Substituted Heterocycles as Therapeutic agents for treating cancer
a heterocycle and cancer technology, applied in the field of tumor suppressor proteins, can solve the problems of difficult development of molecules capable of inhibiting or disrupting the p53/mdm4 interaction
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
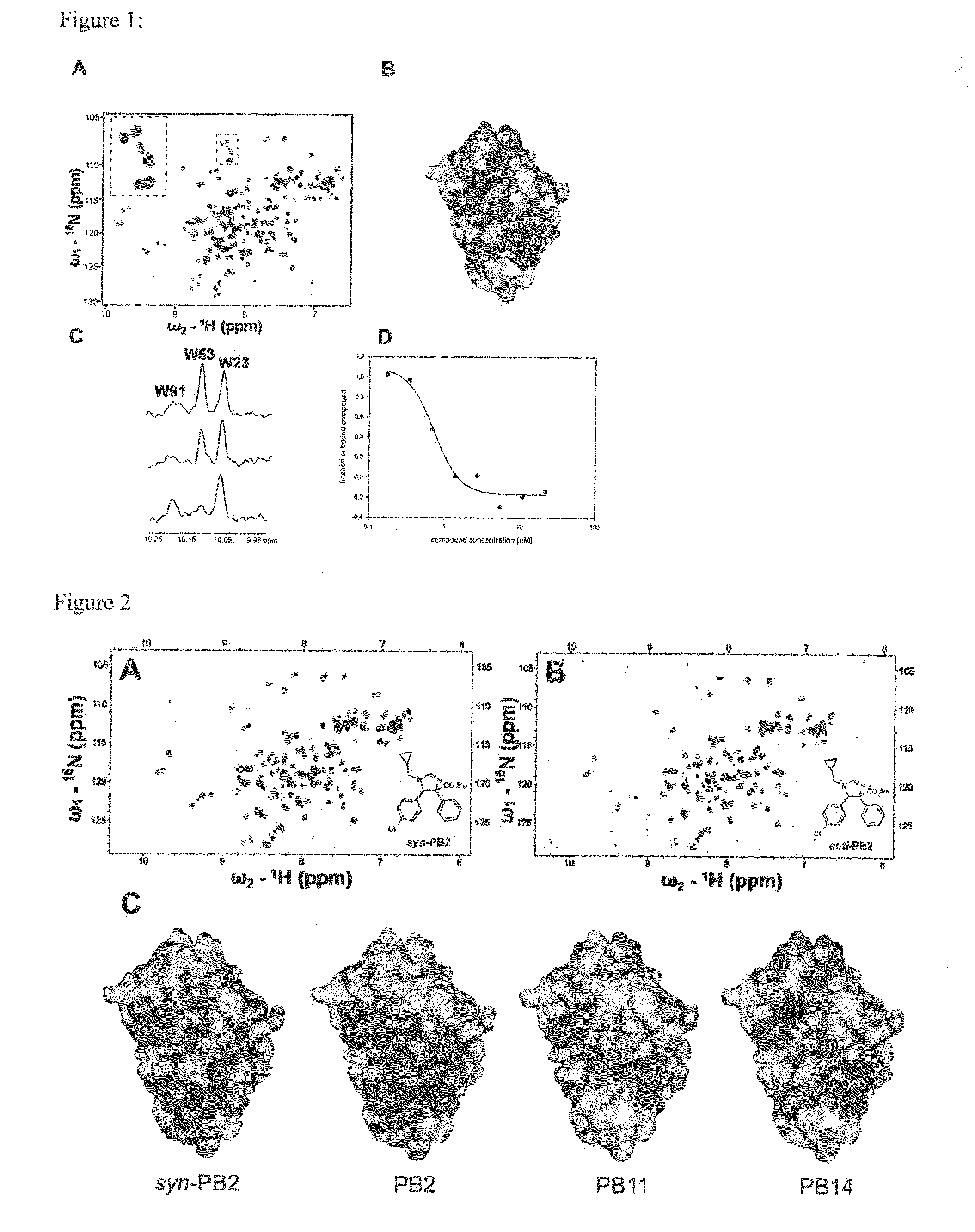
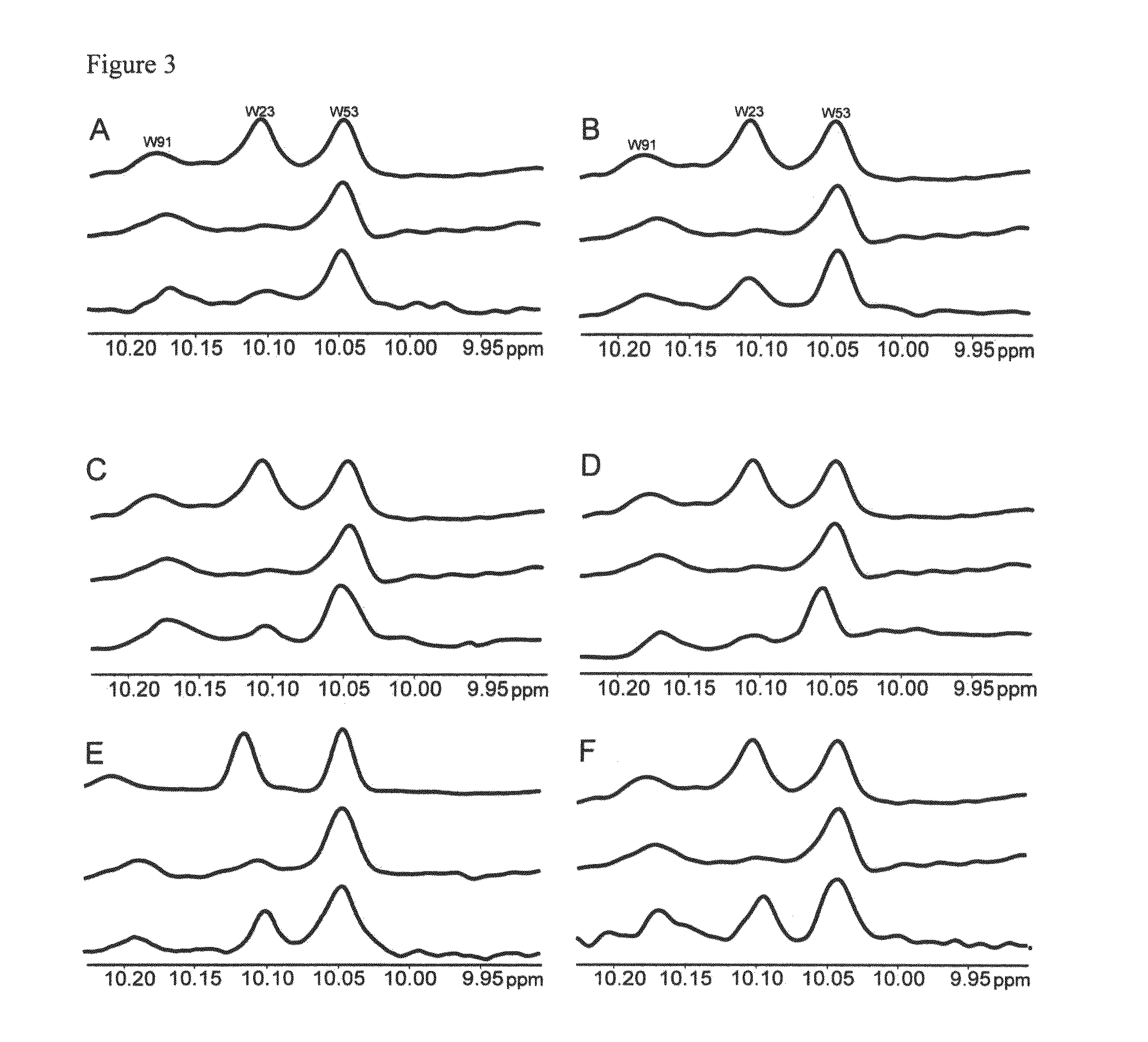
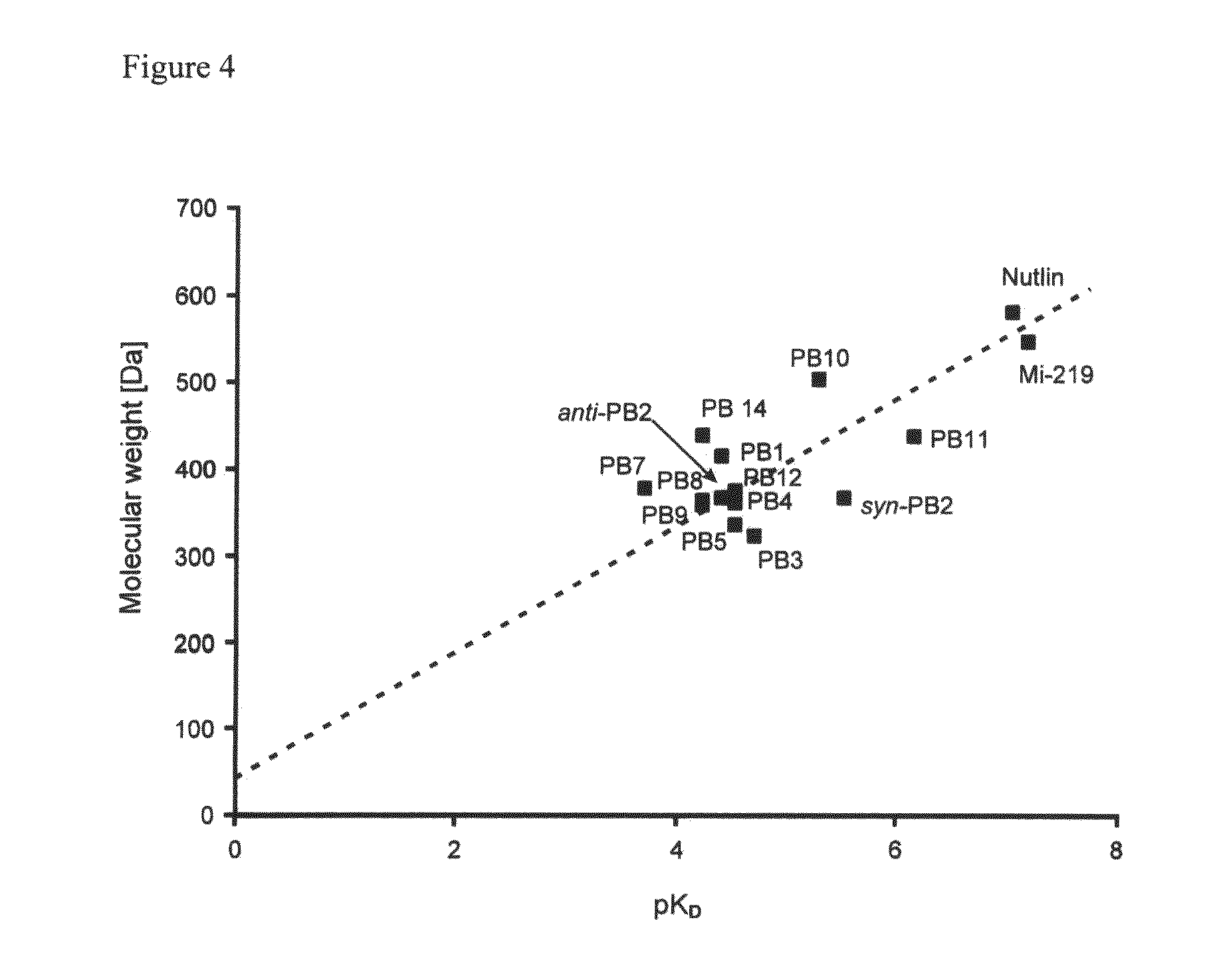
Image
Examples
examples
PB1: 6-Chloro-3-[3-cyclopropylmethyl-5-(3,4-dichlorophenyl)-3H-imidazol-4-yl]-1H-indole
