Treatment with anti-ErbB2 antibodies
An antibody, receptor technology, applied in the treatment of diseases characterized by ErbB2 overexpression, can solve problems such as stimulating tumor growth
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
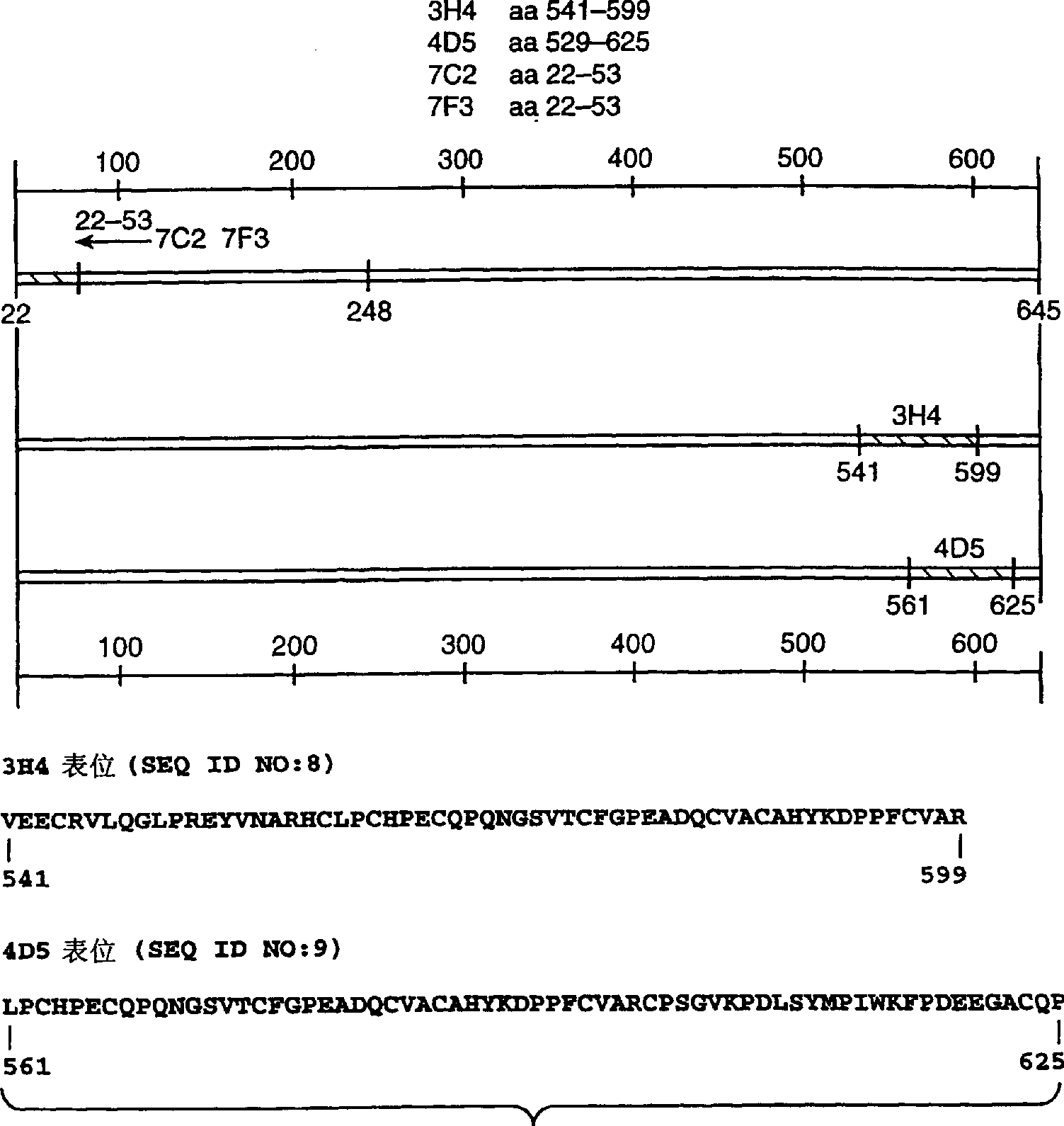
[0154] Materials and methods
[0155] Anti-ErbB2 monoclonal antibody. Press Fendly and other Cancer Res. 50: 1550-1558 (1990) and WO89 / 06692, produced anti-ErbB2 IgG specific for the extracellular domain of ErbB2 1 κ mouse monoclonal antibody 4D5. In short, such as Hudziak et al., Proc. Natl. Acad. Sci. (USA) 84: 7159 (1987) produced NIH3T3 / HER2-3 400 Cells (each cell expresses approximately 1×10 5 ErbB 2 molecules), the cells were harvested with phosphate buffered saline (PBS) containing 25 mM EDTA for immunization of BALB / c mice. At 0, 2, 5, and 7 weeks, mice were injected intraperitoneally containing 10 7 0.5 ml of PBS for cells. In the 9th and 13th weeks, antiserum can immunoprecipitate 32 Mice with p-labeled ErbB2 were injected intraperitoneally with ErbB2 membrane extract purified by malt agglutinin-Sepharose (WGA). Then, 0.1 ml of ErbB2 preparation was injected intravenously, and spleen cells were fused with the mouse myeloma cell line X63-Ag8.653. The ErbB2 binding ability...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com