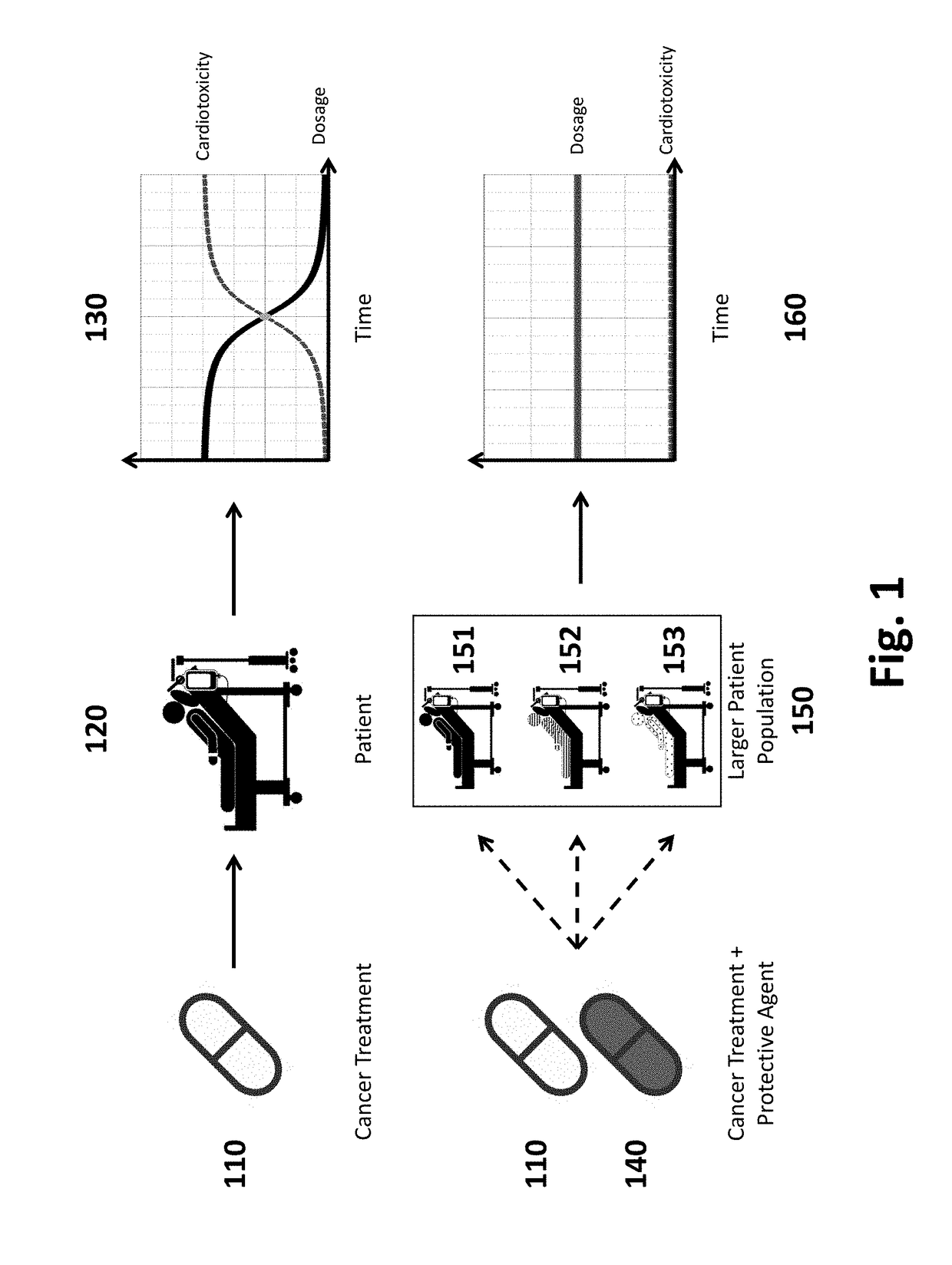
Pharmaceutical compositions and methods for countering chemotherapy induced cardiotoxicity
a technology of chemotherapy and compositions, applied in the direction of drug compositions, enzyme inhibitor ingredients, antibody medical ingredients, etc., can solve the problems of high incidence of heart failure, cell death in both cancer and normal cells, severe limits of the therapeutic potential of these anticancer agents, etc., to reduce the risk of potentially devastating heart failure, prevent heart failure, and improve efficacy and safety
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Provides Long-Term Cardioprotection (Cell Viability)
[0220]Cell samples were prepared by differentiating induced pluripotent stem cells into cardiomyocytes. Cells were cultured for 4 days post-differentiation, changing media at day 3, before performing experiments. Samples were either mock-treated, treated with 1.25 μM doxorubicin, treated with myricetin, or co-treated with 1.25 μM of doxorubicin and myricetin for 72 hours. Following treatment, the samples were incubated Hoeschst 33342 to indicate cell nuclei. Cells were imaged using the INCell Analyzer2200, and images were analyzed to quantify the total number of cells and plotted as a percentage of total cells normalized to control (left), where each data point was obtained from three biological replicates. Representative images (FIG. 3, right) are presented for each sample, where an increase in Hoechst 33342 signal represents an increase ion cell viability.
[0221]Cardiomyocytes were either mock-treated, treated with 1.25 μM doxorub...
example 2
f Myricetin on Doxorubicin-Induced Cardiotoxicity 2 Days Following Treatment (Mitochondrial Toxicity)
[0222]Human iPSC-derived cardiomyocytes were prepared by differentiating induced pluripotent stem cells into cardiomyocytes. Cells were cultured for 4 days post-differentiation, changing media at day 3, before performing experiments. Cardiomyocytes were treated with 1.25 μM doxorubicin (FIG. 4A), or co-treated with 1.25 μM of doxorubicin and 79 μM myricetin (FIG. 4B) for 2 days. Following treatment, the samples were incubated with a tetramethylrhodamine methyl ester (TMRM) dye to indicate mitochondrial health, and Hoechst 33342 to identify cell nuclei. Cells were imaged using the INCell Analyzer2200. Representative images are presented for each sample, wherein a decrease in TMRM signal indicates an increase in mitochondrial toxicity. Myricetin was a potent protector against doxorubicin-induced mitochondrial toxicity, as indicated by a greater TMRM signal in cells co-treated with 1.25...
example 3
f Myricetin on Doxorubicin-Induced Cardiotoxicity 3 Days Following Treatment (Contractility)
[0223]Human iPSC-derived cardiomyocytes were prepared as described above. Samples were either mock-treated, treated with 1.25 μM doxorubicin, treated with 79 μM myricetin, or co-treated with 1.25 μM of doxorubicin and 79 μM myricetin for 72 hours. Following treatment, videos of beating cardiomyocytes were captured using Pulse, and analyzed to quantify beat rate (FIG. 5; left) from plots of cell contraction, where each data point was obtained from three biological replicates. Representative plots of cell contraction (FIG. 5; right) are presented for each sample. Myricetin was a potent protector of cell contractility. Mock-treated cardiomyocytes contracted at 33.33 beats per minute, whereas treatment with 1.25 μM doxorubicin, in the absence of myricetin, completely inhibited contraction. Cardiomyocytes treated with myricetin, or co-treated with myricetin and 1.25 μM doxorubicin, contracted at 3...
PUM
| Property | Measurement | Unit |
|---|---|---|
| time | aaaaa | aaaaa |
| time | aaaaa | aaaaa |
| enantiomeric excess | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com