Method for polyclonal stimulation of t cells by mobile nanomatrices
a nano-matrice and polyclonal technology, applied in general culture methods, blood/immune system cells, biocide, etc., to save the viability of cells and improve the in-vitro stimulation of t-cells
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
embodiments
[0088]In one embodiment of the present invention a first nanomatrix of 1 to 500 nm, preferentially 10 to 200 nm in size consists of a mobile matrix of a polymer of dextran and has attached thereto one agent, e.g. anti CD3 mAb. A second nanomatrix of 1 to 500 nm, preferentially 10 to 200 nm in size consists of a mobile matrix of a polymer of dextran and has attached thereto another agent, e.g. anti CD28mAb. In this case the nanomatrix of the present invention is a nanomatrix wherein at least one first agent and one second agent are attached to separate mobile matrices.
[0089]A mixture of these nanomatrices is contacted with T cells, thereby activating and inducing the T cells to proliferate.
[0090]Fine-tuning of nanomatrices for the stimulation of the T cells is easily performed due to the high ratio of nanomatrices to cells (normally larger than 500:1).
[0091]In another embodiment of the present invention a nanomatrix of 1 to 500 nm, preferentially 10 to 200 nm in size consists of a mo...
example 1
Preparation of Nanomatrices
[0108]Magnetic nanomatrices were produced by a modification of the procedure of Molday and MacKenzie. Ten grams of Dextran T40 (Pharmacia Uppsala, Sweden), 1.5 g FeCl3.6H2O and 0.64 g FeCl2.4H2O are dissolved in 20 ml H2O, and heated to 40° C. While stirring, 10 ml 4N NaOH are added slowly and the solution is heated to 70° C. for 5 min. The particle suspension is neutralized with acetic acid. To remove aggregates the suspension is centrifuged for 10 min at 2,000 g and filtrated through a 0.22 μm pore-size filter (Millex GV, Millipore, Molsheim, France). Unbound Dextran is removed by washing in a high-gradient magnetic field (HGMF). HGMF washing of magnetic nanomatrices is performed in steelwool columns made as described below and placed in a magnetic field of approx. 0.6 Tesla (MACS permanent magnet, Miltenyi Biotec GmbH, Bergisch Gladbach, Germany). Ten milliliters of nanomatrix suspension are applied to a 15×40 mm column of 2 g steelwool. The loaded colu...
example 2
Expansion of T Cells Using Nanomatrices at Various CD3 / CD28 Concentrations and Ratios Versus CD3 / CD28 MACSiBeads
