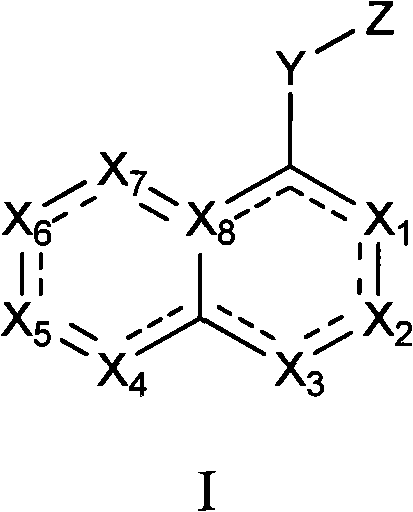
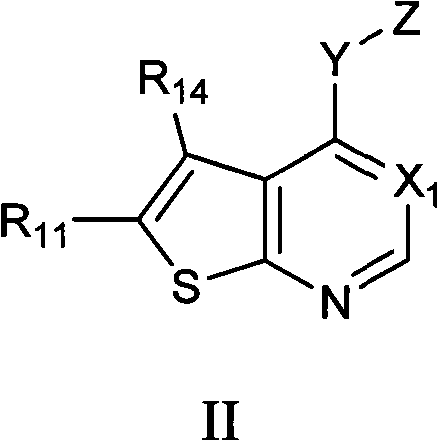
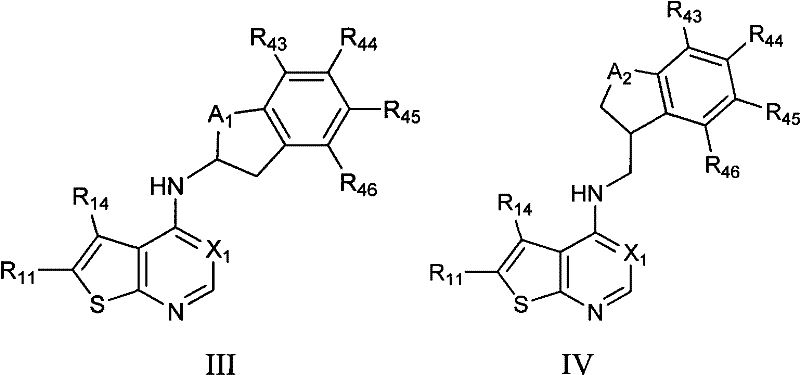
Aminopyrimidine inhibitors of histamine receptors for the treatment of disease
A technology for diseases, alkylation, applied in the field of medicine
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0302]
[0303] 4-(4-(3,4-dichlorophenyl)piperazin-1-yl)-5-methylthieno[2,3-d]pyrimidine
[0304] The title compound was obtained commercially.
Embodiment 2
[0306]
[0307] 5-methyl-4-(4-(5-(trifluoromethyl)pyridin-2-yl)piperazin-1-yl)thieno[2,3-d]pyrimidine
[0308] The title compound was obtained commercially.
Embodiment 3
[0310]
[0311] 4-(4-(2-Chlorophenyl)piperazin-1-yl)-5-methylthieno[2,3-d]pyrimidine
[0312] The title compound was obtained commercially.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com