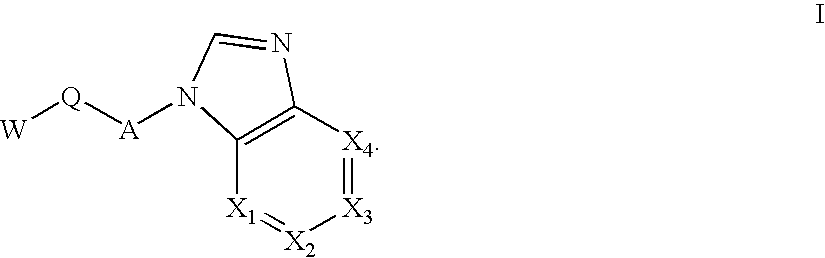
Selective Kinase Inhibitors
a kinase inhibitor and selective technology, applied in the field of selective kinase inhibitors, can solve the problems of difficult design of compounds with acceptable selectivity, inhibiting other members of the family with detrimental long-term consequences, etc., and achieve the effect of suppressing the immune system of a subj
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
6-Chloro-N-[(1R)-1-phenylethyl]pyrazin-2-amine
[0135]
[0136]A solution of R-α-methylbenzylamine (0.57 g, 4.7 mmol) and 2,6-dichloropyrazine (0.6388 g, 4.29 mmol) in dioxane (2.5 mL) was heated at reflux under N2 for 48 hours. The solvent was removed and the product crystallised from toluene-hexane (0.82 g, 82%).
[0137]1H-n.m.r. (CDCl3) δ 1.58 (d, J=6.6 HZ, 3H, CH3), 4.88 (m, 1H, CH), 5.07 (d, 1H, NH), 7.24-7.36 (m, 5H, Ar-H), 7.61 (s, 1H, pyraz-H), 7.79 (s, 1H, pyraz-H).
example 2
N-(tert-butyl)-6-chloropyrazin-2-amine
[0138]
[0139]A mixture of tert-butylamine (14.9 g, 20 mmol), 2,6-dichloropyrazine (6.0 g, 40 mmol), Hünig's base (10 mL) and ethoxyethanol (6 mL) was heated at 130° C. in a sealed tube for 18 hours. The solvent was removed in vacuo and the residue taken up in CHzCl2 (100 mL) and filtered. The filtrate was washed with H2O (2×20 mL), brine (20 mL) and dried (Na2SO4). Chromatography eluting with CH2Cl2 separated the product as a white solid (5.4 g, 72%).
[0140]1H-n.m.r. (CDCl3) δ 1.44 (s, 9H, CH3), 4.68 (br s, 1H, NH), 7.71 (s, 1H, pyraz-H), 7.72 (s, 1H, pyraz-H).
example 3
6-Chloro-N-[(1R)-1-(3-methoxyphenyl)ethyl]pyrazin-2-amine
[0141]
[0142]In a procedure analogous to Example 1, reaction of R-α-methylbenzylamine (1.0 g, 6.6 mmol) and 2,6-dichloropyrazine (0.440 g, 2.95 mmol) furnished the product (517 mg, 67%).
[0143]1H-n.m.r. (CDCl3) δ 1.59 (d, J=6.9 Hz, 3H, CH3), 3.81 (s, 3H, OCH3), 4.87 (m, 1H, CH), 5.47 (br s, 1H, NH), 6.79-7.30 (m, 4H, Ar-H), 7.66 (s, 1H, pyraz-H), 7.79 (s, 1H, pyraz-H).
PUM
| Property | Measurement | Unit |
|---|---|---|
| Fraction | aaaaa | aaaaa |
| Fraction | aaaaa | aaaaa |
| Dimensionless property | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com