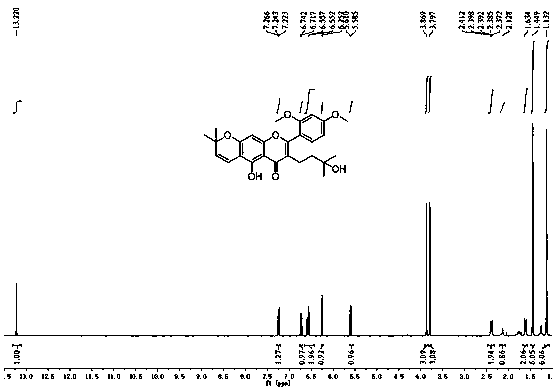
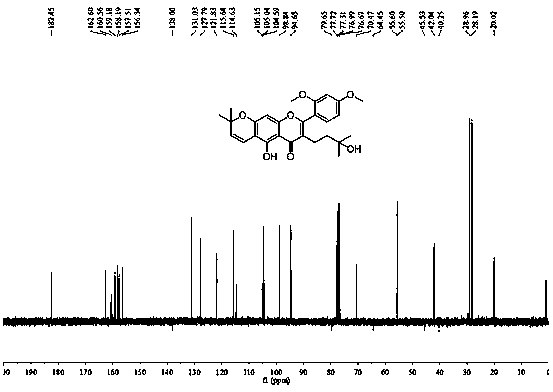
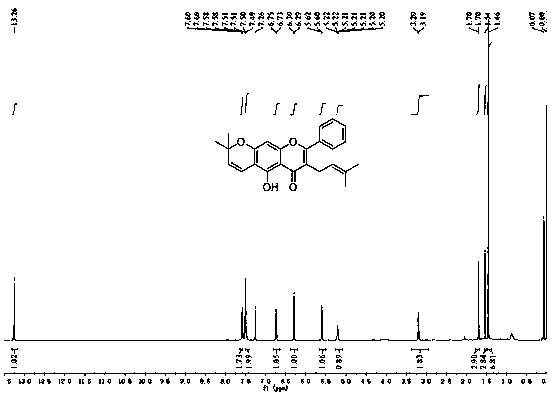
A kind of application and preparation method of morusignin L and its derivatives
A technology of derivatives and reactions, applied in the field of application and preparation of MorusigninL and its derivatives, can solve the problems of limited sources of extraction and separation, low content of natural products, and limitations of druggability research, and achieve cheap synthesis of raw materials and simple and easy operation , the effect of good air stability
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0087] Pharmacological Example 1: Morusignin L and its derivatives 5a, 5b, 5c, 5d, 5e, 6a, 6c-1, 6c-2, 6e-1, 6e-2, 6e-3, 7c or 7e on A549 cells Cytotoxicity
[0088] A549 (human non-small cell lung cancer) was cultured in DMEM medium containing 10% fetal bovine serum, 100U / mL penicillin and 100U / mL streptomycin. Cells were added to 96 wells at a concentration of 4000 cells per well at 37°C with 5% CO 2 Incubate for 24 hours in a humidified incubator.
[0089]Cell viability was determined by the modified MTT method. After the cells were incubated for 24 hours, the newly formulated compound Morusignin L and its derivatives 5a, 5b, 5c, 5d, 5e, 6a, 6c-1, 6c-2, 6e-1, 6e-2, 6e- The dimethyl sulfoxide solution of 3, 7c or 7e was added to each well with a concentration gradient, so that the final concentration of the compound in the well was 6.25 μmol / L, 12.5 μmol / L, 25 μmol / L, 50 μmol / L and 100 μmol / L, respectively. μmol / L. After 48 hours, 10 μL of MTT (5 mg / mL) in phosphate buf...
Embodiment 2
[0091] Pharmacological Example 2: The effect of Morusignin L and its derivatives 5a, 5b, 5c, 5d, 5e, 6a, 6c-1, 6c-2, 6e-1, 6e-2, 6e-3, 7c or 7e on K562 cells Cytotoxicity
[0092] K562 (human chronic myeloid leukemia cells) were cultured in RPMI-1640 medium containing 10% fetal bovine serum, 100 U / mL penicillin and 100 U / mL streptomycin. Cells were added to 96 wells at a concentration of 5000 cells per well at 37°C with 5% CO 2 Incubate for 24 hours in a humidified incubator.
[0093] Cell viability was determined by the modified MTT method. The specific method is as in Pharmacological Example 1. IC of Morusignin L on K562 tumor cells 50 was 10.24 μmol / L; the IC of compound 5a on K562 tumor cells 50 was 28.54 μmol / L; the IC of compound 5b on K562 tumor cells 50 was 15.40 μmol / L; the IC of compound 5c on K562 tumor cells 50 It was 17.31 μmol / L; the IC of compound 5d on K562 tumor cells 50 was 25.15 μmol / L; the IC of compound 5e on K562 tumor cells 50 It was 28.12 μmol / ...
Embodiment 3
[0095] Pharmacological Example 3: Morusignin L and its derivatives 5a, 5b, 5c, 5d, 5e, 6a, 6c-1, 6c-2, 6e-1, 6e-2, 6e-3, 7c or 7e against PC-3 cytotoxicity of cells
[0096] PC-3 (human prostate cancer) cells were cultured in RPMI-1640 medium containing 10% fetal bovine serum, 100U / mL penicillin and 100U / mL streptomycin. Cells were added to 96 wells at a concentration of 5000 cells per well, at 37 o C with 5% CO 2 Incubate for 24 hours in a humidified incubator.
[0097] Cell viability was determined by the modified MTT method. The specific method is as in Pharmacological Example 1. IC of Morusignin L on PC-3 tumor cells 50 9.14 μmol / L; the IC of Morusignin L on PC-3 tumor cells 50 It was 15.71 μmol / L; the IC of compound 5a on PC-3 tumor cells 50 was 47.54 μmol / L; the IC of compound 5b on PC-3 tumor cells 50 It was 37.12 μmol / L; the IC of compound 5c on PC-3 tumor cells 50 It was 25.32 μmol / L; the IC of compound 5d on PC-3 tumor cells 50 is 25.75 μmol / L; the IC of co...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap