Recombinant antibodies for the detection and neutralization of anthrax toxin
a technology of neutralizing antibodies and anthrax, which is applied in the field of antibody engineering, can solve the problems of antibodies not having the affinity and avidity required for widespread use, and the method of prophylaxis treatment of patients exposed to bacillus anthracis /i>cannot achieve the effect of reducing the risk of infection
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
examples
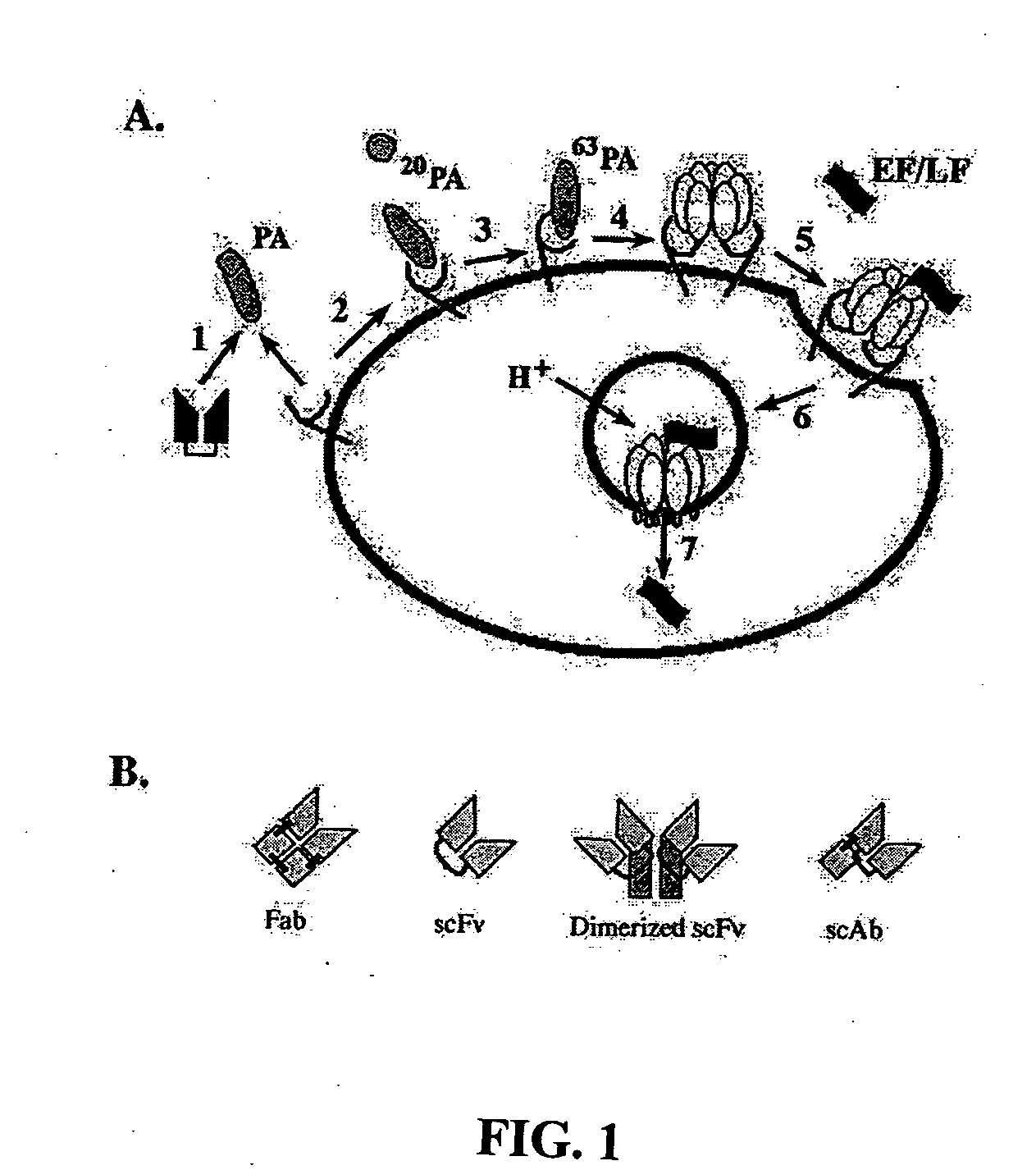
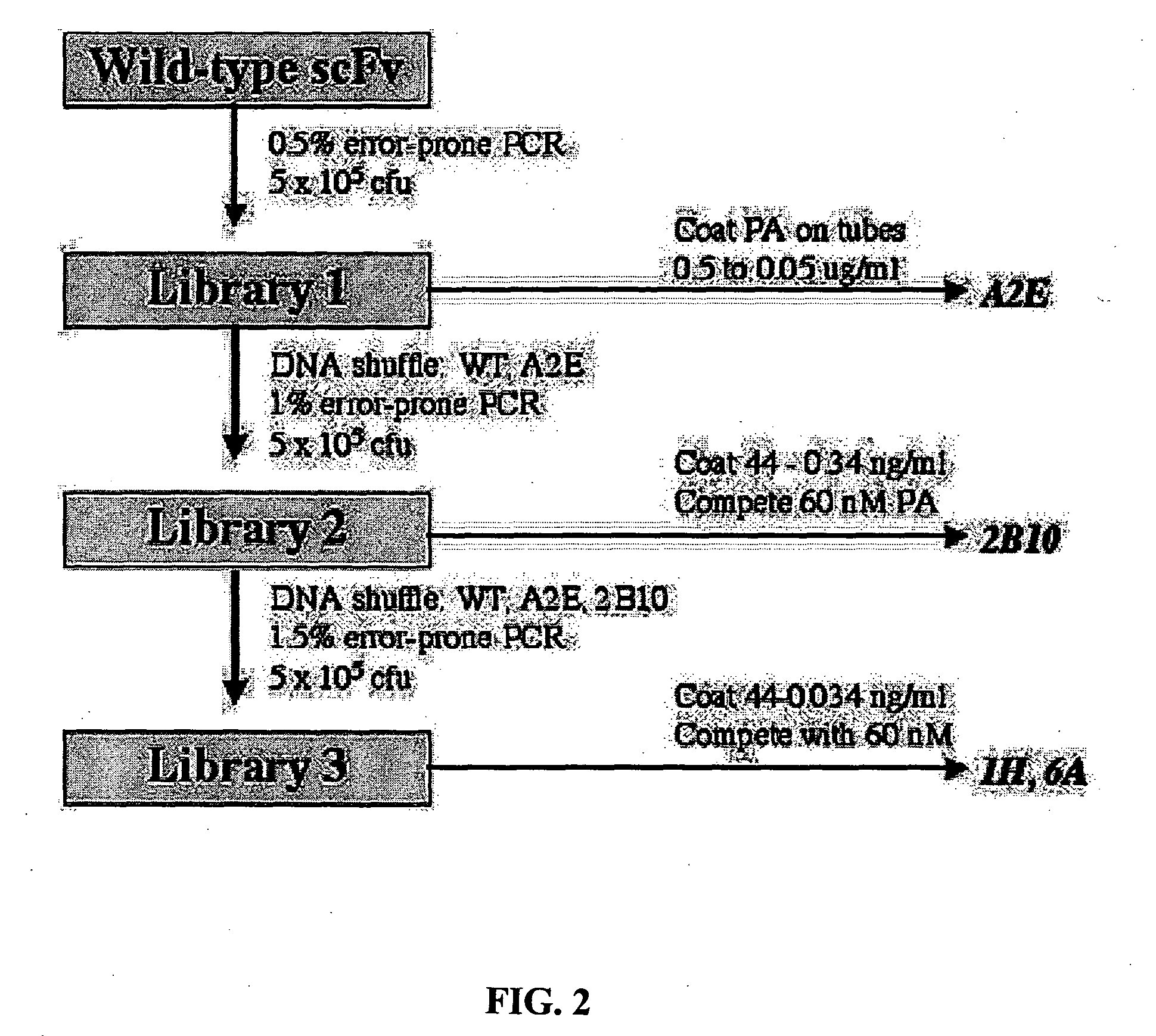
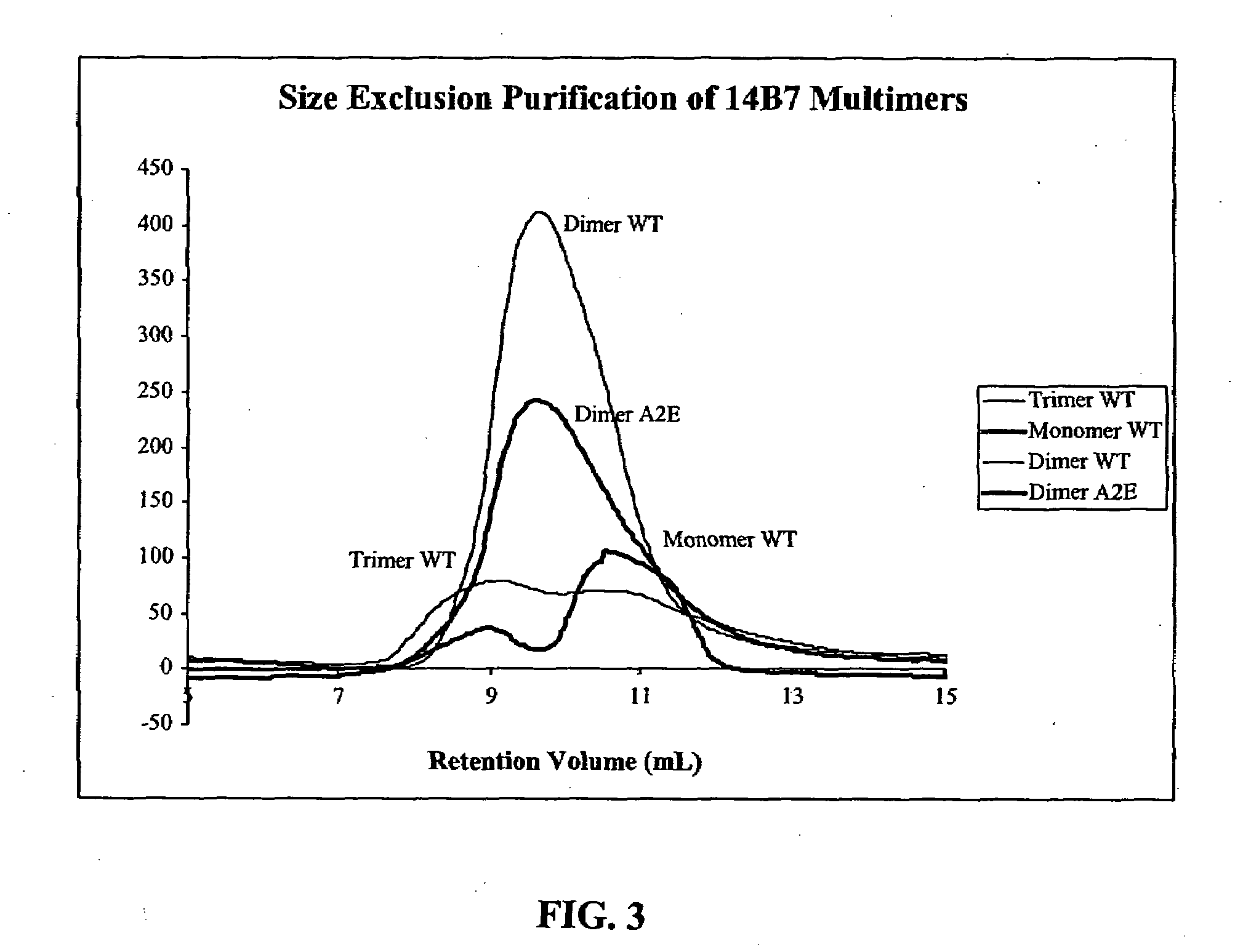
[0056]Cloning from Hybridomas. The heavy and light variable chain regions are cloned from anti-PA hybridoma 14B7 via RT-PCR (Krebber A, Bornhauser S, Burmester J, Honegger A, Willuda J, et al. 1997. Reliable cloning of functional antibody variable domains from hybridomas and spleen cell repertoires employing a reengineered phage display system. J Immunol Methods 201: 35-55). VH and VL genes are joined by overlap PCR and cloned into pAK100 phage display vector using 5 prime and 3 prime SfiI sites. Single colonies in E. coli strain are grown in a 96-well plate, and phage displaying scFv are produced and screened by ELISA to identify PA-reactive clones.
[0057]Antibody Affinity Maturation. Error-prone libraries of the 14B7 scFv gene are constructed using manganese and biased nucleotide ratios (Daugherty P, Chen G, Iverson B, Georgiou G. 2000. Quantitative analysis of the effect of the mutation frequency on the affinity maturation of antibodies. Proc Natl Acad Sci USA In press). DNA shuff...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Atomic weight | aaaaa | aaaaa |
| Dissociation constant | aaaaa | aaaaa |
| Dissociation constant | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com