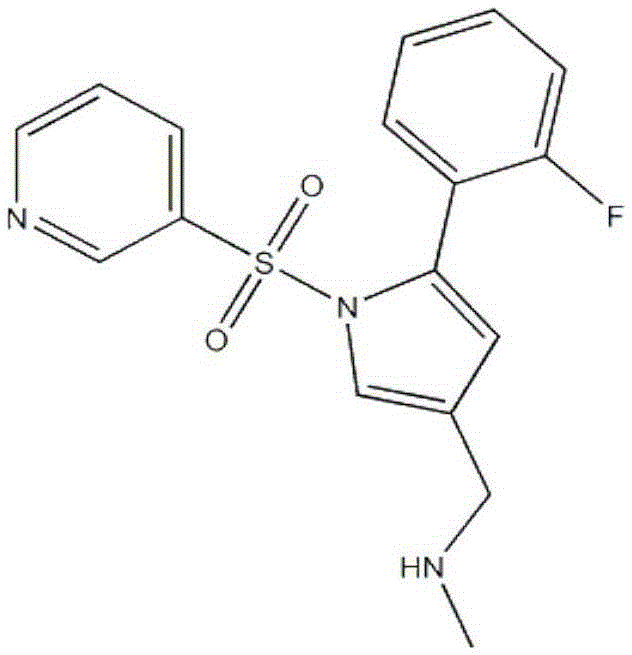
Compound preparation containing vonoprazan and non-steroidal anti-inflammatory drugs
A technology of non-steroidal anti-inflammatory drugs and compound preparations, applied in the field of medicine
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0013]
[0014]
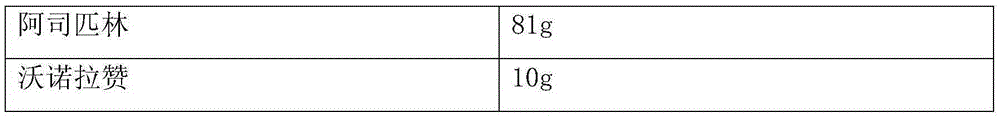
[0015] Preparation method: Mix the prescribed amount of aspirin, tartaric acid and 85g lactose, wet granulate with 10% starch slurry, boil and dry; mix the aspirin granules with 8g microcrystalline cellulose and 7g talcum powder, press into tablets, and wrap the isolation layer Mix Vonorazan, aspartame 0.1g, sodium carboxymethylcellulose 5g and lactose 5g, add water to moisten, wrap around the tablet core, and dry to obtain.
Embodiment 2
[0017] Indomethacin 75mg, Vonorazan 10mg;
[0018] Indomethacin
[0019] Preparation method: Mix the prescription amount of indomethacin, tartaric acid and 90g lactose, wet granulate with 10% starch slurry, and boil and dry; mix indomethacin granules with 8g low-substituted hydroxypropyl cellulose and 7g talcum powder Mix evenly, compress into tablets, and wrap the isolation layer; mix Vonorazan, 0.1g of aspartame, 5g of sodium carboxymethylcellulose and 6g of lactose, add water to moisten, wrap around the tablet core, and dry to obtain .
Embodiment 3
[0021] Ketoprofen 25mg, Wonorazan 10mg;
[0022] Ketoprofen
[0023] Preparation method: mix ketoprofen, tartaric acid and 100g lactose in the prescribed amount, wet granulate with 10% starch slurry, boil and dry; mix ketoprofen granules with 38g microcrystalline cellulose and 7g talcum powder, and compress into tablets , wrapped with an isolation layer; mix Vonorazan, 0.1g of aspartame, 5g of carboxymethylcellulose sodium and 25g of lactose, moisten with water, wrap around the tablet core, and dry to obtain.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com