and reference substance solution, inject in the gas chromatogr...
Embodiment 2
[0112] In the same way as in Example 1, the solvent for adjusting need testing solution and reference substance solution is acetone;
[0113] Injection port temperature: 60°C;
[0114] Flame ionization detector temperature: 180°C;
[0115] Carrier gas flow rate: 2.2ml / min;
[0116] Split ratio: 5:1.
[0117] Three batches of test samples were detected according to the above-mentioned assay method, and the results are shown in Table 6.
[0118] Table 6: Test results of three batches of test products
[0119] batch number
Embodiment 3
[0121] In the same way as in Example 1, the solvent for adjusting need testing solution and reference substance solution is normal hexane;
[0122] Injection port temperature: 75°C;
[0123] Flame ionization detector temperature: 250°C;
[0124] Carrier gas flow rate: 3.2ml / min;
[0125] Split ratio: 15:1.
[0126] Three batches of test samples were detected according to the above-mentioned determination method, and the results are shown in Table 7.
[0127] Table 7: Test results of three batches of test products
[0128] batch number
PUM
| Property | Measurement | Unit |
|---|---|---|
| boiling point | aaaaa | aaaaa |
| length | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com