Rotary cassette system for dry powder inhaler
a technology of dry powder and cassette system, which is applied in the field of inhalation devices, can solve the problems of large clusters making it to the deep lungs then decreasing, affecting the absorption rate of packaged drugs, etc., and achieves the effect of compact assembly and simple structur
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
[0027]In the following description, reference is made to the accompanying drawings, which form a part hereof, and in which is shown, by way of illustration, various embodiments of the present disclosure. It is understood that other embodiments may be utilized and changes may be made without departing from the scope of the present disclosure.
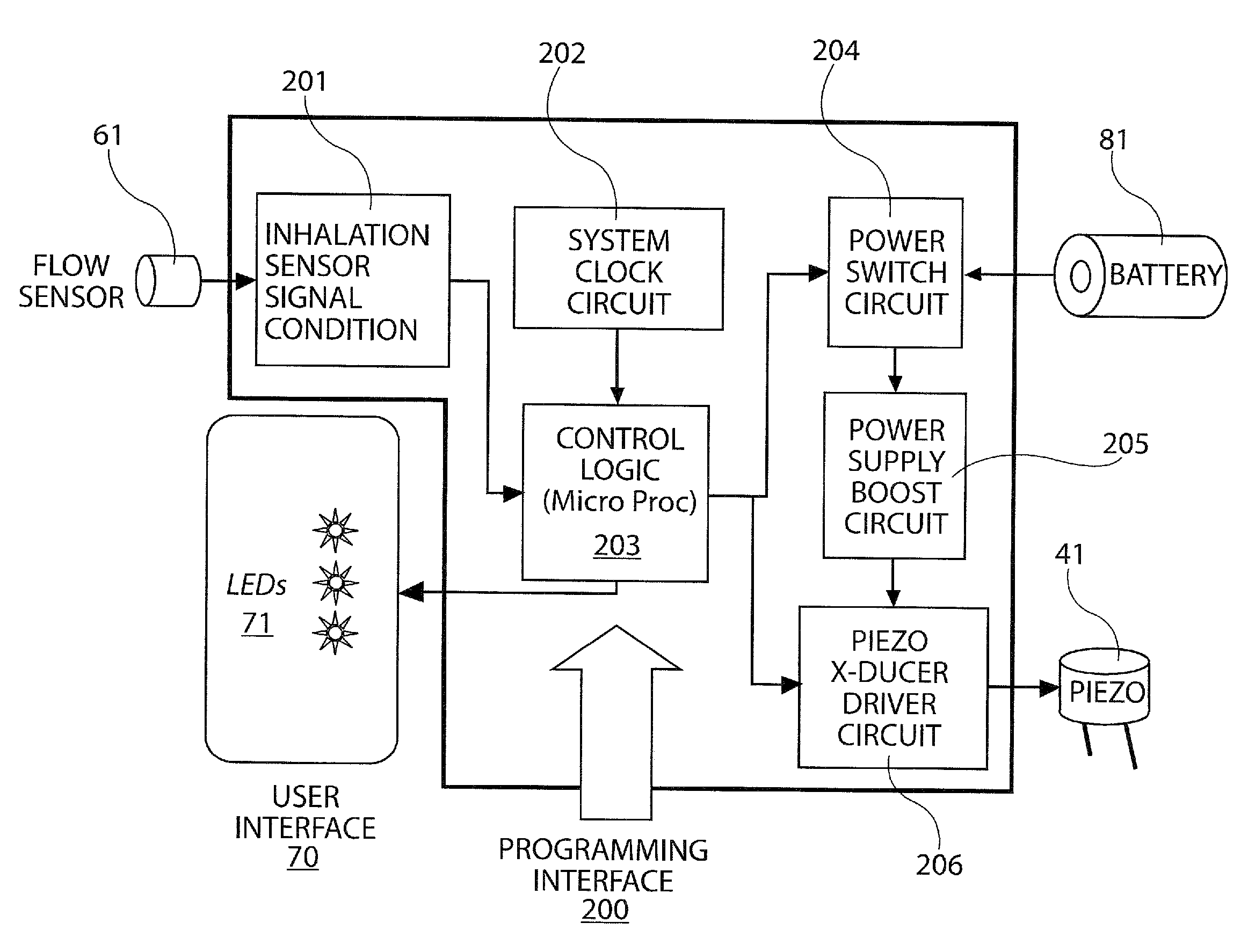
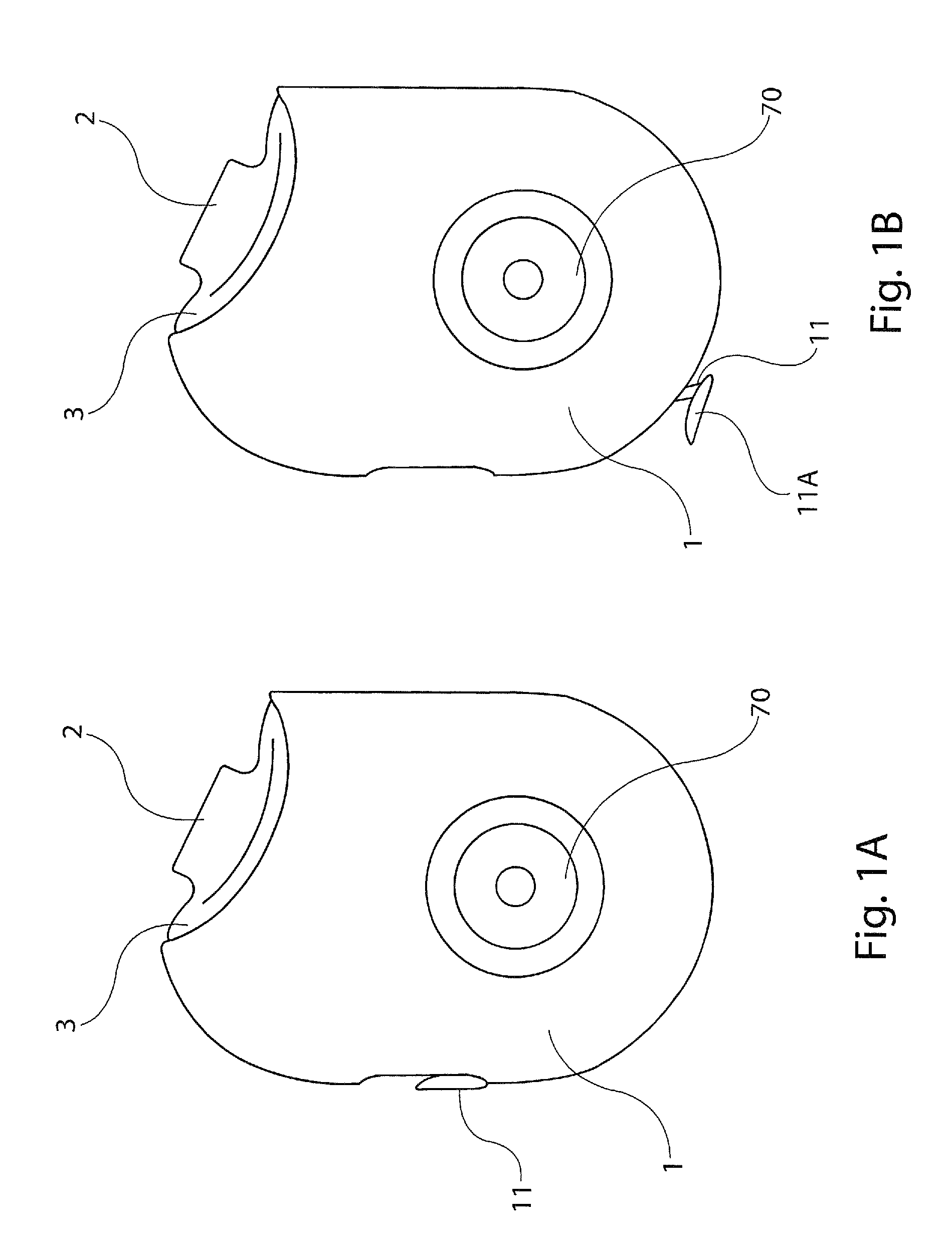
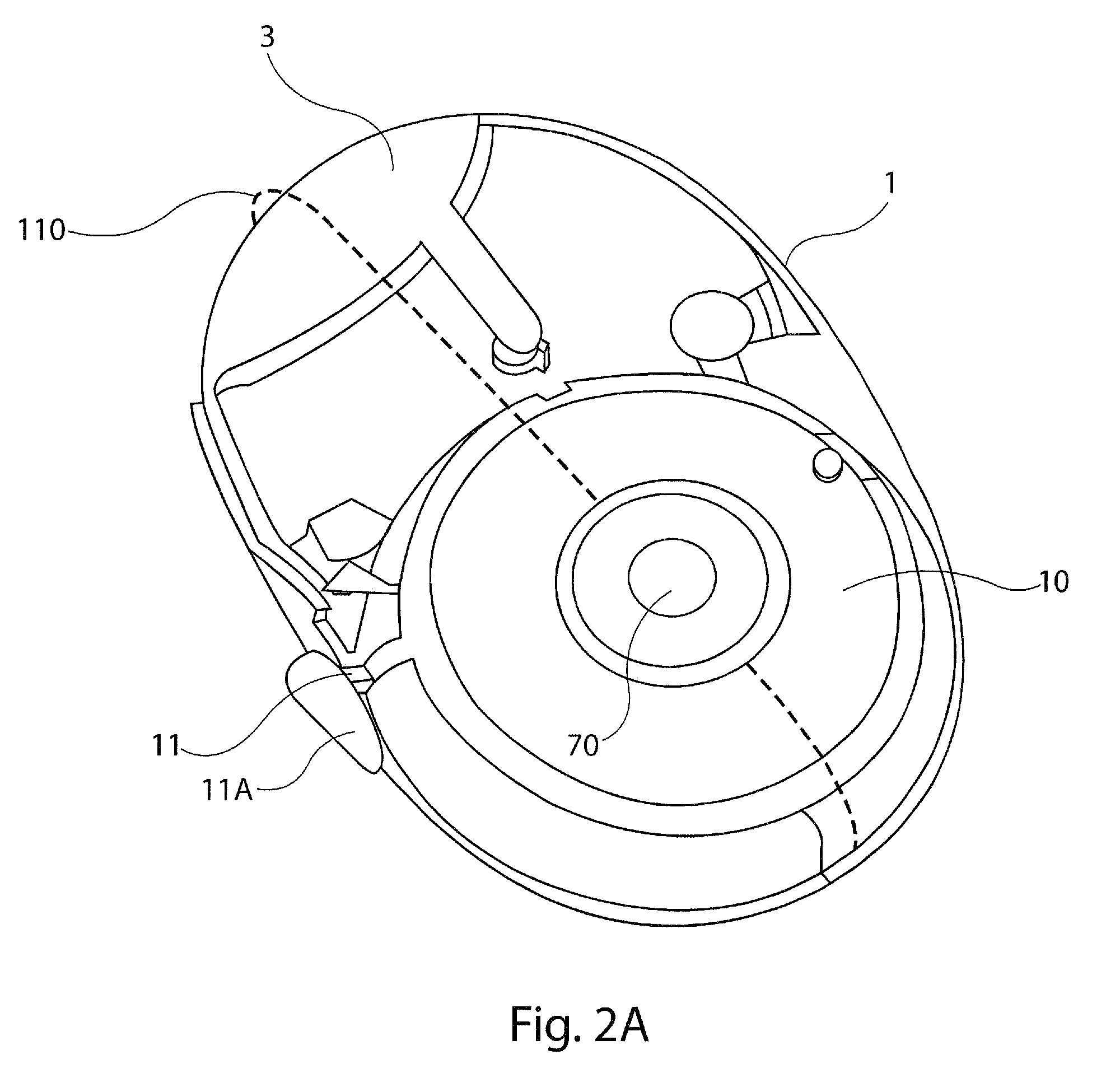
[0028]The present disclosure provides a device for delivering medicament to the airway of a user, wherein the device generally comprises a housing with a mouthpiece affixed and a cover for the mouthpiece. The housing is adapted to hold a plurality of individual blister packs containing, for example, powdered medicament. However, the medication could be a liquid form medication. The blisters are arranged such that individual blisters may be loaded into a clamping position, whereupon the blister is pierced and a vibrating device is used to aggregate the contents of the blister, which is subsequently inhaled by the user. Preferably the blisters are ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com