Materials Development: Ion-Exchange Polymers And Catalytic Layers For BPMED
BPMED Ion-Exchange Polymer Technology Background and Objectives
Bipolar membrane electrodialysis (BPMED) technology has evolved significantly since its inception in the mid-20th century. Initially developed as an extension of conventional electrodialysis, BPMED has transformed from a laboratory curiosity into a promising industrial separation technology. The fundamental principle—utilizing ion-exchange membranes to facilitate controlled ion transport under an electric field—has remained consistent, while material science advancements have dramatically improved performance metrics.
The evolution of ion-exchange polymers for BPMED applications has followed three distinct phases. The first generation, developed in the 1950s-1970s, consisted primarily of polystyrene-based materials with limited stability and selectivity. The second generation (1980s-2000s) introduced fluorinated polymers and composite structures, significantly enhancing chemical stability and operational lifetime. The current third generation focuses on nanoscale engineering and functional group optimization to achieve unprecedented ion selectivity and reduced electrical resistance.
Catalytic layers, a critical component in BPMED systems, have similarly undergone substantial development. Early systems relied on simple metal oxide catalysts with limited water dissociation efficiency. Modern catalytic layers incorporate advanced materials such as transition metal complexes, conductive polymers, and more recently, nanoscale architectures that dramatically enhance water splitting kinetics while minimizing energy consumption.
The primary technical objectives for next-generation BPMED materials development center around four key parameters: enhanced ion selectivity, improved mechanical and chemical stability, reduced electrical resistance, and optimized water dissociation kinetics at the bipolar interface. These objectives are driven by the increasing industrial demand for energy-efficient separation processes, particularly in applications involving acid and base recovery from waste streams, salt splitting operations, and the production of high-purity chemicals.
Current research trajectories indicate a convergence toward multifunctional materials that simultaneously address multiple performance limitations. This includes the development of composite membranes with distinct functional layers, incorporation of nanomaterials to enhance specific properties, and the exploration of bio-inspired designs that mimic natural ion transport mechanisms.
The ultimate goal of BPMED materials development is to create systems capable of operating at higher current densities with lower energy consumption, extended operational lifetimes, and reduced manufacturing costs. These improvements would position BPMED as a competitive alternative to conventional chemical processes in various industrial applications, particularly in scenarios where environmental sustainability and resource recovery are prioritized.
Market Analysis for BPMED Applications
The Bipolar Membrane Electrodialysis (BPMED) technology market is experiencing significant growth driven by increasing industrial demand for sustainable acid and base production methods. Current market valuation stands at approximately 320 million USD globally, with projections indicating a compound annual growth rate of 8.7% over the next five years. This growth trajectory is primarily fueled by stringent environmental regulations and the rising costs of traditional chemical production methods across various industries.
The chemical processing sector represents the largest market segment for BPMED applications, accounting for roughly 42% of the total market share. Within this segment, the production of high-purity acids and bases for specialty chemical manufacturing demonstrates particularly strong demand growth. The food and beverage industry follows as the second-largest application sector, where BPMED systems are increasingly utilized for deacidification processes and pH adjustment in production lines.
Geographically, Europe currently leads the BPMED market with approximately 38% market share, attributed to its stringent environmental regulations and strong focus on sustainable industrial processes. North America follows at 29%, while the Asia-Pacific region, particularly China and India, represents the fastest-growing market with annual growth rates exceeding 12%.
A critical market driver for advanced ion-exchange polymers and catalytic layers is the push toward higher energy efficiency in BPMED systems. End-users report that energy consumption represents 40-60% of operational costs in current BPMED installations, creating strong market pull for materials that can reduce this expenditure. Materials that can demonstrate a 15% or greater reduction in energy requirements while maintaining performance are positioned to capture significant market share.
Water treatment applications represent an emerging high-growth segment, with municipal and industrial wastewater treatment facilities increasingly exploring BPMED for selective ion removal and resource recovery. This segment is projected to grow at 14.2% annually, outpacing the overall market growth rate.
Market analysis indicates that durability and operational lifetime of ion-exchange membranes and catalytic layers remain key purchasing factors. Current materials typically require replacement every 2-3 years, representing a substantial operational cost. Materials innovations that can extend operational lifetime to 5+ years while maintaining performance characteristics could command premium pricing, with customers indicating willingness to pay 30-40% more for such improvements.
