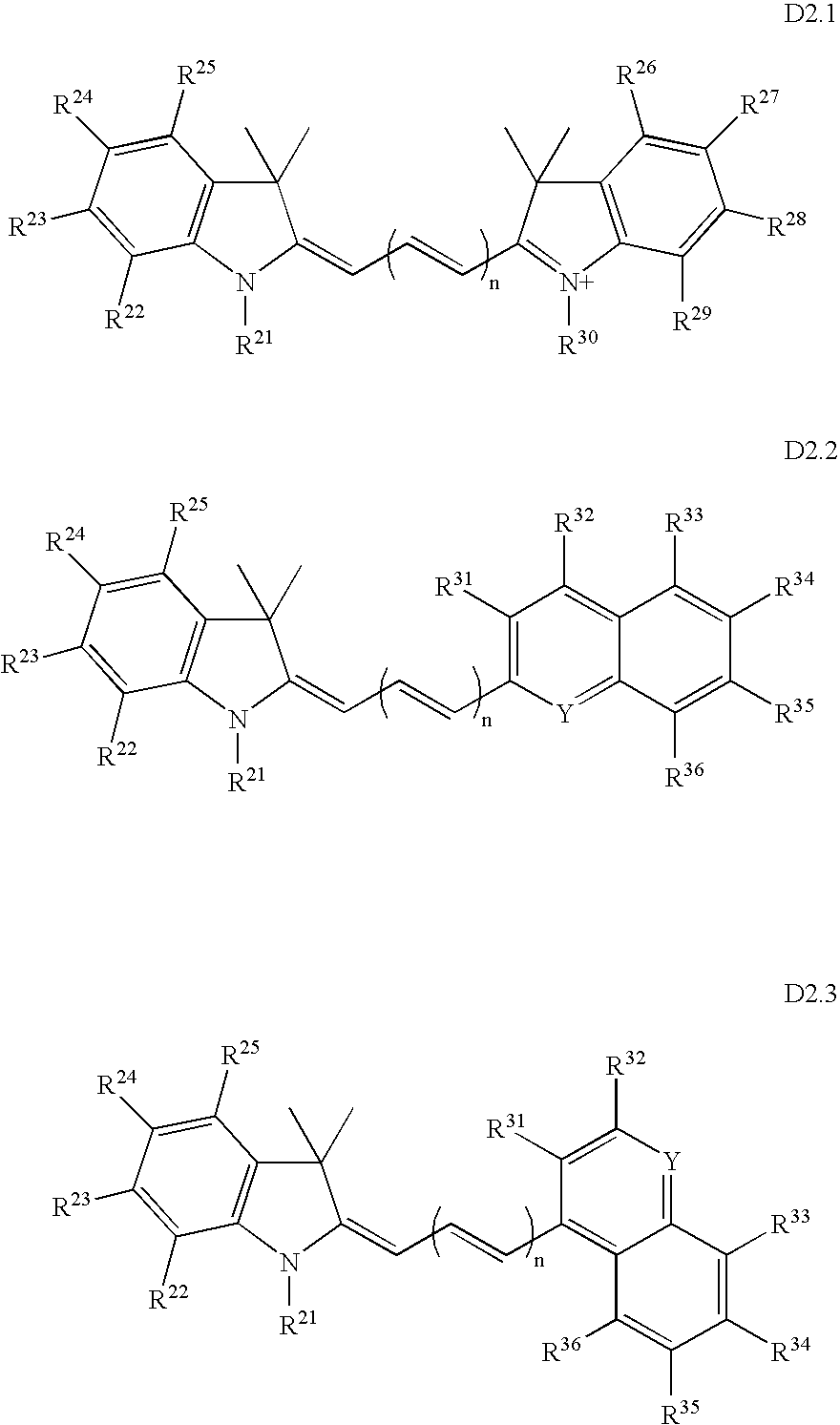
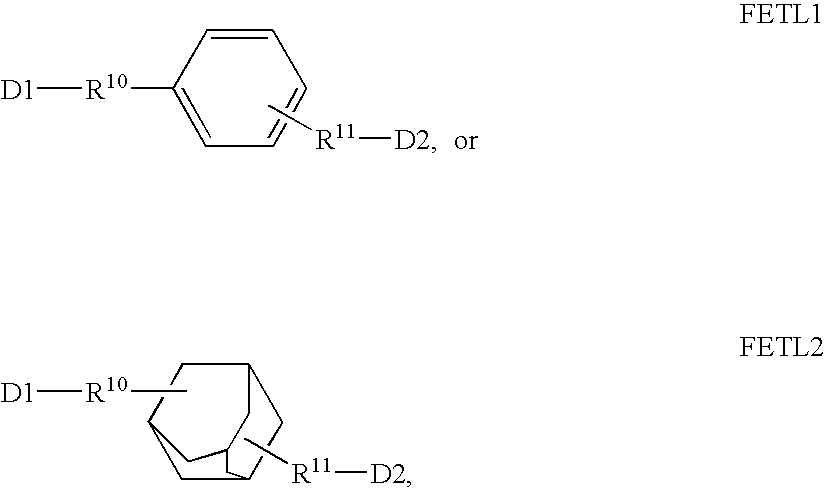Fluorescent dyes, energy transfer couples and methods
a technology of fluorescence and energy transfer, applied in the field of fluorescence dyes and fluorescence energy transfer, can solve the problems of imposing five severe constraints on selection, adding undesired time and expense, and difficult to find a set of dyes whose emission spectra are spectrally resolved
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
[0381] 2,3,4,5-Tetrachloro-6-(2-(4′-carboxyphenyl)-7-chloro-6-hydroxy-3-oxo-3H-xanthen-9-yl)-benzoic acid
[0382] 1A. 2,3,4,5-Tetrachloro-6-(5-chloro-2,4-dihydroxy-benzoyl)-benzoic acid (0.43 g, 1.0 mmol) and 2′,4′-dihydroxy-biphenyl-4-carboxylic acid (3, 0.23 g, 1.0 mmol) in methanesulfonic acid (10 mL) was placed in a pre-heated oil bath (T=130° C.) and stirred over a period of 2 h. The reaction mixture was allowed to cool to room temperature and quenched by pouring into ice cold water (200 mL). The solution was allowed to stand over a period of 2 h and the crude product was extracted with ethylacetate (EtOAc, 2×100 mL). The combined organic portions were washed with water (1×100 mL), and saturated sodium chloride (1×100 mL), dried (Na2SO4), and concentrated to give crude 1 as a dark brown solid. Purification via column chromatography (silica gel, 100 g, 1 / 1 EtOAc:hexanes 0.5% AcOH) gave the title compound (0.26 g, 42%) as an orange solid. Absorbance maximum (λmax 540 nm, 1×PBS), ...
example 2
[0388] 2,3,4,5-Tetrachloro-6-(2,7-bis(4′-carboxyphenyl)-6-hydroxy-3-oxo-3H-xanthen-9-yl)-benzoic acid)
