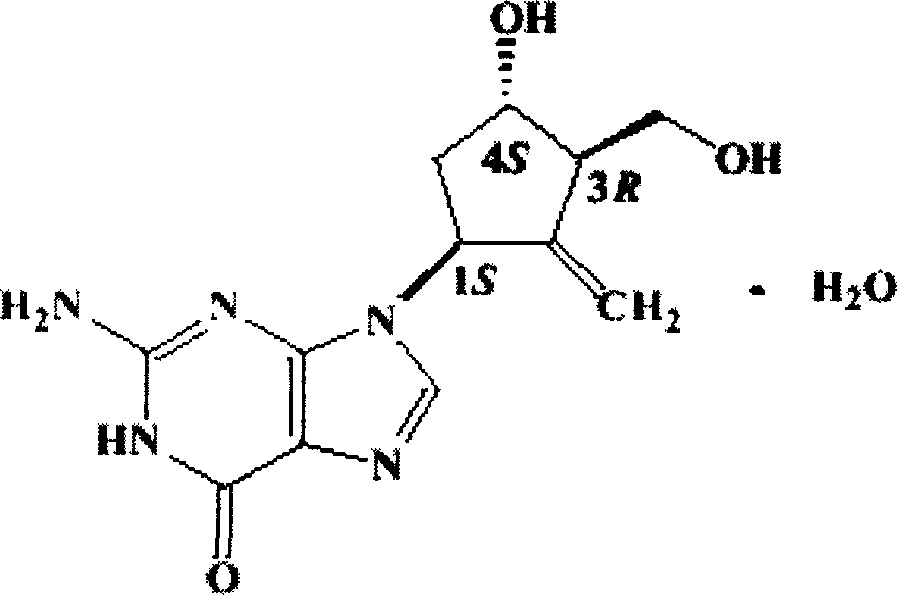
Entecavir soft capsule and its preparation method
A technology of entecavir and soft capsules, which is applied in the field of entecavir soft capsules for the treatment of hepatitis B and its preparation, and can solve the problems that many pharmaceutical factories, government departments, scientific research institutes and other institutions are conducting research work on the drug
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0021] prescription:
[0022] Entecavir 0.5g
[0023] PEG400 190g
[0024] Propylene glycol 9.5g
[0025] A total of 1000 capsules were made
[0026] Preparation:
[0027] First heat about 80% PEG400 to 40-60°C, add entecavir, stir to dissolve, then add the prescribed amount of propylene glycol, stir evenly, then add the remaining amount of PEG400 to obtain a clear solution; adjust the temperature control system of the dropping pill machine, Heat the dropper of the dropping pill machine and keep it at 50-90°C, cool the temperature of the condensing agent and keep it at -5-40°C; adjust the size of the dropper to control the weight of the soft capsule content, and press it to get it.
Embodiment 2
[0029] prescription:
[0030] Entecavir 1g
[0031] PEG400 190g
[0032] Propylene glycol 7g
[0033] Tween-80 2
[0034] A total of 1000 capsules were made
[0035] Preparation:
[0036] First heat about 80% PEG400 to 40-60°C, add entecavir, stir to dissolve, then add the prescribed amount of propylene glycol and Tween-80, stir well, then add the remaining amount of PEG400 to obtain a clear solution; adjust the dropping pill machine The temperature control system makes the dropper temperature of the dropping pill machine heated and kept at 50-90°C, the temperature of the condensing agent is cooled and kept at -5-40°C, and the size of the dripper is adjusted to control the weight of the soft capsule content, pressing , that is.
Embodiment 3
[0038] prescription:
[0039] Entecavir 1g
[0040] Edible vegetable oil 494.5
[0041] Beeswax 2.5g
[0042] Tween-80 2.0g
[0043] A total of 1000 capsules were made
[0044] Preparation:
[0045] Grind Entecavir into ultra-fine powder, add it to edible vegetable oil, stir to make it evenly suspended, add the prescribed amount of beeswax and Tween-80, and continue to stir to obtain a good oily suspension; adjust the temperature control of the dropping pill machine system, to heat and maintain the dropper temperature of the dropping pill machine at 50-90°C, and cool and maintain the temperature of the condensing agent at -5-40°C; adjust the size of the dripper to control the weight of the contents of the soft capsule, and compress, that is have to.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com