The competitive landscape shows increasing consolidation, with five major manufacturers controlling approximately 67% of the global market for ion-exchange polymers used in BPMED applications. However, significant opportunities exist for new entrants with innovative materials solutions, particularly in specialized application niches where performance requirements cannot be met by standard offerings.
Current Challenges in Ion-Exchange Polymers and Catalytic Layers
Despite significant advancements in bipolar membrane electrodialysis (BPMED) technology, several critical challenges persist in the development of ion-exchange polymers and catalytic layers. The primary limitation of current ion-exchange membranes (IEMs) is their insufficient chemical stability under extreme pH conditions, particularly in highly acidic or alkaline environments that are common in BPMED operations. This instability leads to membrane degradation, reduced lifespan, and increased operational costs.
Water dissociation efficiency at the bipolar membrane interface remains suboptimal, creating a significant bottleneck in BPMED performance. Current catalytic layers often exhibit diminished activity over time, requiring frequent replacement and maintenance. The water splitting catalysts incorporated into these layers face challenges in maintaining consistent performance across varying operational conditions.
Ion selectivity presents another substantial challenge, as existing membranes frequently demonstrate inadequate selectivity between monovalent and multivalent ions. This limitation restricts the application scope of BPMED in processes requiring high purity outputs, such as pharmaceutical manufacturing and specialized chemical production.
The mechanical properties of ion-exchange polymers pose additional difficulties, with many materials exhibiting dimensional instability during operation. Swelling, shrinking, and deformation under pressure differentials and temperature fluctuations compromise membrane integrity and system efficiency. Furthermore, the interface between the cation and anion exchange layers in bipolar membranes often suffers from delamination issues during extended operation.
Manufacturing scalability represents a significant hurdle for advanced ion-exchange materials. Novel polymer compositions with superior properties often involve complex synthesis procedures that are difficult to scale up economically. The incorporation of catalytic nanoparticles into membrane structures in a uniform and reproducible manner remains technically challenging at industrial scales.
Electrical resistance across membranes continues to limit energy efficiency in BPMED systems. Current materials exhibit a trade-off between ion selectivity and electrical resistance, with improvements in selectivity typically resulting in higher resistance and consequently higher energy consumption.
Fouling susceptibility further complicates BPMED operations, as organic and inorganic foulants accumulate on membrane surfaces and within porous structures, progressively reducing performance. Existing anti-fouling strategies often compromise other critical membrane properties, creating a complex design challenge for materials scientists.
The development of environmentally sustainable ion-exchange polymers presents an emerging challenge, with traditional materials often relying on fluorinated compounds that raise environmental concerns. The transition to greener alternatives while maintaining performance specifications requires significant innovation in polymer chemistry and membrane fabrication techniques.
Current Material Solutions for BPMED Systems
01 Ion-exchange polymers in fuel cell applications
Ion-exchange polymers are widely used in fuel cell applications, particularly as proton exchange membranes (PEMs). These polymers facilitate the transport of protons while acting as an electronic insulator and reactant barrier. The catalytic layers in fuel cells often incorporate these polymers to create a three-phase boundary where electrochemical reactions occur. Perfluorosulfonic acid polymers like Nafion are commonly used due to their excellent proton conductivity and chemical stability.- Ion-exchange polymers in fuel cell applications: Ion-exchange polymers are widely used in fuel cell applications, particularly as proton exchange membranes (PEMs). These polymers facilitate the transport of protons while acting as an electronic insulator and reactant barrier. The catalytic layers in fuel cells often incorporate these polymers to create a three-phase boundary where electrochemical reactions occur. Perfluorosulfonic acid polymers like Nafion are commonly used due to their excellent proton conductivity and chemical stability under fuel cell operating conditions.
