Glycated cd59 peptides, their preparation, and uses thereof
a technology of cd59 and peptides, which is applied in the field of glycated cd59 peptides, their preparation, can solve the problems of achieve the effects of facilitating the unregulated activation of complement and excessive and accelerated deposition of mac, stimulating proliferation of fibroblasts, and increasing the synthesis of extracellular matrix proteins
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
General Peptide Coupling Procedure
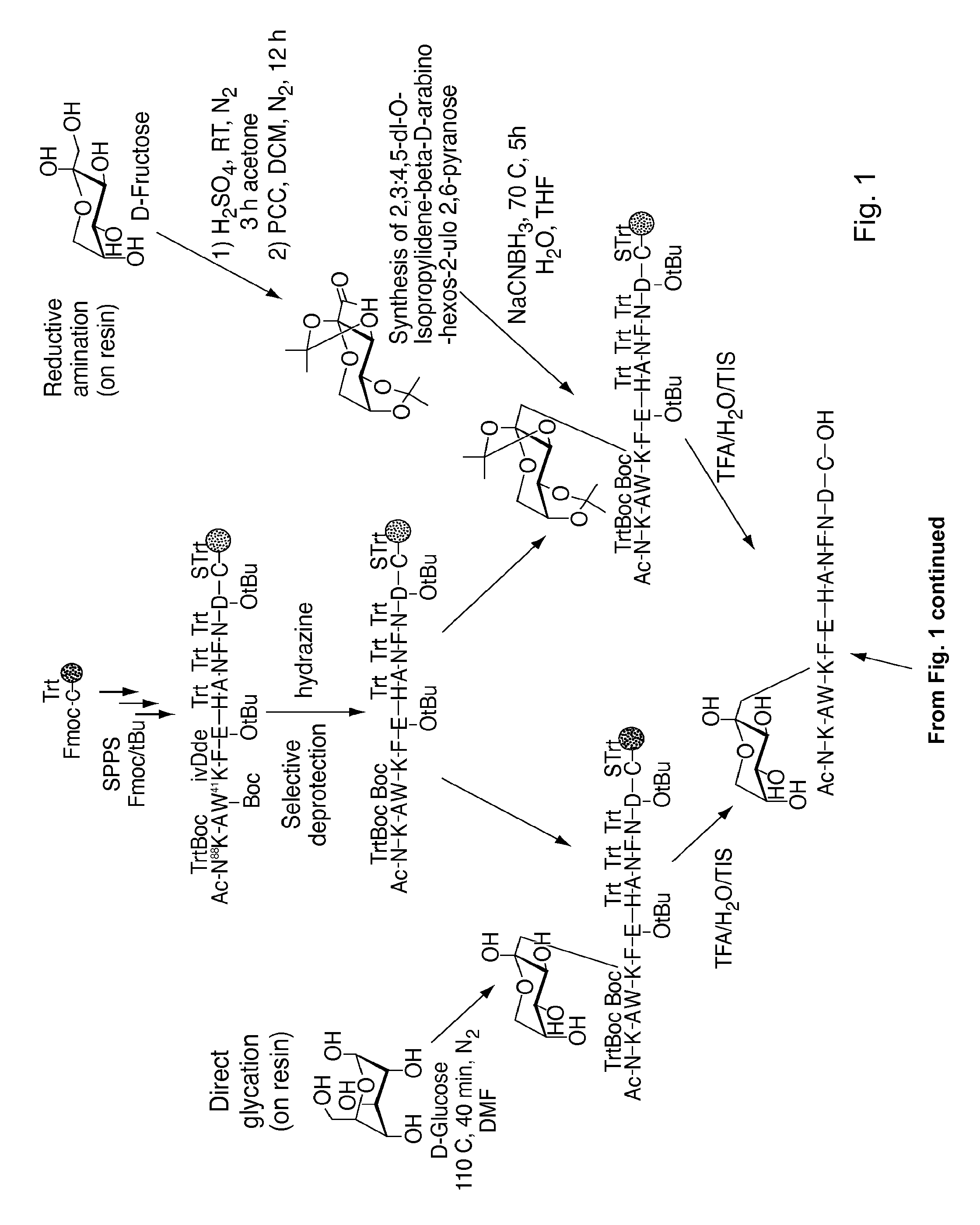
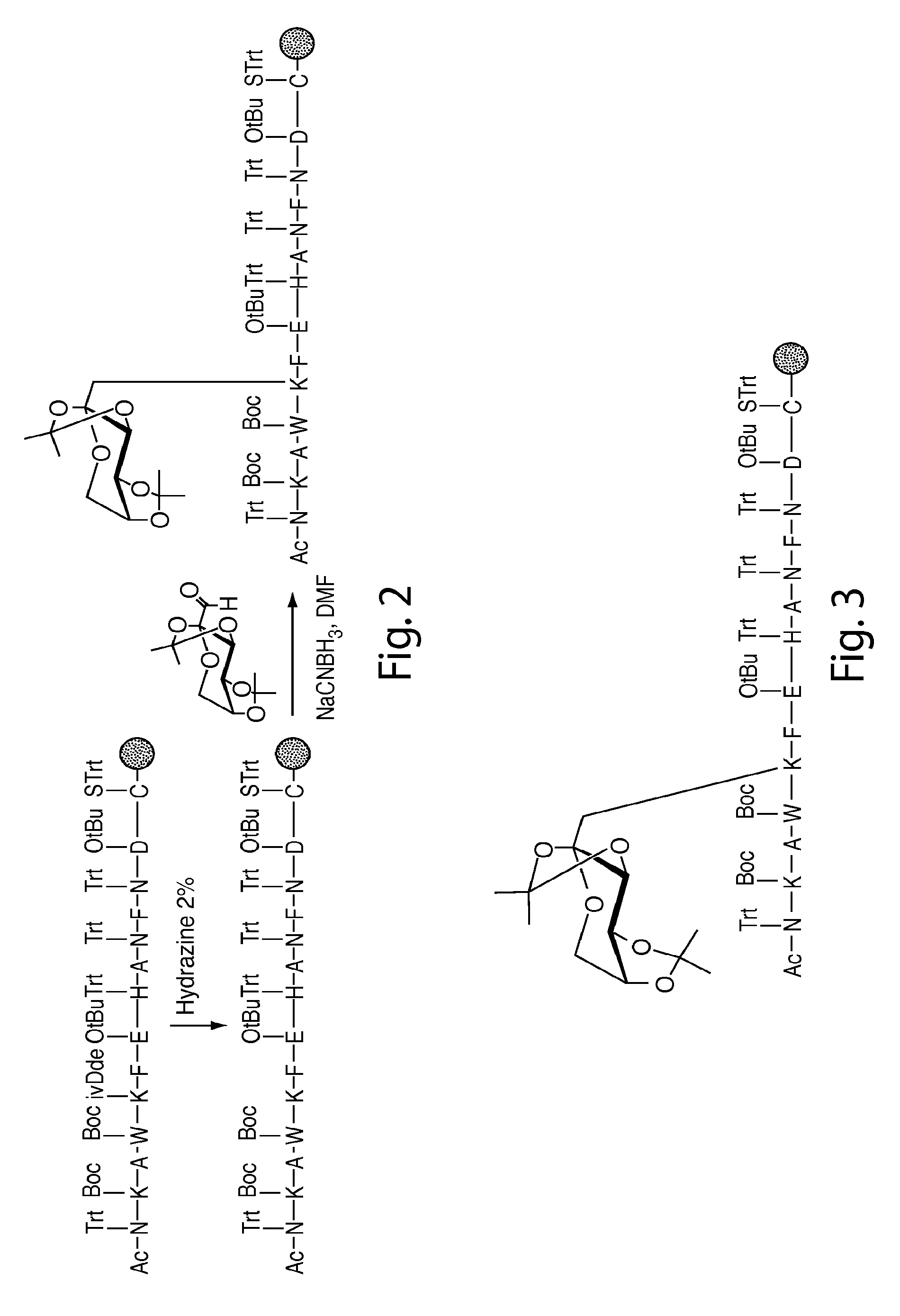
[0205]Nα-Ac[Lys41(Nε-1-deoxyfructosyl)]hCD59(37-50)-OH polypeptide (SEQ ID NO:3) was manually synthesized via stepwise solid-phase polypeptide synthesis from 155 mg of Fmoc-Cys(Trt)-Rink resin starting material (0.41 meq / g, 0.0633 mmol) using PyBOP, HOBt, and DIEA coupling reagents in 5 / 5 / 10-fold molar excess. Successive Nα-Fmoc protected amino acids were added in a 5-fold molar excess and stirred 2-3 hours at ambient temperature in the dark. Side-chain protecting groups of the Nα-Fmoc protected amino acids included: Asn(Trt), Asp(OtBu), Glu(OtBu), His(Nim-Trt), Lys(Nε-Boc) and Trp(Nin-Boc). Synthesis of derivatives of the above polypeptide follow a similar procedure.
