Preparation method and preparation device of beta-caryophyllene alcohol derivative
A technology for caryophyllene alcohol and a preparation device is applied in the preparation of organic compounds, the preparation of carboxylic acid esters, organic chemical methods, etc., and can solve the problems of complicated preparation process and poor operability, and achieves improved controllability and improved operability. Preparation efficiency, avoid the effect of conversion
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
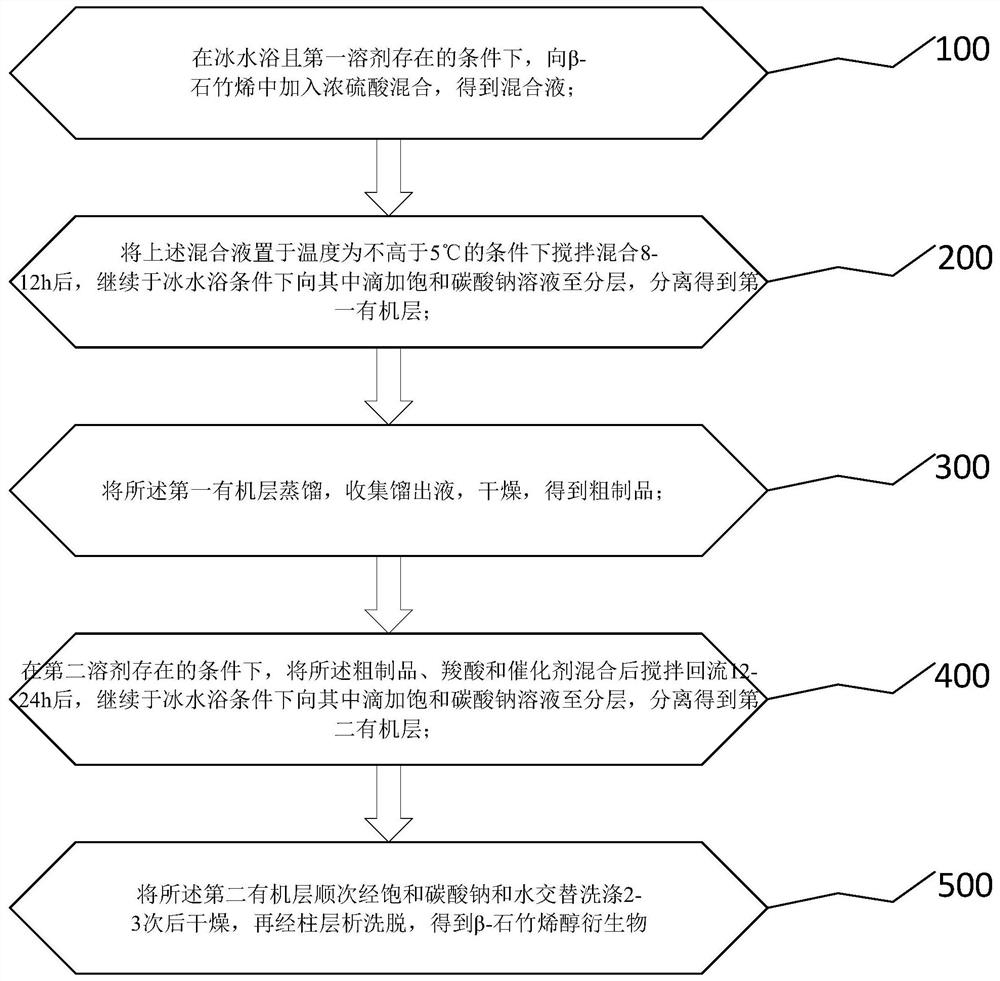
[0061] like figure 1 As shown, the invention provides a kind of preparation method of β-caryophyllenol derivative, comprising:
[0062] 100. Add concentrated sulfuric acid to β-caryophyllene in the presence of solvent A in an ice-water bath and mix to obtain a β-caryophyllene mixed solution;
[0063] 200. After stirring and mixing the β-caryophyllene mixture at a temperature not higher than 5°C for 8-12 hours, continue to add a saturated sodium carbonate solution dropwise to it in an ice-water bath until the layers are separated to obtain the second an organic layer;
[0064] 300. Distill the first organic layer, collect the distillate, and dry to obtain a crude product;
[0065] 400. In the presence of solvent B, mix the crude product, carboxylic acid and catalyst, stir and reflux for 12-24 hours, then continue to add saturated sodium carbonate solution dropwise to it in an ice-water bath until the layers are separated, and the second organic layer;
[0066] 500. The seco...
