Application of depolymerized holothurian glycosaminolycan in preparation of medicine for preventing and treating thromboembolism diseases
一种糖胺聚糖、血栓栓塞的技术,应用在解聚海参糖胺聚糖在制备防治血栓栓塞疾病药物中的应用领域,能够解决抗凝药物安全隐患、给药方式繁复、安全隐患等问题,达到有利于药物使用、显著抗凝血作用、递增趋势缓和的效果
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
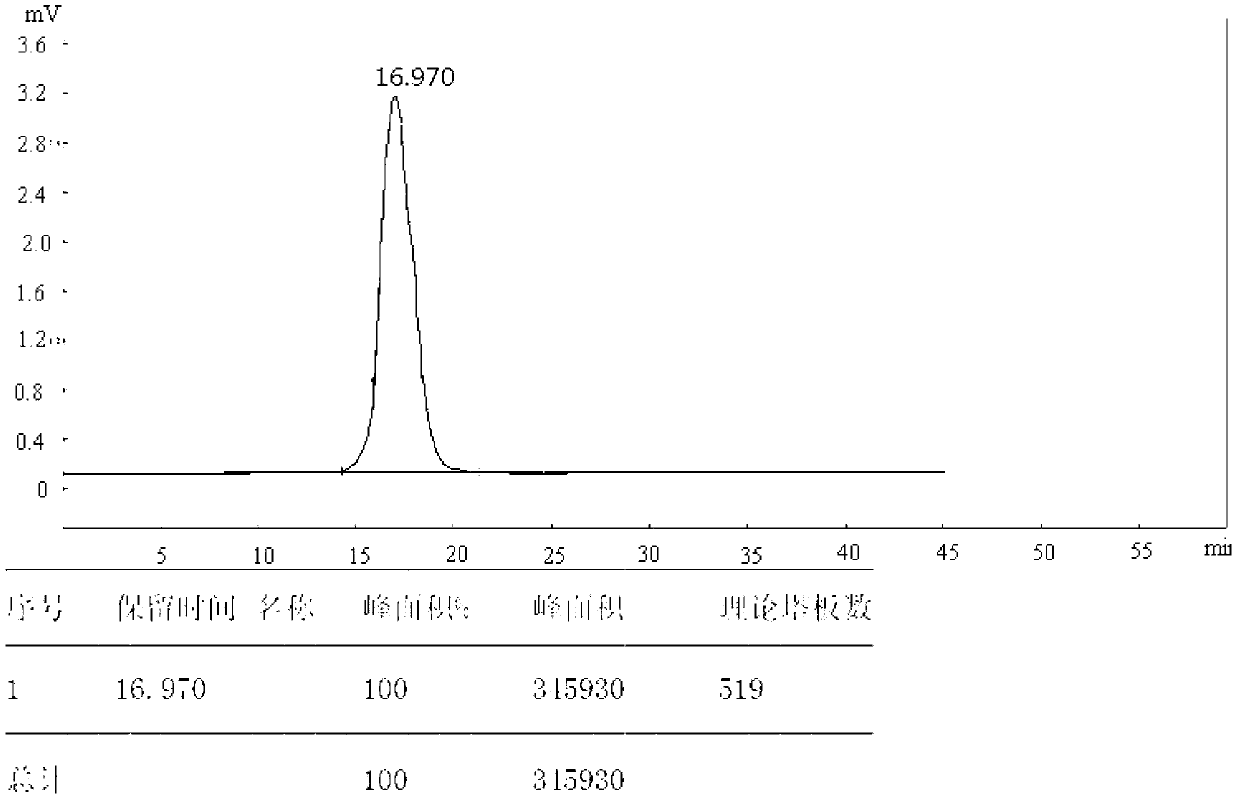
Image
Examples
Embodiment 1
[0029] Extraction of sea cucumber glycosaminoglycans:
[0030] Weigh 5 kg of Yuzu sea cucumber medicinal material and soak in water overnight. Drain the body wall of the sea cucumber, mince it, weigh it and add water to 40Kg, put it in a water bath at 60°C, add 6mol / L sodium hydroxide to adjust the pH to 8.0±0.2, add 100ml of protease Alcalase (Novozymes (Shenyang) Biotechnology Co., Ltd.) stirring, enzymatic hydrolysis for 4 hours, inactivation above 85°C for 10 minutes, cooling to 50°C±2°C, adding 6mol / L sodium hydroxide to adjust the pH to 8.0±0.2, and adding 10g of compound trypsin (Wuxi City Xuemei Enzyme Preparation Technology Co., Ltd. (Xuemei Brand) stirred for 4 hours, boiled for 10 minutes, and cooled. Centrifuge at 4°C, collect the supernatant, add 6mol / L hydrochloric acid to adjust the pH to 2.5±0.2, refrigerate at 4°C for 2 hours, centrifuge, collect the supernatant, add 6mol / L sodium hydroxide to adjust the pH to 7.0±0.2, add 0.8 double ethanol, and let stand o...
Embodiment 2
[0041] Pharmacodynamic experiment of depolymerized sea cucumber glycosaminoglycan
[0042] 2.1 In vitro anticoagulation experiment
[0043] 2.1.1 Test material
[0044] Samples for testing:
[0045] Name: depolymerized sea cucumber glycosaminoglycan (26,000Da~45,000Da), the following abbreviation: DHG;
[0046] Source: Shanghai Korrun Biomedical Co., Ltd.;
[0047] Batch number: 20110308;
[0048] Preparation: Dilute with normal saline for injection to the required concentration after precision drawing.
[0049] experimental animals
[0050] Strain: Rabbit;
[0051] Source: Shanghai Chenxing Experimental Rabbit Co., Ltd.;
[0052] Gender: Male;
[0053] Weight: 1800 grams;
[0054] Animal certificate number: SCXK (Shanghai) 2007-0010.
[0055] 2.1.2 Test equipment
[0056] Platelet aggregation coagulation factor analyzer (model LG-PABER Beijing Shidi Scientific Instrument Company).
[0057] 2.1.3 Experimental method
[0058] On the day of the experiment, add 80 μl...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com