Injection of bone marrow-derived cells and medium for angiogenesis
A myeloid cell, angiogenesis technique with applications in enhancing collateral vessel formation) and tissue perfusion. ,Intramyocardial injection of autologous bone marrow and transfected bone marrow cells can solve problems such as gaps
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
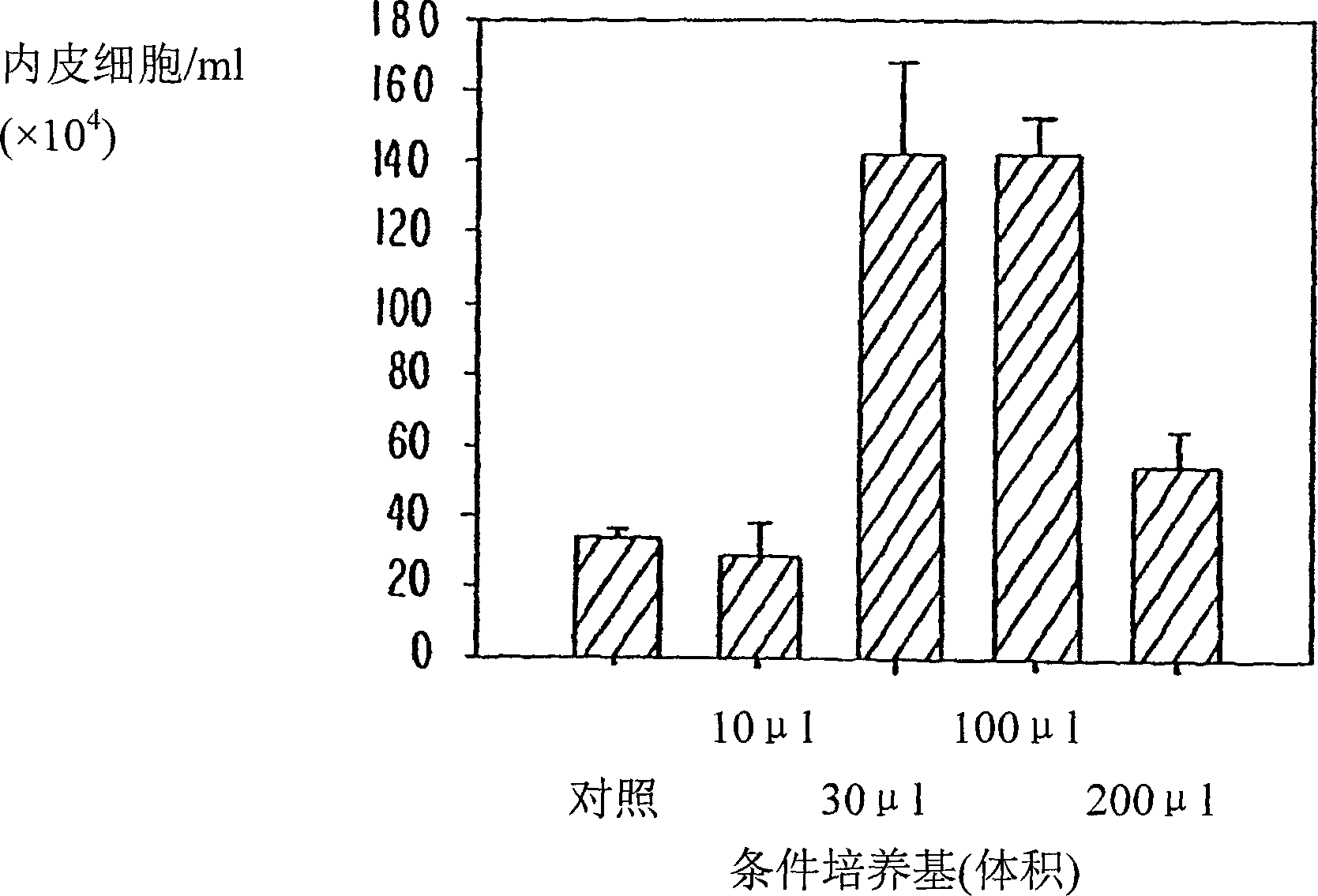
Effects of Bone Marrow Culture Medium on the Proliferation of Endothelial Cells
[0064] A study was performed to determine whether the obtained aspirated porcine autologous bone marrow cells secreted VEGF, a potent angiogenic factor, and MCP-1, which was recently identified as an important angiogenic cofactor. Bone marrow was cultured in vitro for four weeks. Conditioned medium was added to cultured porcine aortic endothelial cells (PAECs), and proliferation was assessed four days later. Conditioned media were analyzed for VEGF and MCP-1 levels by ELISA. During four weeks of culture, BM cells secreted VEGF and MCP-1 such that their concentrations increased in a time-dependent manner. The resulting medium enhanced the proliferation of PAECs in a dose-related manner. The results suggest that BM cells can secrete potent angiogenic factors such as VEGF and MCP-1, and can induce proliferation of vascular endothelial cells.
porcine bone marrow culture
[0065] Bone marrow (BM)...
Embodiment 2
Effect of Hypoxia on Secretion of VEGF by Cultured Porcine Bone Marrow Cells
[0074] It was confirmed that hypoxia significantly increases the expression of VEGF in cultured bone marrow endothelial cells, and the results suggest that exposure to hypoxia in vitro can further increase the expression of hypoxia-induced angiogenic factors, which can further increase the expression of bone marrow to be injected in ischemic muscle tissue. Collateral enhancement effects of cells and their conditioned media. Porcine bone marrow was harvested and successively filtered through 300μ and 200μ stainless steel mesh filters. BMCs were then isolated by Ficoll-Hypaque gradient centrifugation and cultured at 33 °C with CO in T-75 culture flasks 2 is 5%. When the cells became confluent at approximately 7 days, they were trypsinized 1:3 to separate. After 4 weeks in culture, BMCs were either exposed to hypoxic conditions (placed in a chamber containing 1% oxygen) for 24 to 120 hours,...
Embodiment 3
[0076] demonstrated that the conditioned medium of bone marrow cells induces the formation of structural vascular tubes in vitro using a co-culture technique of porcine endothelial cells and vascular smooth muscle cells. This effect on vascular tube formation was not observed in the absence of exposure to bone marrow-conditioned media. The results suggest that myeloid cells and the factors they secrete exert pro-angiogenic effects.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com