Bungotoxin Antigen Epitope Gene and Its Application in Gene Vaccine and Antigen Preparation
A technology of antigenic sites and vaccines, applied in the field of biomedicine
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0069] (1) Acquisition of SEQ ID NO: 1
[0070] (1) Antigenic sites obtained by bioinformatics analysis
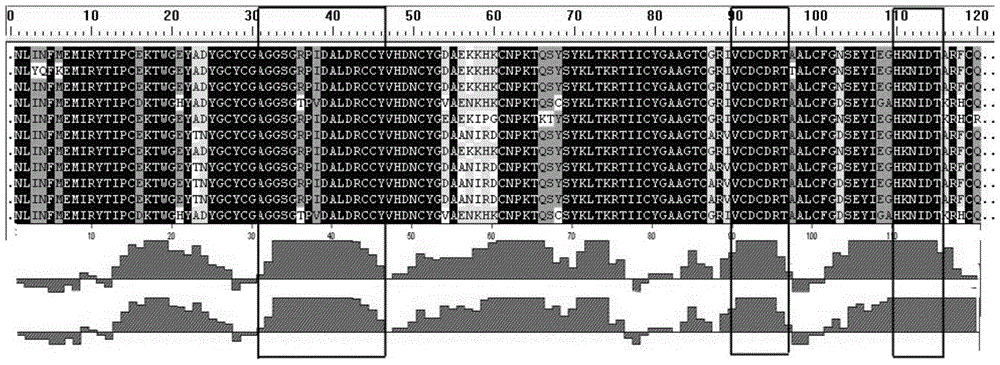
[0071] Two representative sequences extracted from the long chain of α-bungarotoxin (BGT), the A and B chains of β-BGT, and the κ-BGT class: AF056407, AF056407, AJ242012, AJ242011, Y12100, Y12101, Y12265, Y12266 (The above is the ID number in NCBIGenbank).
[0072] Using SignalP software ( http: / / www.cbs.dtu.dk / services / SignalP-2.0 / ) to analyze the signal peptide and propeptide sequences of the above 8 sequences, remove the signal peptide and propeptide sequences and retain the mature peptide sequence for analysis. The secondary structure of the amino acid sequence is predicted by the Jameson-Wolf method combined with the Gamier-Robson and Chou-Fasman method, the hydrophobicity is analyzed by the Kyte-Doolittle method, the flexibility is analyzed by the Karplus-Schulz method, and the site surface possibility is analyzed by the Emini method, Finally, the combination of...
Embodiment 2
[0099] (3) Application of SEQ ID NO: 3 in preparation of antigen by prokaryotic expression
[0100] Design of SEQ ID NO: 3
[0101] On the basis of SEQ ID NO: 1, the Not I (gtg cgg ccg c) and BamH I (gga tcc gc) restriction site sequences on SEQ ID NO: 1 were replaced with BamH I (gta gga tcc) and SalI ( taa gtc gac gta) restriction site sequence and then handed over to a professional company for whole gene synthesis.
[0102] (1) Submit SEQ ID NO: 3 to Beijing Liuhe Huada Gene Technology Co., Ltd. for synthesis. The pUC57 plasmid containing the synthetic gene and the prokaryotic expression plasmid pET-28a(+) were subjected to Sal I / BamH I double enzyme digestion, respectively, and the digestion conditions were carried out according to the instructions of the corresponding enzymes.
[0103] (2) The digested product was confirmed to be about 350 bp in size by agarose electrophoresis, and the target fragment was excised, and then recovered according to the instructions of the ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com