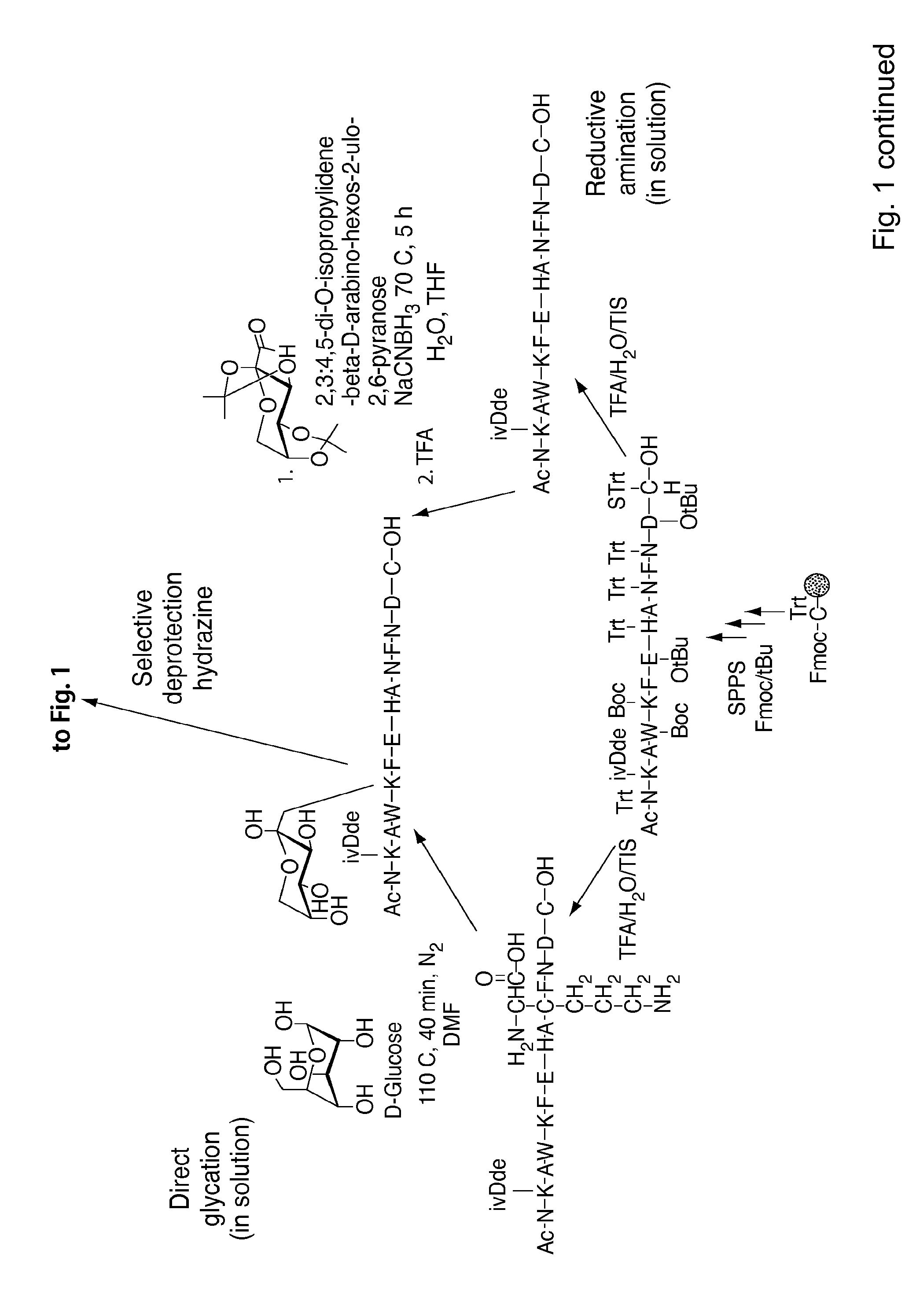
[0206]The Amadori modification: The Amadori modification of lysine proved successful only when fully protected Amadori building blocks of lysine were used. Thus, the fully protected Amadori building block (1), Nα-Fmoc-Lys[Nε-(2,3:4,5-di-O-isopropylidene-1-deoxyfructosyl, Nε-Boc)]-OH, wa...
example 2
Synthesis of fully protected Nα-Fmoc-Lys[Nε-(2,3:4,5-di-O-isopropylidene-1-deoxyfructosyl, Nε-Boc)]-OH (1)
[0209]
Step 1: Synthesis of Nα-Fmoc-Lys[Nε-(2,3:4,5-di-O-isopropylidene-1-deoxyfructosyl)]-OH (2)
[0210]A fresh solution of NaCNBH3 (600 mg, 9.5 mmol) in 5 mL H2O / THF (1:1, v / v) was added to a stirred solution of Nα-Fmoc-Lys-OH (1.4 g, 3.8 mmol) and 2,3:4,5-di-O-isopropylidene-aldehydo-β-D-arabino-hexos-2-ulo-2,6-pyranose (2.45 g, 9.5 mmol) in 15 mL H2O / THF (1:1, v / v) under N2 at 50° C. After 4 h the solvent was removed under reduced pressure and the crude product (RfA for the product=0.8, RfA for the starting material=0.45; UV & ninhydrin positive) was purified (RP-FC) using a linear gradient 30-50% B in A over 20 minutes. The intended product eluted at 37% B in A. The pooled fractions were lyophilized. Pure (2) was obtained as a white solid (500 mg, 22%). LC-ESI-MS m / z=611 ([M+H]+), calcd MW=610 at Rt=3.06 minutes using a linear gradient of 30-80% B in A over 5 min; at Rt=3 min ...
