Use of Exendins and Agonists Thereof for the Treatment of Gestational Diabetes Mellitus
a technology of gestational diabetes and exendins, which is applied in the direction of peptide/protein ingredients, extracellular fluid disorders, metabolic disorders, etc., can solve the problems of increasing the incidence of hypoglycemia, and achieve the effect of prolonging the effect of blood glucos
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
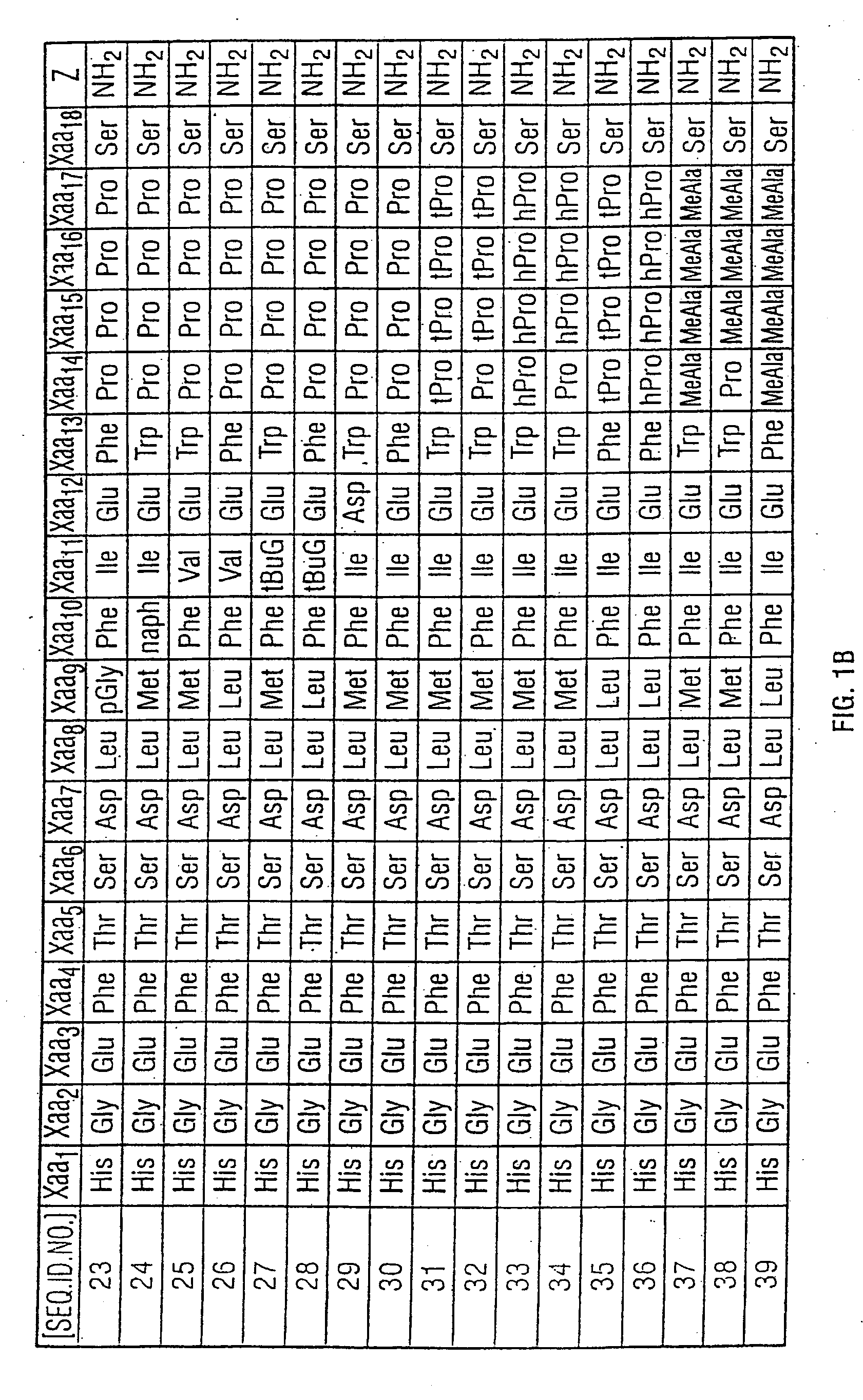
Preparation of Amidated Peptide Having SEQ ID NO. 9
[0175]The above-identified peptide was assembled on 4-(2′-4′-dimethoxyphenyl)-Fmoc aminomethyl phenoxy acetamide norleucine MBHA resin (Novabiochem, 0.55 mmole / g) using Fmoc-protected amino acids (Applied Biosystems, Inc.). In general, single-coupling cycles were used throughout the synthesis and Fast Moc (HBTU activation) chemistry was employed. However, at some positions coupling was less efficient than expected and double couplings were required. In particular, residues Asp9, Thr7 and Phe6 all required double coupling. Deprotection (Fmoc group removal) of the growing peptide chain using piperidine was not always efficient. Double deprotection was required at positions Arg20, Val19 and Leu14. Final deprotection of the completed peptide resin was achieved using a mixture of triethylsilane (0.2 mL), ethanedithiol (0.2 mL), anisole (0.2 mL), water (0.2 mL) and trifluoroacetic acid (15 mL) according to standard methods (Introduction t...
example 2
Preparation of Peptide Having SEQ ID NO. 10
[0178]The above-identified peptide was assembled on 4-(2′-4′-dimethoxyphenyl)-Fmoc aminomethyl phenoxy acetamide norleucine MBHA resin (Novabiochem, 0.55 mmole / g) using Fmoc-protected amino acids (Applied Biosystems, Inc.), cleaved from the resin, deprotected and purified in a similar way to Example 5. Used in analysis were Solvent A (0.1% TFA in water) and Solvent B (0.1% TFA in ACN). Analytical RP-HPLC (gradient 25% to 75% Solvent B in Solvent A over 30 minutes) of the lyophilized peptide gave product peptide having an observed retention time of 21.5 minutes. Electrospray Mass Spectrometry (M): calculated 4168.6. found 4171.2.
EXAMPLE 3
Preparation of Peptide Having SEQ ID NO. 11
[0179]The above-identified peptide was assembled on 4-(2′-4′-dimethoxyphenyl)-Fmoc aminomethyl phenoxy acetamide norleucine MBHA resin (Novabiochem, 0.55 mmole / g) using Fmoc-protected amino acids (Applied Biosystems, Inc.), cleaved from the resin, deprotected and pu...
example 3
Preparation of Peptide Having SEQ ID NO. 12
[0180]The above-identified peptide was assembled on 4-(2′-4′-dimethoxyphenyl)-Fmoc aminomethyl phenoxy acetamide norleucine MBHA resin (Novabiochem, 0.55 mmole / g) using Fmoc-protected amino acids (Applied Biosystems, Inc.), cleaved from the resin, deprotected and purified in a similar way to Example 1. Used in analysis were Solvent A (0.1% TFA in water) and Solvent B (0.1% TFA in ACN). Analytical RP-HPLC (gradient 35% to 65% Solvent B in Solvent A over 30 minutes) of the lyophilized peptide gave product peptide having an observed retention time of 19.7 minutes. Electrospray Mass Spectrometry (M): calculated 4212.6. found 4213.2.
PUM
| Property | Measurement | Unit |
|---|---|---|
| width | aaaaa | aaaaa |
| concentrations | aaaaa | aaaaa |
| concentration | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com