Modified pictet-spengler reaction and products prepared therefrom
a technology of pictet spengler and product, applied in the field of pictet spengler reaction, can solve the problems of affecting the reaction yield and requiring several days to perform, and achieve the effect of increasing the yield of the desired diastereomer
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
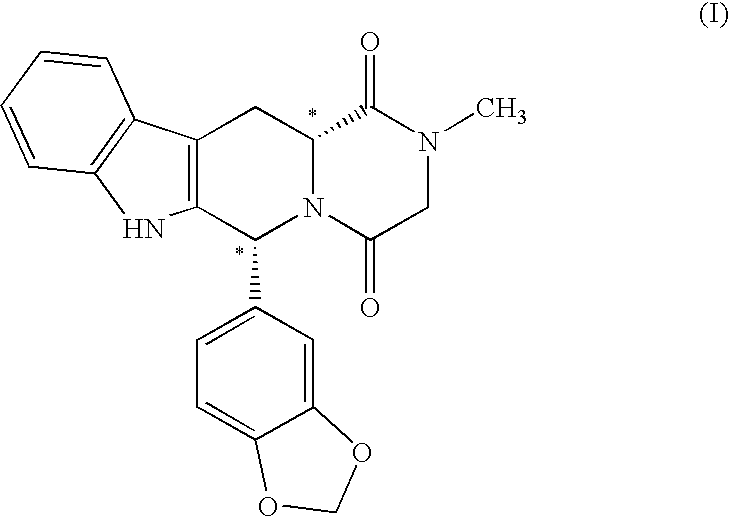
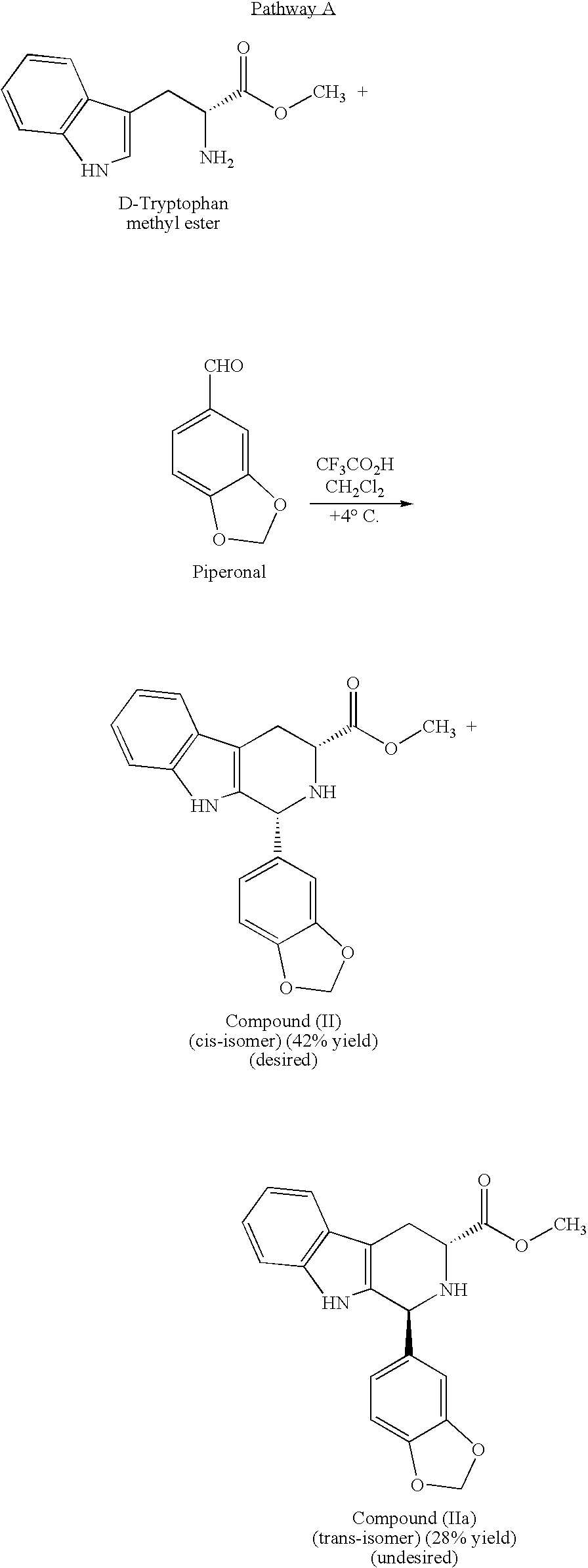
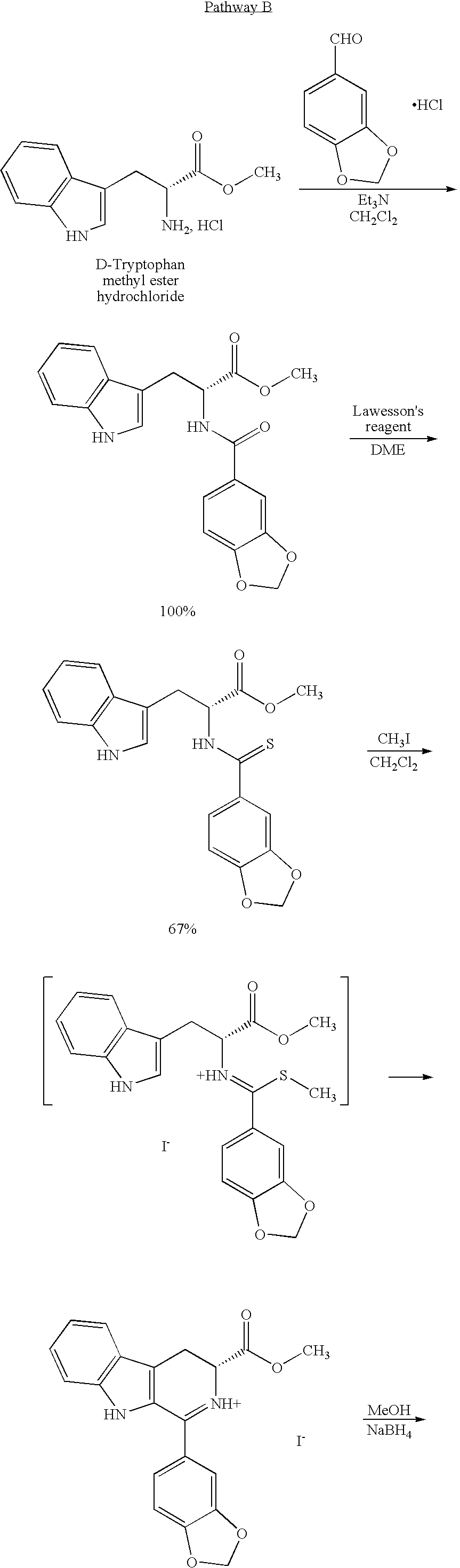
[0019]The present invention is directed to a method of preparing a desired diastereomer of a polycyclic compound having two asymmetric carbon atoms as members of a ring system. The method utilizes an improved Pictet-Spengler reaction that provides a desired tetrahydro-β-carboline diastereomer in high yield, high purity, and in a short process time. The improved Pictet-Spengler reaction also avoids the use of TFA in the reaction.
[0020]Although the synthesis of Compounds (I) and (II) are particularly discussed herein, the present method is not limited to these compounds. The present method also can be used to synthesize the desired diastereomer of other tetrahydro-β-carbolines by a judicious selection of starting tryptophan ester, e.g., the D- or L-form, the starting aldehyde, and the reaction solvents utilized in the present modified Pictet-Spengler cyclization reaction.
[0021]Typically, the Pictet-Spengler reaction proceeds through generation of an imine under neutral conditions, the...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com