[0389] Tetrachloropthallic anhydride (31 mg, 0.11 mmol) and 2′,4′-dihydroxy-biphenyl-4-carboxylic acid (50 mg, 0.22 mmol) in methanesulfonic acid (2 mL) was placed in an pre-heated oil bath (T=130° C.) and stirred over a period of 0.75 h. The reaction mixture was allowed to cool to room temperature and quenched by pouring into ice cold water (50 mL). The solution was allowed to stand over a period of 2 h and the crude product was extracted with ethylacetate (EtOAc, 3×25 mL). The combined organic portions were washed with water (1×50 mL), and saturated sodium chloride (1×50 mL), dried (Na2SO4), and concentrated to give crude 1 as a dark brown solid. Purification via preparative layer chromatography (silica gel RP-18, 1 mm, 95 / 35 100 mM TEAB: AcCN, X 2) giving 4 as the major product. λmax 550 nm, MeOH; λem 575 nm, MeOH.
example 3
[0390] 2,3,4,5-Tetrachloro-6-(4-(4′-carboxyphenyl )-5-hydroxy-9-oxo-9H-benzo[a]xanthen-12-yl)-benzoic acid
[0391] Tetrachloropthallic anhydride (0.32 g, 1.1 mmol) and 2′,4′-dihydroxy-biphenyl-4-carboxylic acid (0.26 g, 1.1 mmol), 1,3-dihydroxynaphtalene (0.18 g, 1.1 mmol), in methanesulfonic acid (4 mL) was placed in an pre-heated oil bath (T=110° C.) and stirred over a period of 17 h. The reaction mixture was allowed to cool to room temperature and quenched by pouring into ice cold water (100 mL). The solution was allowed to stand over a period of 2 h and the crude product was extracted with ethylacetate (EtOAc, 3×25 mL). The combined organic portions were washed with water (1×50 mL), and saturated sodium chloride (1×50 mL), dried (Na2SO4), and concentrated to give crude 1 as a dark brown solid. Purification via column chromatography (silica gel 50 g, 4 / 1 EtOAc:hexanes, 1% AcOH) giving the title compound as the major product. ABS λmax 540 nm, 1×PBS; EM λem 570 nm, 1×PBS.
PUM
| Property | Measurement | Unit |
|---|---|---|
| emission band half-width | aaaaa | aaaaa |
| temperature | aaaaa | aaaaa |
| temperature | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - Generate Ideas
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com