[0065]
[0066]6-Chloro-1H-indole3-carbaldehyde (180 mg, 1 mmol) were dissolved in 2 mL of MeOH, and 85.6 μL (1 mmol) cyclopropylmethyl amine was added dropwise. The reaction mixture was stirred for 4 h at room temperature and 1,2-dichloro-4-[isocyano(toluene-4-sulfonyl)methyl]benzene (340 mg, 1 mmol) and piperazine (86 mg, 1 mmol) were added and stirred over night at room temperature. The solvent was evaporated and the crude product purified by chromatography on silica with a gradient of 3:1 to 2:1 heptane / ethyl acetate to yield 6-chloro-3-[3-cyclopropylmethyl-5-(3,4-dichlorophenyl)-3H-imidazol-4-yl]-1H-indole (PB1) 356 mg (86%); 1H-NMR (CDCl3, 600 MHz): δ 0.17 (d, J=4.80 Hz, 2H), 0.54 (d, J=7.86 Hz, 2H), 0.99-1.03 (m, 1H), 3.59 (d, J=6.90 Hz, 2H), 7.07-7.12 (m, 2H), 7.17-7.21 (m, 2H), 7.25-7.26 (m, 1H), 7.46 (m, 1H), 7.74-7.75 (m, 1H), 7.86 (s, 1H), 9.63 (s, 1H); 13C-NMR (CDCl3, 150 MHz): δ 4.0, 11...
PUM
| Property | Measurement | Unit |
|---|---|---|
| NMR chemical shifts | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
| dual wavelength UV detector | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com