Medicine compound for treating obese hypertensives and preparation method thereof
A technology for obesity-type hypertension and a composition is applied in the field of pharmaceutical compositions for treating obese-type hypertension patients and the field of preparation thereof, and achieves the effect of significant curative effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
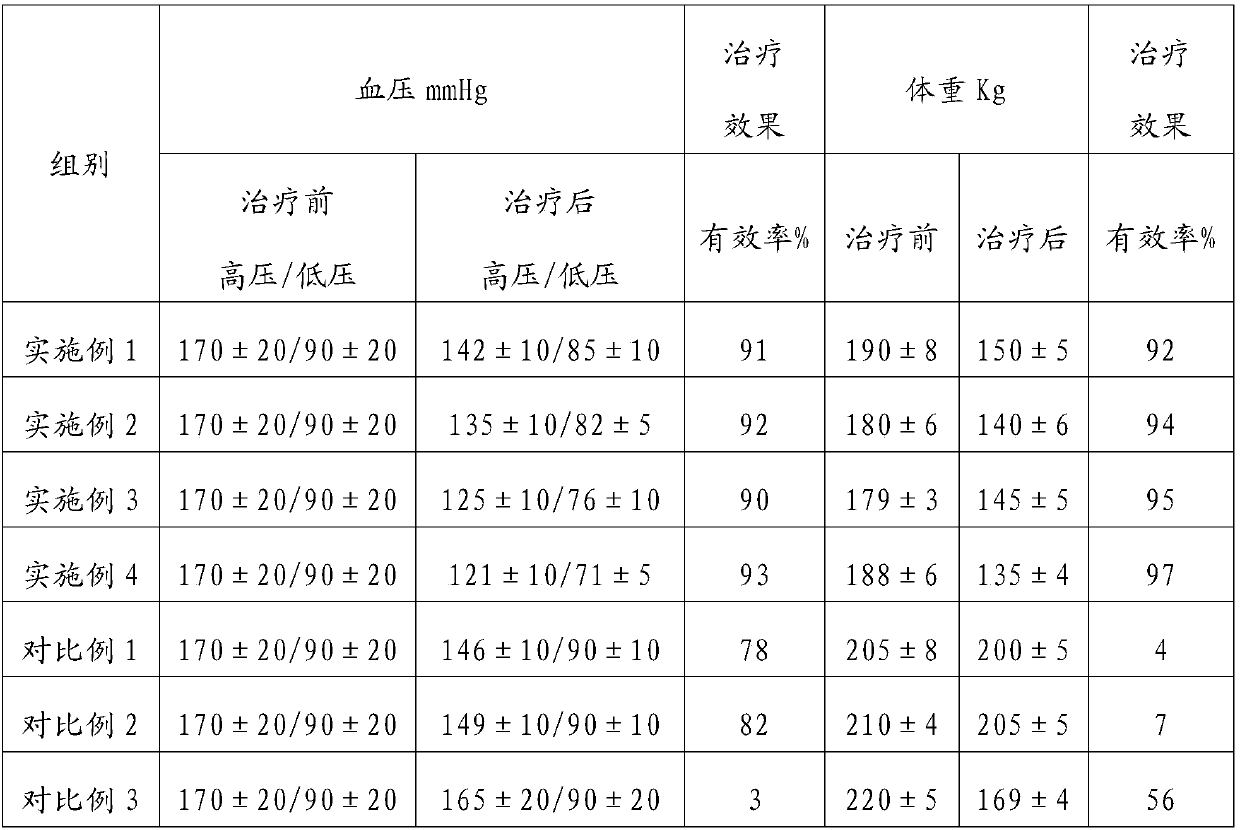
Embodiment 1
[0015] In parts by weight, 50 parts of alprenol and 100 parts of orlistat are prepared into alprenol sustained-release pellets and orlistat sustained-release pellets with clinically acceptable auxiliary materials, and then mixed Made into capsules to obtain pharmaceutical composition capsules.
Embodiment 2
[0017] In parts by weight, 70 parts of alprenol and 100 parts of orlistat are prepared into alprenol sustained-release pellets and orlistat sustained-release pellets with clinically acceptable auxiliary materials, and then mixed Made into capsules to obtain pharmaceutical composition capsules.
Embodiment 3
[0019] In terms of parts by weight, 50 parts of metyrolol and 100 parts of orlistat were taken, and clinically acceptable auxiliary materials were used to prepare metyrolol sustained-release pellets and orlistat sustained-release pellets respectively, and then mixed to prepare Form into capsules to obtain pharmaceutical composition capsules.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com