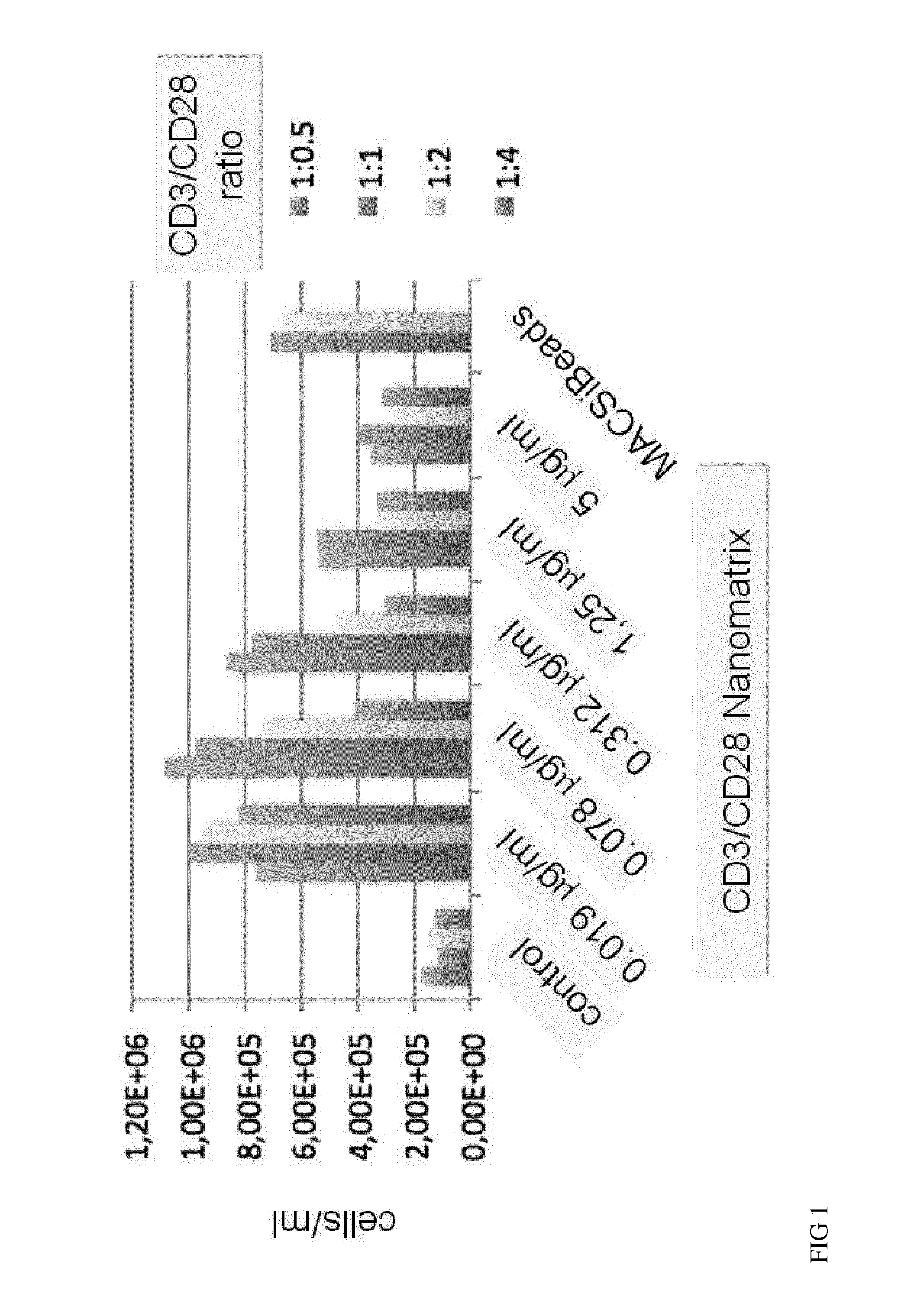
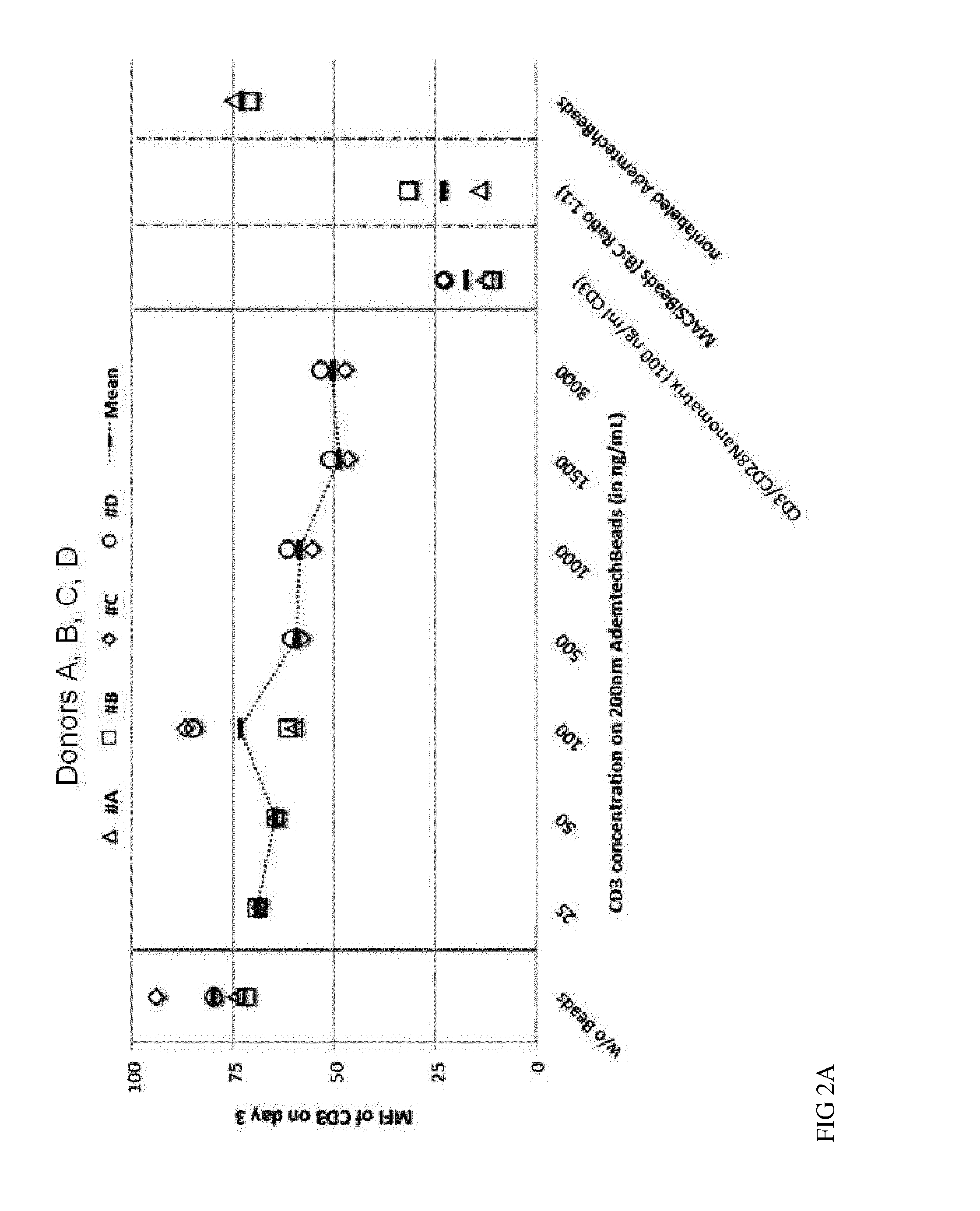
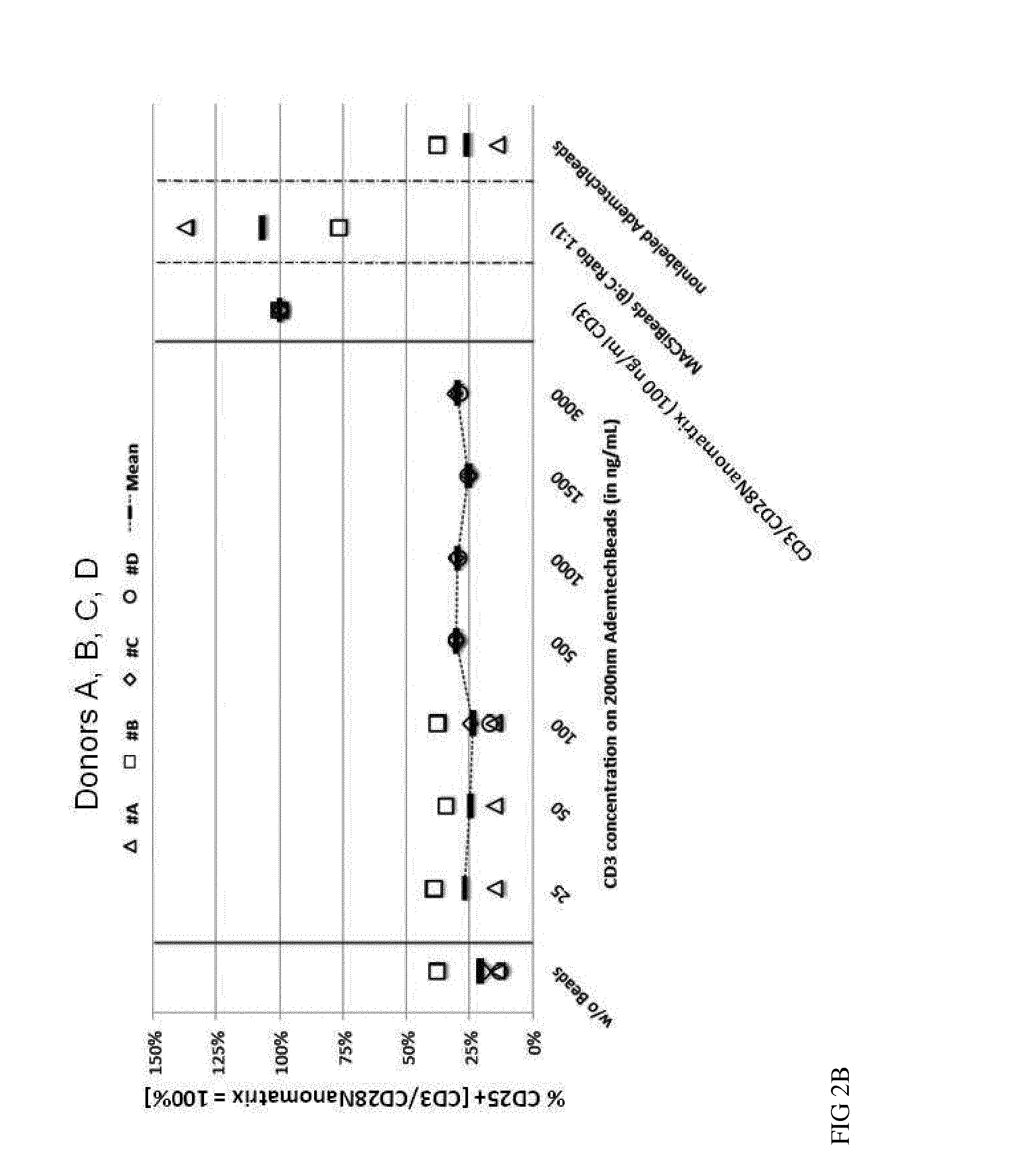
[0110]The current state-of-the-art reagents for activation of highly purified T cells comprise activating antibodies against CD3 / CD28 immobilized either on the surfaces of a cell culture dish or large cell-sized (4-5 μm) particles. Both techniques are error prone and technically difficult to realize and standardize, especially under GMP-compatible production conditions. In contrast nanomatrices can be easily prepared and conveniently be used for cell culture under GMP-conditions. Therefore we compared the T cell activation potential by analysing the expansion potential of the CD3 / CD28 coated nanomatrices at various concentrations and CD3 / CD28 ratios with commercially available cell stimulation beads (MACSiBeads, ø 4.5 μm, Miltenyi Biotec GmbH). As can be seen in FIG. 1 the nanomatrices expand T cells efficiently even at very low CD3 concentrations (20-100 ng / ml) which are ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| size | aaaaa | aaaaa |
| size | aaaaa | aaaaa |
| particle sizes | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com