Mphosph1 peptides and vaccines including the same
An amino acid, selected from the technology, applied in the field of cancer treatment, bioscience
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment
[0444] Materials and methods
[0445] cell line
[0446] T2, HLA-A*0201-positive B-lymphoblastoid cell line, HLA-A*0206-positive B-lymphoblastoid cell line, HT1376, J82, COS7 and UM-UC3 were purchased from ATCC. MKN-45 was purchased from JCRB.
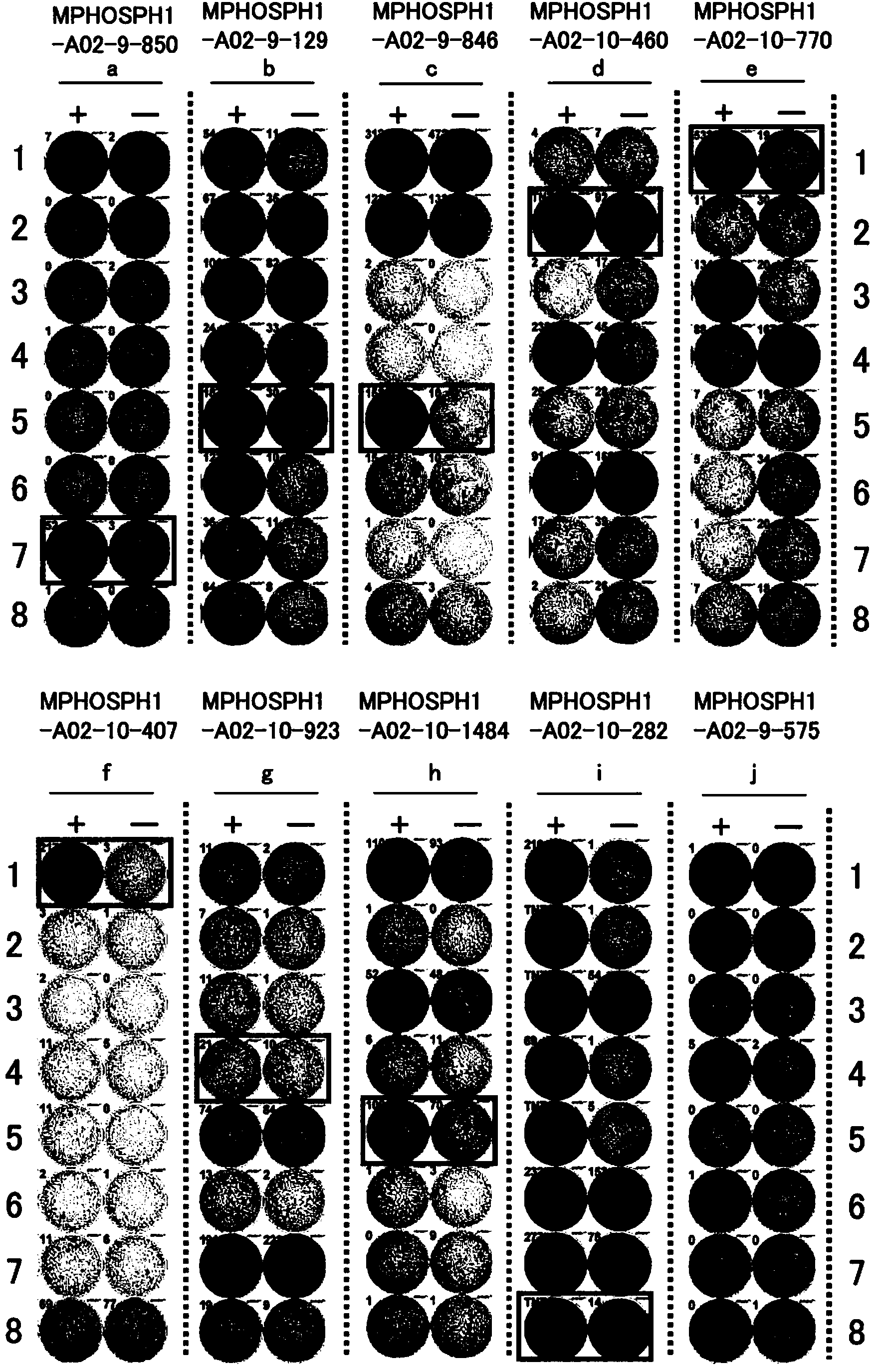
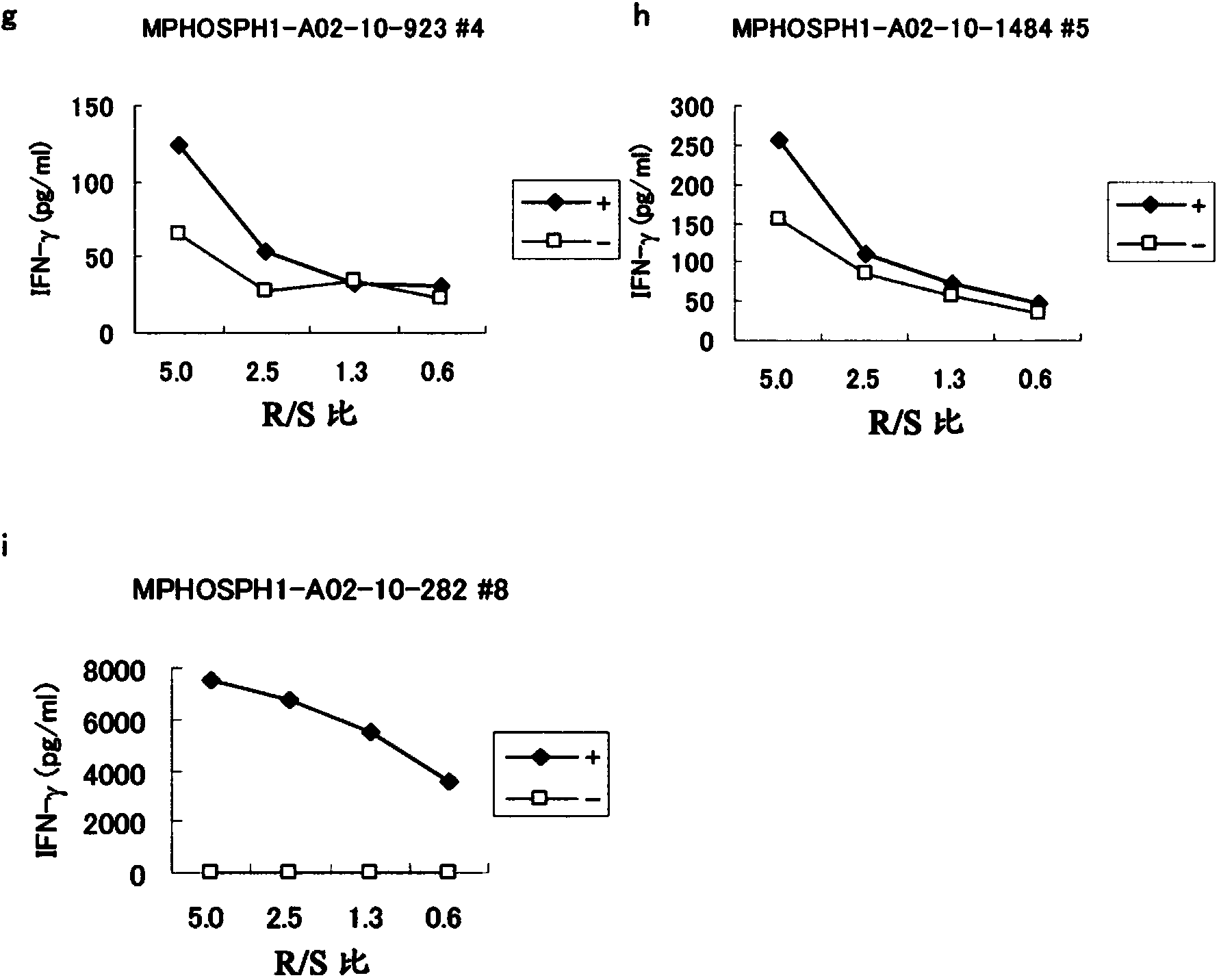
[0447] Candidate selection for peptides derived from MPHOSPH1
[0448] Using the binding prediction software "BIMAS" (http: / / www-bimas.cit.nih.gov / molbio / hla_bind) (Parker et al., J Immunol 1994,152(1):163-75; Kuzushima et al., Blood 2001, 98(6):1872-81) predicted 9- and 10-mer peptides derived from MPHOSPH1 (GenBank Accession No: EAW63006 (SEQ ID NO: 40)) that bind the HLA-A*0201 molecule. These peptides were synthesized by BioSynthesis (Lewisville, Texas) according to standard solid phase synthesis and purified by reverse phase high performance liquid chromatography (HPLC). The purity (>90%) and identity of the peptides were determined by analytical HPLC and mass spectrometry, respectively. Peptides were dissolved in dimet...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com