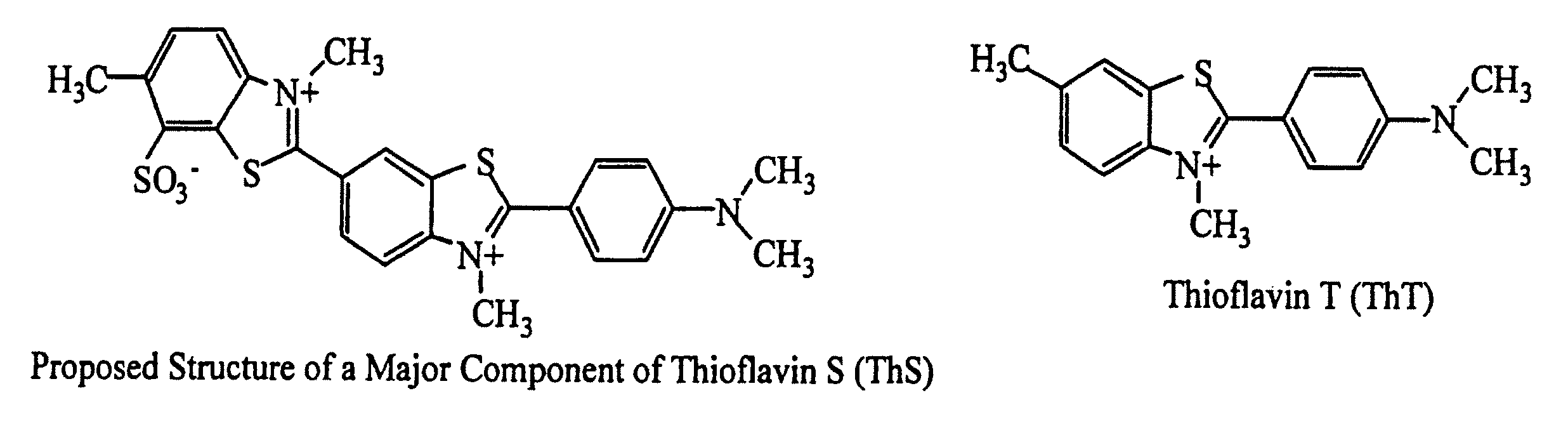
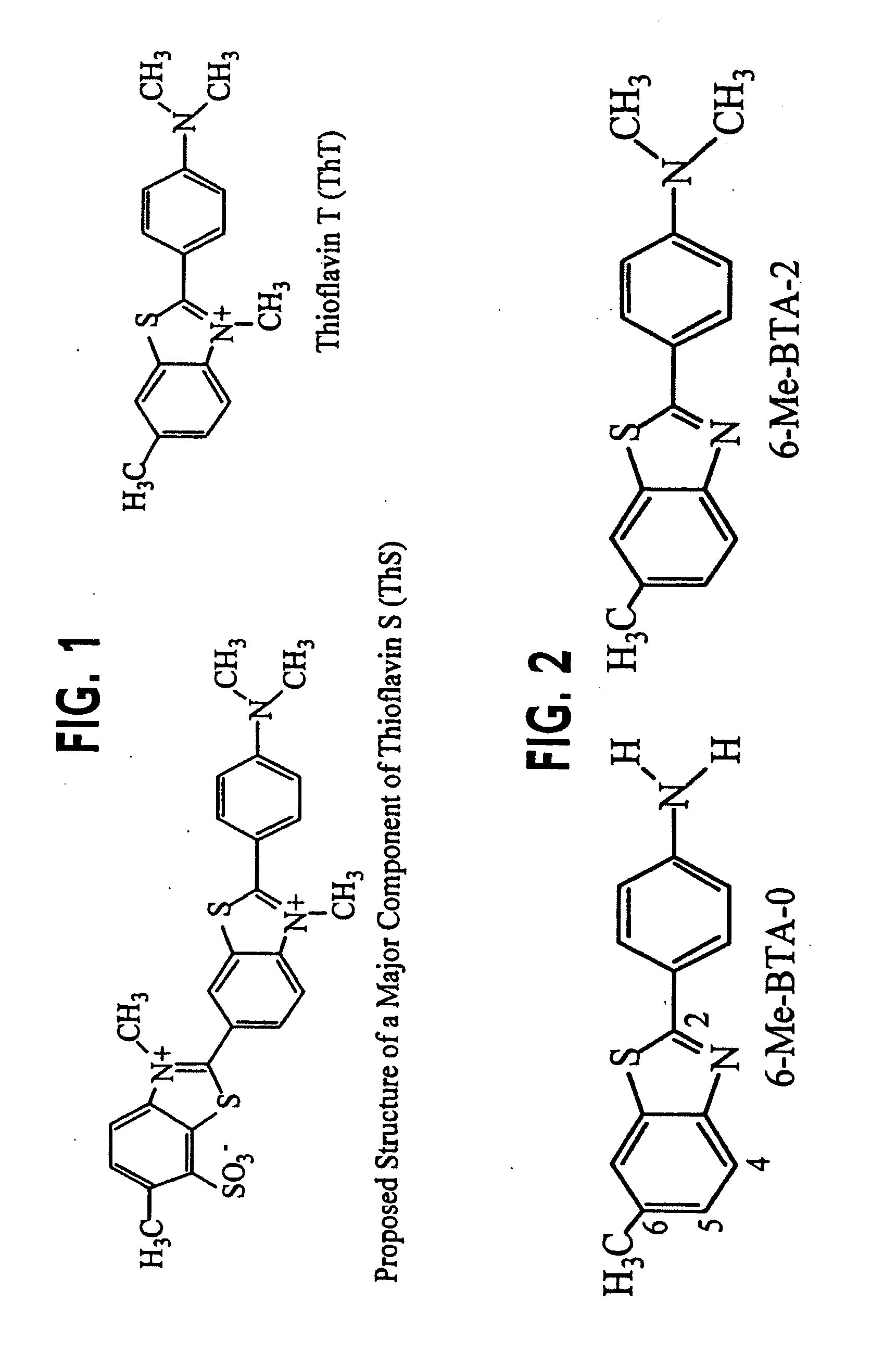
Thioflavin derivatives for use in the antemortem diagnosis of alzheimers disease and in vivo imaging and prevention of amyloid deposition
a technology of thioflavin derivatives and in vivo imaging, which is applied in the direction of drug compositions, organic chemistry, nervous disorders, etc., can solve the problems of delayed assessment of neuritic plaque counts, difficulty, and unknown time of ons
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
examples
[0094]All of the reagents used in the synthesis were purchased from Aldrich Chemical Company and used without further purification. Melting points were determined on MeI-TEMP II and were uncorrected. The 1H NMR spectra of all compounds were measured on Bruker 300 using TMS as internal reference and were in agreement with the assigned structures. The TLC was performed using Silica Gel 60 F254 from EM Sciences and detected under UV lamp. Flash chromatography was performed on silica gel 60 (230-400 mesh. purchased from Mallinckrodt Company. The reverse phase TLC were purchased from Whiteman Company.
synthesis examples
Example 1
Synthesis of Primuline Base Derivatives
[0095]Route 1: Example of the synthesis of Primuline compounds is according to the reaction scheme shown below:
[0096]The primuline derivatives are prepared based on Schubert's method (Schubert, M. Zur Kenntnis der Dehydrothiotoluidin- and Primulin-sulfosäuren, Justus Liebigs Ann. Chem. 558, 10-33, 1947) through condensation of 2-amino-5-methylthiophenol with 2-(p-nitrophenyl)-benzothiazole-6-carboxylic