Methods and devices for the long-term culture of hematopoietic progenitor cells
a technology of hematopoietic progenitor cells and culture methods, which is applied in the field of hematopoietic cells, can solve the problems of inability to maintain viability and pluripotency of such cells for a long time, limited longevity of cultures, and disappointing gene transfer efficiency to date, so as to increase the efficiency of gene transfer when carried out with cells cultured on the matrix
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
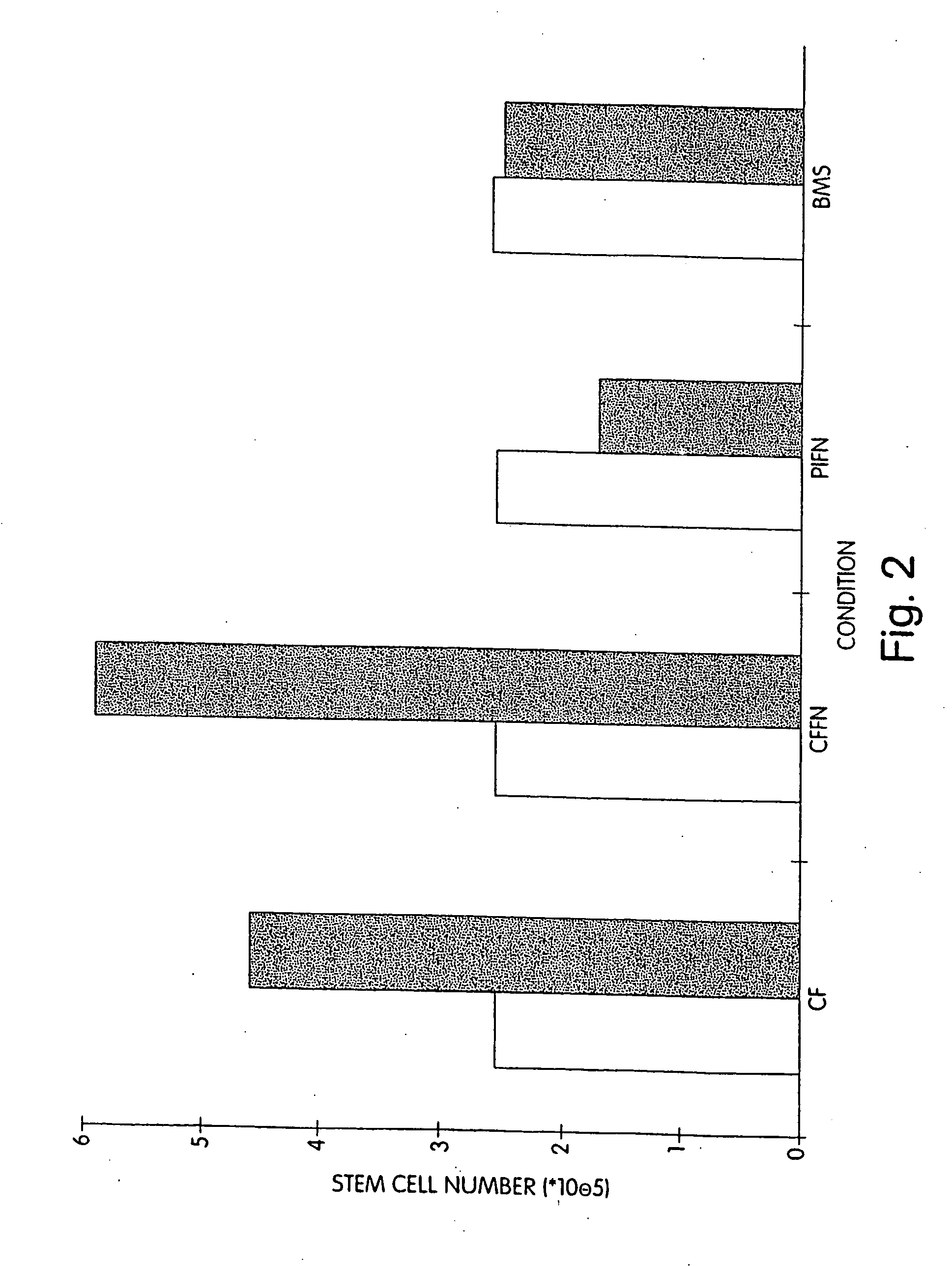
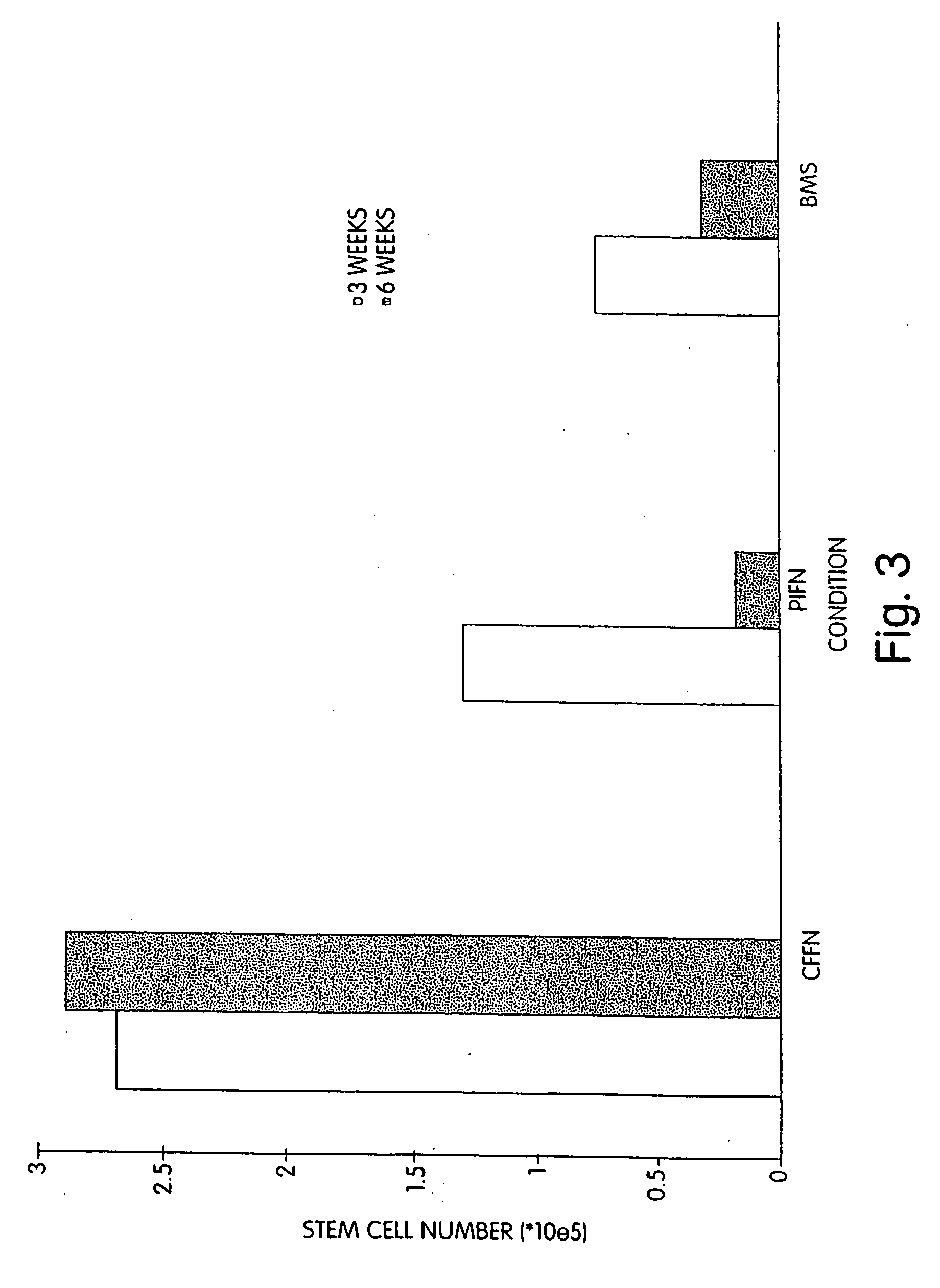
[0096] We performed extended-culture survival studies examining CD34+ HPC cell numbers at 1, 3, and 6 weeks in the absence of supplemented cytokines. Cultures were carried out in fibronectin coated Cellfoam units and compared with bone marrow stroma and fibronectin coated plastic dishes CD34+ HPCs cultured in Cellfoam without cytokine supplementation exhibited enhanced survival and marked enrichment compared to parallel control cultures. The loss of HPCs in control systems supports documentation of their inability to support HPCs without exogenous cytokines. Plastic dish cultures performed similar to BMS. Conversely, at 1 week CD34+ cell counts in Cellfoam were 2.5-3 fold higher than other systems analyzed and had increased 80-110% over input numbers. By 3 and 6 weeks, as many as 6 to 10 times CD34+ cells were detected in Cellfoam versus controls. This increase in cell number was reproducible and in the absence of cytokines. In addition, we were able to count an immature population ...
example 2
