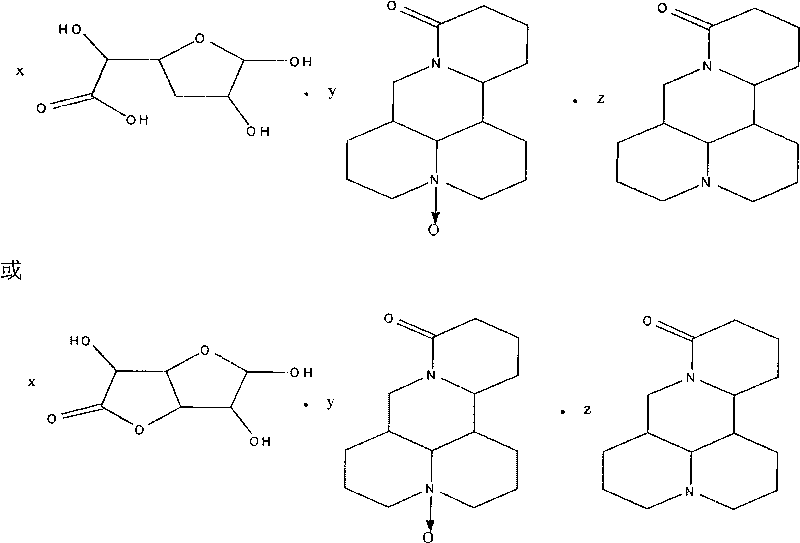
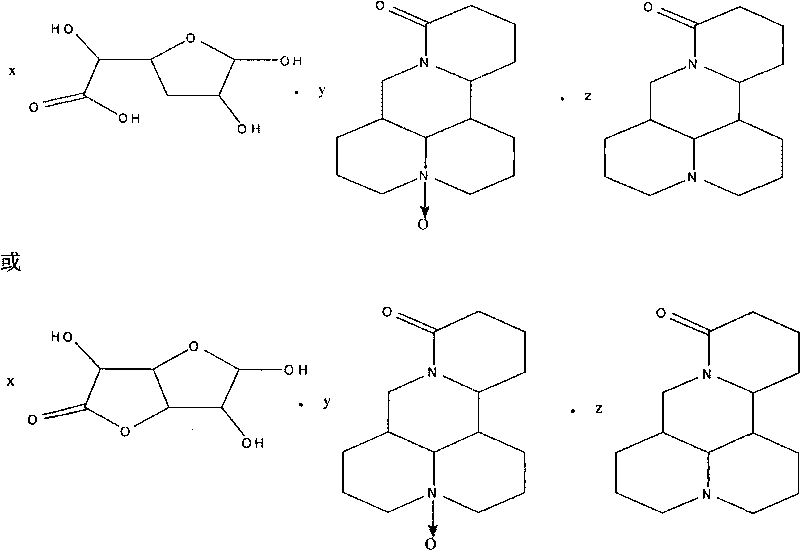
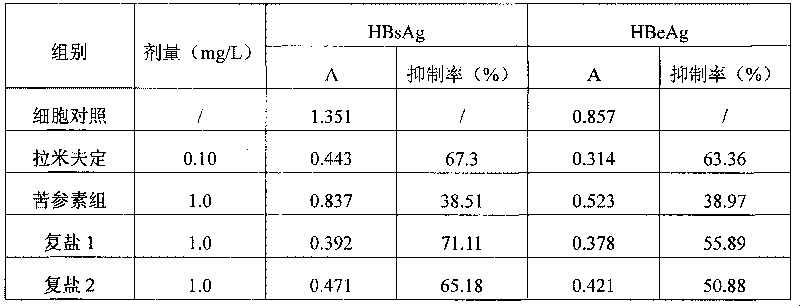
Complex salt consisting of glucuronic acid or glucurolactone and kushenin or matrine and application thereof
A technology of glucuronic acid and glucuronolactone, which is applied in the field of diseases and can solve problems such as reducing
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0049] Preparation method of glucuronolactone and matrine double salt:
[0050] Add about 5 grams of matrine and glucuronolactone in an equimolar ratio into a 100ml reaction bottle, add 50ml of methanol, heat and stir, reflux in a water bath, add an appropriate amount of water dropwise, reflux for 2h, then concentrate the solution, cool, and refrigerate for 12-24h , precipitated white crystals, filtered, washed, and dried in vacuo to obtain 4.31 g of white solids with a yield of 86.2%.
Embodiment 2
[0052] The preparation method of glucuronolactone and matrine double salt:
[0053] Add approximately 5 grams of matrine and glucuronolactone in an equimolar ratio into a 100ml reaction bottle, add 50ml of acetone, heat and stir, reflux in a water bath, add an appropriate amount of water dropwise, reflux for 2 hours, then concentrate the solution, cool, and refrigerate for 12-24 hours , precipitated white crystals, filtered, washed, and dried in vacuo to obtain 4.16 g of white solids with a yield of 83.2%.
Embodiment 3
[0055] The preparation method of glucuronic acid and matrine double salt:
[0056] Put approximately 5 grams of matrine and glucuronic acid in an equimolar ratio into a 100ml reaction bottle, add 50ml of ethanol, heat and stir, reflux in a water bath, add an appropriate amount of water dropwise, reflux for 2 hours, then concentrate the solution, cool, and refrigerate for 12-24 hours. White crystals were precipitated, filtered, washed, and dried in vacuo to obtain 4.45 g of white solids with a yield of 89.0%.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com