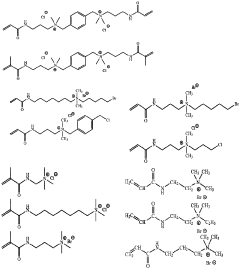
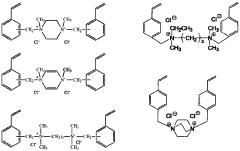
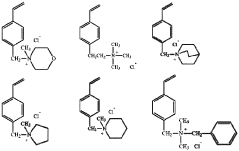
- Synthesis and modification of ion-exchange polymers: Various methods for synthesizing and modifying ion-exchange polymers have been developed to enhance their performance in catalytic applications. These include copolymerization techniques, surface modification, and incorporation of functional groups to improve ion conductivity, mechanical strength, and chemical stability. Modified polymers can exhibit enhanced catalytic activity, selectivity, and durability. Techniques such as grafting, cross-linking, and blending are employed to tailor the properties of ion-exchange polymers for specific catalytic applications.
- Catalytic layers with embedded ion-exchange polymers: Catalytic layers incorporating ion-exchange polymers provide unique environments for chemical reactions. These layers typically consist of catalyst particles (often noble metals) dispersed within an ion-exchange polymer matrix. The polymer facilitates ion transport to and from catalytic sites while providing structural support. The design of these layers focuses on optimizing the interface between the catalyst and polymer to enhance reaction efficiency. Parameters such as polymer loading, catalyst distribution, and layer thickness significantly impact the performance of these catalytic systems.
- Environmental applications of ion-exchange polymer catalysts: Ion-exchange polymers combined with catalytic materials are increasingly used in environmental applications such as water purification, air pollution control, and waste treatment. These systems can effectively remove contaminants through combined ion-exchange and catalytic degradation mechanisms. The polymers provide selective binding sites for pollutants while the catalytic components facilitate their transformation into less harmful substances. This dual functionality makes these materials particularly effective for treating complex environmental contaminants.
- Novel composite materials combining ion-exchange polymers with inorganic catalysts: Recent developments include composite materials that combine ion-exchange polymers with inorganic catalytic components such as metal oxides, zeolites, or nanoparticles. These hybrid materials exhibit synergistic properties that enhance catalytic performance. The polymer component provides ion selectivity and a controlled microenvironment, while the inorganic component contributes specific catalytic functions. These composites often show improved stability, reusability, and catalytic efficiency compared to their individual components. Applications include chemical synthesis, energy conversion, and advanced separation processes.
02 Synthesis and modification of ion-exchange polymers
Various methods for synthesizing and modifying ion-exchange polymers have been developed to enhance their properties for specific applications. These include copolymerization techniques, surface modification, and incorporation of functional groups. Modified ion-exchange polymers can exhibit improved mechanical strength, thermal stability, and ion selectivity. Techniques such as radiation grafting, chemical crosslinking, and the addition of inorganic fillers are commonly employed to tailor the properties of these materials.Expand Specific Solutions03 Catalytic layers with ion-exchange polymers for electrochemical applications
Catalytic layers incorporating ion-exchange polymers are crucial components in various electrochemical devices. These layers typically consist of catalyst particles (often noble metals like platinum) dispersed within an ion-exchange polymer matrix. The polymer provides ionic conductivity while the catalyst facilitates the electrochemical reactions. The structure and composition of these layers significantly impact the performance of electrochemical devices, with factors such as catalyst loading, polymer content, and layer thickness being critical parameters.Expand Specific Solutions04 Water treatment and purification applications
Ion-exchange polymers are extensively used in water treatment and purification processes. These polymers can selectively remove ions from aqueous solutions through ion-exchange mechanisms. Catalytic layers containing ion-exchange polymers can enhance the efficiency of water treatment processes by combining ion-exchange capabilities with catalytic degradation of contaminants. Applications include desalination, softening, deionization, and removal of heavy metals and organic pollutants from water.Expand Specific Solutions05 Novel composite materials combining ion-exchange polymers with inorganic components
Hybrid materials that combine ion-exchange polymers with inorganic components represent an important advancement in the field. These composites often exhibit synergistic properties that enhance performance in various applications. Inorganic components such as metal oxides, silica, and clay minerals can improve the mechanical, thermal, and chemical stability of ion-exchange polymers. These hybrid materials find applications in catalysis, separation processes, sensors, and energy storage and conversion devices.Expand Specific Solutions
Key Industry Players in BPMED Technology
The bipolar membrane electrodialysis (BPMED) technology for ion-exchange polymers and catalytic layers is currently in a growth phase, with increasing market adoption driven by sustainability demands. The global market is expanding, estimated to reach significant scale as industries seek efficient separation technologies. Technologically, the field shows moderate maturity with established players like FUJIFILM Corp. and Kuraray leading membrane development, while companies such as DuPont and Tokuyama focus on ion-exchange materials. Chinese institutions including Sinopec and Zhejiang University of Technology are rapidly advancing catalytic layer research. European entities like Commissariat à l'énergie atomique contribute significant innovations in polymer chemistry. The competitive landscape features both specialized membrane manufacturers and diversified chemical corporations, with increasing collaboration between academic institutions and industry players to overcome performance and durability challenges.