[0097] In addition, we evaluated the multipotency of the population of cells isolated from multi-week cytokine cultures. The assays used were conventional methylcellulose colony-formation assays to evaluate myeloid and erythroid colony-forming cells and a published lymphopoiesis assay to evaluate T cell precursor activity. We observed that HPCs isolated from Cellfoam cultures retain red blood cell (RBC) and white blood cell (WBC) colony forming ability to a greater extent than parallel control cultures. In all cultures the CFU-GM and BFU-E were evaluated; the myeloid:erythroid ratio was approximately 2:1. At 3 weeks, Cellfoam cultures yielded up to 31 times as many colonies compared to controls, an increase of 16 fold over input capabilities (see FIG. 4). By 6 weeks, HPCs had lost essentially all of their colony-forming ability in BMS and plastic-fibronectin cultures. HPCs from Cellfoam displayed a 1000 fold greater capacity to produce colonies over control-isolated cells (see FIG. ...
example 3
[0098] The ability of cultured HPCs to foster T-cell lymphopoiesis was assessed in an in vitro T-cell differentiation assay. After termination of Cellfoam and control cultures at 3 and 6 weeks, an aliquot of the combined adherent / non-adherent factions were co-cultured with primary fetal thymic stroma. We evaluated the ability to produce mature T cells as assessed by CD4 and CD8 single positivity and CD4CD8 double positivity antibody staining. When cells were harvested at 3 and 6 weeks from Cellfoam and control cultures and placed in the T-cell assay, only cells recovered from Cellfoam generated T-cell progeny at both time points. Cells recovered from FN / plastic failed to generate T-cell progeny. Cells from BMS cultures generated T-cell progeny at 3 weeks but not at 6 weeks. Progeny derived from Cellfoam included CD4+CD8+ thymocytes, as well as CD4+ and CD8+ cells. Progeny derived from Cellfoam cultures included CD4+CD8+thymocytes as well as CD4+ and CD8+ single positive cells while ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com