Embodiment 1
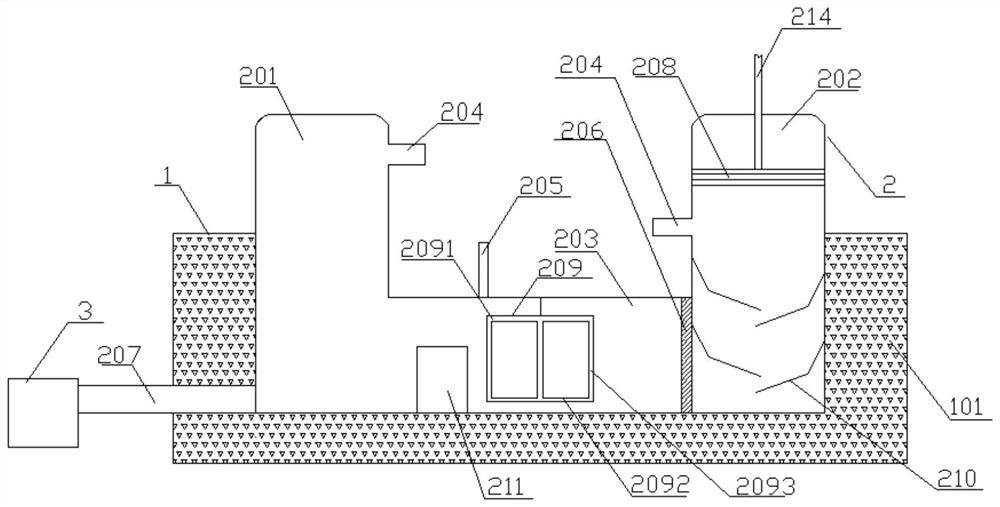
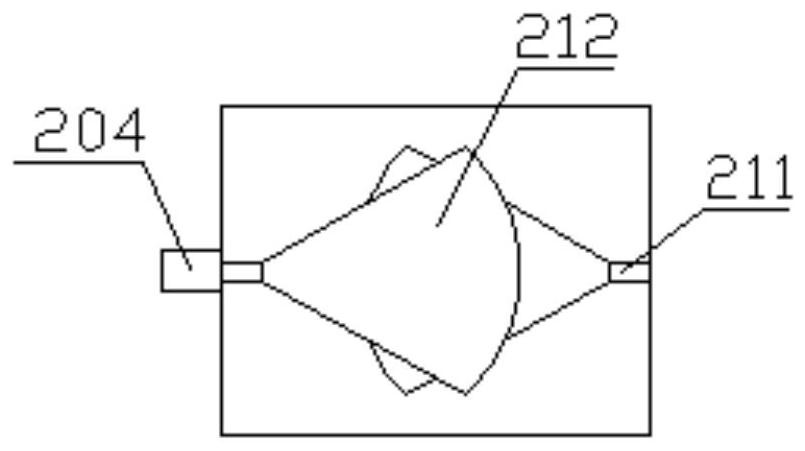
[0109] Fill ice water into the cold and hot water storage tank, add 5g (0.02447mol) β-caryophyllene and 10mL ether to the first storage cylinder, start the stirring paddle to rotate and stir; add 2g of 98% concentrated sulfuric acid to the second storage In the cylinder, slowly push the sealing plate through the air pump until the sulfuric acid enters the connecting pipe; continue to stir for 10h under this condition, then add saturated sodium carbonate dropwise through the titration port to separate layers, open the discharge pipe, collect the first organic layer, distill and dry , get the crude product;
[0110] Mix the above-mentioned crude product, 0.02mol acetic acid and 180mL petroleum ether into the first cylinder for stirring, mix 5mL of 4-dimethylaminopyridine and 20mL petroleum ether into the second cylinder, push it into the connecting pipe for further mixing, Feed through the discharge pipe into the stirring reflux unit and stir and reflux for 18 hours, then import...
Embodiment 2
[0112] The operation was carried out as in Example 1, except that benzoic acid was used instead of acetic acid, and the β-caryophyllenol derivative A2 was collected (5.93 g of product A1, yield 74.79%).
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com