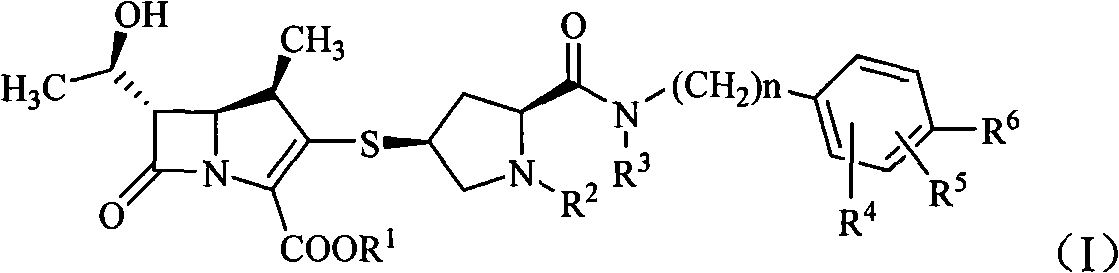
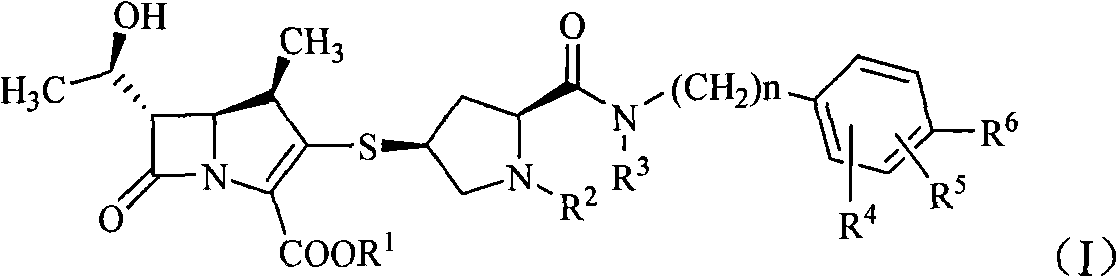
Formamide alkylbenzene substituted mercapto pyrrolidine carbapenem compounds
A compound, methyl technology, applied in the field of mercaptopyrrolidine carbapenem compounds substituted by carboxamide alkylbenzene, can solve the problems of not meeting clinical needs, weak antibacterial activity of MRSA, and low clinical utilization
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0090] The preparation of embodiment 1 (2S, 4S)-4-mercapto-2-formyl [N-benzyl-2-methylpropyl-2-amino]-1-(tert-butoxycarbonyl) pyrrolidine prepare
[0091] 14.5 g (50 mmol) of (2S,4S)-4-acetylthio-2-carboxy-1-(tert-butoxycarbonyl)pyrrolidine and 150 ml of anhydrous tetrahydrofuran were added to a dry reaction flask. Under the protection of nitrogen, 13g (80mmol) of 1,1-carbonyldiimidazole was added at room temperature, reacted for 0.5h, and 8.3g (51mmol) of N-benzyl-2-methylpropyl-2-amine was added below 0°C. Tetrahydrofuran solution 100ml, continue to react for 1h. Then 60ml of 1mol / L hydrochloric acid was added dropwise, extracted with ethyl acetate (100ml×2), the organic phase was washed with water and saturated sodium chloride solution successively, concentrated under reduced pressure, the residue was added with 150ml of 3mol / L hydrochloric acid, stirred for 2h, and The dilute alkali solution was adjusted to be alkaline, and a solid was precipitated, which was recrysta...
Embodiment 2
[0092] Example 2 (2S, 4S)-4-mercapto-2-formyl [N-(4-fluorobenzyl)-2-methylpropyl-2-amino]-1-(tert-butoxycarbonyl)pyrrole Preparation of alkane
[0093] Refer to Example 1 for the specific preparation method. Cast (2S, 4S)-4-acetylthio-2-carboxy-1-(tert-butoxycarbonyl)pyrrolidine 14.5g (50mmol), N-(4-fluorobenzyl)-2-methylpropyl- 2-amine 9.2 g (51 mmol). 16.3 g of the product was obtained, yield: 79.3%.
Embodiment 3
[0094] Example 3 (4R, 5S, 6S)-3-[(2S, 4S)-2-formyl[N-benzyl-2-methylpropyl-2-amino]-1-(tert-butoxycarbonyl ) Pyrrolidin-4-yl]thio-6-[(1R)-1-hydroxyethyl]-4-methyl-7-oxo-1-azabicyclo[3,2,0]hept 2-ene- 2-Carboxylic acid p-nitrobenzyl ester preparation of
[0095] In a dry reaction flask, add (4R, 5S, 6S)-3-diphenoxyphosphoryloxy-6-[(1R)-1-hydroxyethyl]-4-methyl-7-oxo-1- Azabicyclo[3,2,0]hept-2-ene-2-carboxylic acid p-nitrobenzyl ester 11.9g (20mmol) in 120ml of acetonitrile solution, cooled to below -10°C, add 5ml of diisopropylethylamine and (2S, 4S)-4-mercapto-2-formyl [N-benzyl-2-methylpropyl-2-amino]-1-(tert-butoxycarbonyl)pyrrolidine 8.6g (22mmol) Acetonitrile solution 80ml, stirred at 0°C for 15h. After the reaction was completed, 300 ml of ethyl acetate was added to dilute, washed with water and saturated brine in sequence, the organic layer was dried and concentrated to obtain 10.1 g of solid, yield: 68.3%.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com