chloride and subsequent reduction of the nitro group with tin chloride in ethanol. Substituted derivatives of primuline base are synthesized with the appropriate substituted p-nitrobenzoylchlorides and R7-R10 substituted 2-aminothiophenol.
[0097]Following the same strategy as above, the other claimed primulin derivatives may be synthesized by substituting the appropriate substituted 3-mercapto-4-aminobenzoic acid derivative (e.g. 2-, 5-, or 6-methyl-3-mercapto-4-aminobenzoic acid), the appropriate 4-nitro-benzoyl chloride derivative (e.g....
example 2
Synthesis of 2-[2-(4′-aminophenyl)-ethylenyl)-benzothiazole derivatives
[0098]Route 3: Example of the synthesis of BTEA-0, 1, 2 and BTAA-0, 1, 2, which are representative of the group of BTEA and BTAA compounds was according to the reaction scheme shown below:
(a) Trans-2-(4-Nitrophenylethenyl)benzothiazole (11)
[0099]trans-4-Nitrocinnamyl chloride 10 (1.77 g, 9.5 mmol, 1.2 eq.) in DMF (20 ml) was added dropwise to a solution of 2-aminothiophenol 9 (1.0 g, 8.0 mmol) in DMF (15 ml) at room temperature. The reaction mixture was stirred at room temperature for overnight. The reaction mixture was poured into a solution of 10% sodium carbonate (100 ml). The participate was collected by filtration under reduced pressure. Recrystallization from methanol gave 1.92 g (85.1%) of the product 11.
(b) 2-(4-Aminophenylethenyl)benzothiazole (12)
[0100]A mixture of 2-(4-nitrophenylethenyl)benthiazole 11 (500 mg, 1.7 mmol) and tin(II) chloride dihydrate (1.18 g, 5.2 mmol) in anhydrous ethanol (20 ml) was...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com