example 3
Synthesis of Nα-Fmoc-Lys[Nε-(1-deoxyfructosyl)]-OH (3)
[0212]
[0213]FmocLys-OH (1 g, 2.71 mmol) and D-glucose (1.22 g, 6.77 mmol, 2.5 eq) were suspended in 40 mL of dry DMF under an atmosphere of N2. The reaction was heated to 110° C. for 10 minutes then poured into an ice bath. After a few minutes the Fmoc-Lys-OH dissolved in DMF as a yellow solution. The reaction had to be stopped before the solution was allowed to rapidly turned brown, which indicates the occurrence of diglycated product. The DMF was evaporated under vacuum and the crude residue was purified by RP-FC. TLC analysis with DCM / MeOH (1:4) eluant was visualized with ninhydrin. Pure (3) was obtained as a white solid (390 mg, 67%). LC-ESI-MS calcd: MW=530. found m / z=531 ([M+H]+), Rt=2.13 min using a linear gradient of 30-50% B in A over min 5 min; analytical RP-HPLC was employed using a linear gradient of 40-80% B in A over 3 min; R, =1.45 min. 1H NMR (CDCl3, 400 MHz,): δ 7.74 (d, 2H, J3,4=J5,6=7.4 Hz, fluorenyl 4-H and 5-...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com