Stable polypeptide protein covalent inhibitor targeting anti-apoptotic protein BFL-1
A BFL-1, anti-apoptotic protein technology, applied in the direction of peptide/protein components, medical preparations containing active ingredients, peptides, etc., can solve problems such as poor efficacy, broaden the scope of application, and solve drug resistance of tumor cells Effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0029] The present invention adopts the sulfonium salt stabilized polypeptide technology reported in the previous literature (D.Wang, M.Yu, et al.Chem.Sci.10, 4966-4972), through the methionine, cysteine and dialkylene on the polypeptide The sulfonium salt cyclic peptide can be reacted with an alkylation reagent, which can not only stabilize the polypeptide, but also covalently modify the cysteine on the interaction site of the target protein, block the combination of anti-apoptotic proteins and pro-apoptotic proteins, and promote The release of apoptotic proteins promotes the apoptosis of tumor cells.
[0030] The inventors synthesized a number of different polypeptides, and focused on seven of them, as shown in Table 1. Then through the covalent reaction to BFL-1 to screen the polypeptide molecule with good covalent effect. Figure 11It is a schematic diagram of anti-tumor covalent polypeptide inhibitor promoting cell apoptosis through covalent reaction with BFL-1.
[...
Embodiment 2
[0035] Preparation and separation and purification steps of the polypeptide of embodiment 2:
[0036] According to the amino acid sequence of solid-phase synthesis of polypeptides, the core steps of preparing the above-mentioned stable polypeptides are as follows (taking B4-MC as an example):
[0037] Specific operation steps ( figure 1 )for:
[0038] (1) Polypeptide solid-phase synthesis: Weigh Rink amide MBHA resin into a peptide tube, add dichloromethane (DCM), and swell with nitrogen gas for 30 minutes. Add 50% (v / v) morpholine in N,N-dimethylformamide (DMF) solution, blow nitrogen gas for 30 minutes, and remove the Fmoc protecting group. After washing the resin alternately with DMF and DCM, the prepared Fmoc-AA-OH (5eq, 0.4M, DMF) solution, 6-chlorobenzotriazole-1,1,3,3-tetramethylurea Fluorophosphate ester (HCTU) (5eq, 0.38M, DMF) solution and N,N-diisopropylethylamine (DIPEA) (10eq) were mixed well, then added to the resin and blown with nitrogen for 1h. Take out th...
Embodiment 3
[0041] Example 3 Polypeptide Molecule and Protein BFL-1 Covalent in Vitro
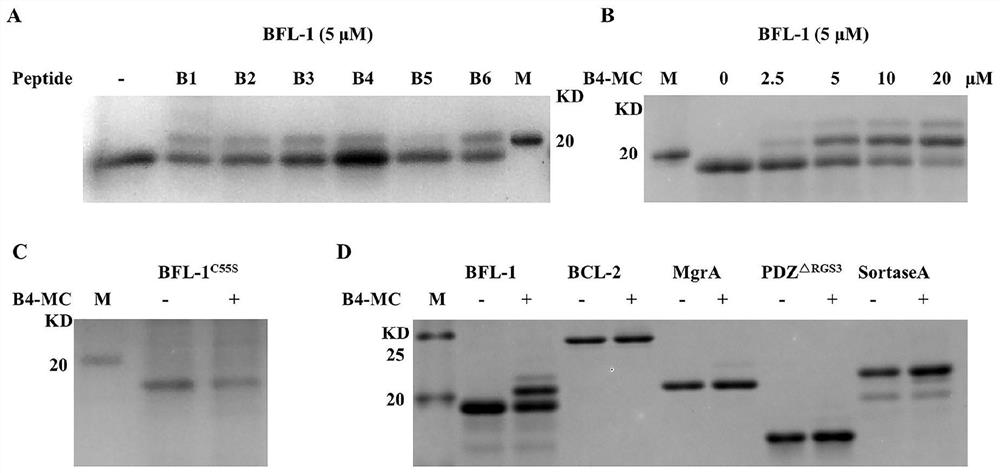
[0042] Methionine and cysteine on different polypeptides react with dialkylating reagents to form sulfonium salt cyclic peptides and co-incubate with proteins respectively, and the protein and sulfonium salt polypeptides undergo a covalent reaction, and obvious upward shifting bands can be seen . The covalent reaction between protein and peptide is dose-dependent. The polypeptide has good specificity, and it mainly reacts covalently with the cysteine at position C55 of BFL-1. The cysteine at position 55 of BFL-1 is mutated to serine, and the polypeptide basically does not covalently interact with BFL-1. valence reaction. In addition, some proteins containing cysteine were selected, and the sulfonium salt polypeptide did not covalently react with it ( figure 2 ).
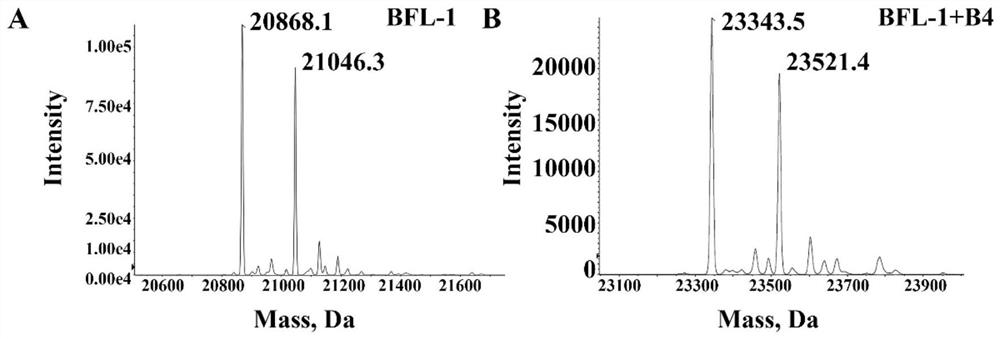
[0043] The polypeptide B4-MC was selected to be co-incubated with the protein BFL-1, and the primary mass spectrometry verified...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap