Drug releasing coatings for medical devices
A medical device and device technology, applied in arterial pulsation system, coated balloon catheter, rapid delivery of therapeutic agents to specific tissues or body cavities to treat diseases, can solve complications, easy hydrolysis or oxidation, and moisture sensitivity And other issues
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment A
[0284] Preparation of coating solution (if necessary, add not more than 10% by volume of a small amount of water sufficient to dissolve all solutes):
[0285] Formulation 1.1 - Mix 30-90mg rapamycin, 1-2% (based on the weight of rapamycin) butylated hydroxytoluene (BHT), 15-90mg Tween 80, 30-90mg docusate sodium (sulfonate diethylhexyl succinate) and 1-3ml ethanol.
[0286] Formulation 1.2 - Combine 30-90mg rapamycin, 1-2% (based on weight of rapamycin) butylated hydroxyanisole (BHA), 15-90mg Tween 80, 30-90mg docusate sodium (Sodium diethylhexyl sulfosuccinate) and 1-3ml ethanol were mixed.
[0287] Formulation 1.3 - Mix 30-90 mg rapamycin without added BHT or BHA, 15-90 mg Tween 80, 30-90 mg docusate sodium (diethylhexyl sodium sulfosuccinate) and 1-3 ml ethanol .
[0288] Formulation 1.4 - Combine 30-90mg rapamycin, 1-2% (based on the weight of rapamycin) BHA or BHT, 15-90mg Solutol HS 15, 5-30mg sodium lauryl sulfate and 1-3ml ethanol mix.
[0289] Formulation 1.5 - C...
Embodiment B
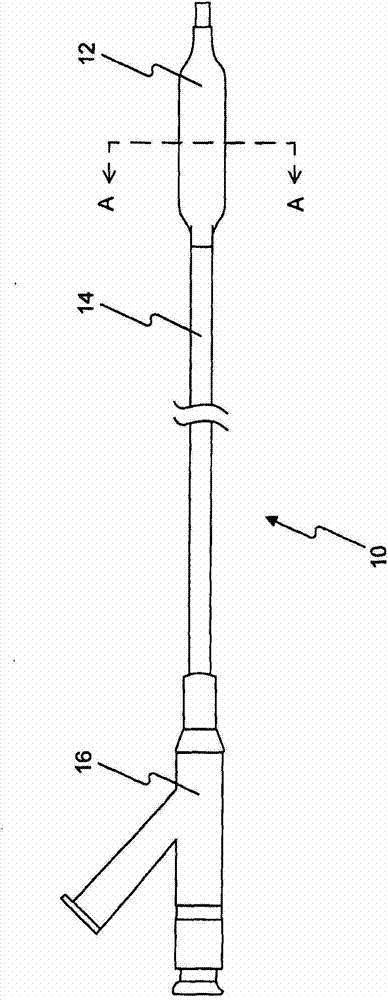
[0311] Five PTCA balloon catheters (3 mm in diameter and 20 mm in length) were coated using the method described in US Patent Application Publication No. 2010-0055294-A1 , which is hereby incorporated by reference in its entirety. Inflate the PTCA balloon catheter at 1-3 atmospheres. Inflated balloons were loaded, sprayed or dipped into the formulations of Example A (1.1-1.3). Then, the balloons were dried, drug-loaded, sprayed or dipped again until balloons with sufficient amount of drug (3 μg / mm) were obtained. The coated balloons are folded, then repackaged and sterilized for analytical testing. For Formulation 1.1 containing BHT, 79% of rapamycin was recovered; for Formulation 1.2 containing BHA, 100% was recovered; and for Formulation 1.3 without BHT or BHA, 14%-50% was recovered. Antioxidants (BHT or BHA) prevent rapamycin from being oxidized or degraded.
Embodiment C
[0313] Six PTCA balloon catheters (3.5 and 3.0 mm in diameter and 20 mm in length) were inflated at 1-3 atmospheres. The inflated balloons were loaded with formulations 1.1-1.25 in Example A. A sufficient amount of drug (3-4 μg / mm2) was obtained on the balloon. The inflated balloon is folded and allowed to dry. The coated folded balloon is then repackaged, sterilized, and optionally vacuum dried for animal testing.
[0314] step : Insertion of coated PTCA balloon catheters into target sites (LAD, LCX, and RCA) in the coronary vessels of 25-45 lb pigs. The balloon is inflated to about 12 atmospheres of pressure. The overdraw ratio (the ratio of balloon diameter to tube diameter) is about 1.15-1.40. During 30-60 seconds of inflation, the drug is delivered to the target tissue. Then, the balloon catheter is deflated and recovered from the animal. Target vessels were harvested 0.25-24 hours after the procedure. The drug content in the target tissue and the residual drug r...
PUM
| Property | Measurement | Unit |
|---|---|---|
| melting point | aaaaa | aaaaa |
| thickness | aaaaa | aaaaa |
| melting point | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com