FUJIFILM Corp.
China Petroleum & Chemical Corp.
Critical Patents and Innovations in Ion-Exchange Membranes
- A bipolar membrane comprising an anionic and cationic ion-exchange membrane with a gel based on sulphate and/or hydrated metal sulphite, excluding indium, cerium, manganese, and copper sulphates, is developed, promoting water dissociation with reduced voltage requirements.
- The CEL and AEL are made visibly distinguishable through differences in their color properties, using dyes or colored photoinitiators that create distinct color differences measurable by CIEDE2000 standards, allowing for easy identification and correct orientation in membrane stacks.
Environmental Impact and Sustainability Considerations
The development of ion-exchange polymers and catalytic layers for Bipolar Membrane Electrodialysis (BPMED) must be evaluated not only for technical performance but also for environmental impact and sustainability considerations. Current manufacturing processes for these materials often involve fluorinated compounds and toxic solvents, raising significant environmental concerns. The production of perfluorinated polymers, commonly used in ion-exchange membranes, generates persistent pollutants that bioaccumulate in ecosystems and pose long-term environmental risks.
Life cycle assessment (LCA) studies indicate that traditional ion-exchange membrane production contributes substantially to global warming potential, acidification, and resource depletion. The energy-intensive synthesis processes and use of petroleum-derived precursors further exacerbate the carbon footprint of these materials. Additionally, end-of-life considerations remain problematic, as most current ion-exchange polymers are not biodegradable and present challenges for recycling or safe disposal.
Recent research has focused on developing more sustainable alternatives, including bio-based ion-exchange polymers derived from renewable resources such as cellulose, chitosan, and lignin. These materials offer promising environmental advantages, including reduced toxicity, lower carbon footprint, and improved biodegradability. For catalytic layers, researchers are exploring earth-abundant metals to replace precious metal catalysts, significantly reducing resource depletion concerns while maintaining electrochemical performance.
Water consumption represents another critical environmental factor in BPMED technology deployment. While the technology itself aims to address water treatment challenges, the manufacturing processes for membranes and catalytic layers can be water-intensive. Innovative green chemistry approaches are emerging to reduce solvent use and implement closed-loop water recycling systems in production facilities.
Regulatory frameworks worldwide are increasingly emphasizing the need for sustainable materials in clean energy technologies. The European Union's REACH regulations and similar initiatives globally are driving the transition away from hazardous substances in membrane materials. This regulatory landscape is accelerating research into environmentally benign alternatives that maintain or exceed the performance of conventional materials.
Economic sustainability must also be considered alongside environmental factors. The commercial viability of new, greener materials depends on achieving cost parity with established technologies. Current research indicates that scale-up of bio-based alternatives could potentially reduce costs while simultaneously decreasing environmental impact, creating a positive feedback loop for sustainable innovation in the BPMED sector.
Scalability and Manufacturing Challenges
The scaling of ion-exchange polymers and catalytic layers for Bipolar Membrane Electrodialysis (BPMED) from laboratory to industrial scale presents significant manufacturing challenges. Current production methods for high-performance membranes often involve complex, multi-step processes that are difficult to standardize across large production volumes. The precision required for controlling membrane thickness, ion-exchange capacity, and catalytic layer deposition creates bottlenecks in mass production scenarios.
Material consistency represents a primary challenge, as variations in polymer composition can dramatically affect membrane performance. Industrial-scale polymerization processes must maintain tight control over reaction conditions to ensure uniform molecular weight distribution and cross-linking density. Even minor deviations can lead to inconsistent ion transport properties, compromising the efficiency of BPMED systems.
For catalytic layers, the uniform deposition of nanoscale materials across large membrane surfaces remains problematic. Laboratory techniques such as spin coating or electrodeposition work effectively for small samples but face significant hurdles when scaled to commercial dimensions. The catalytic activity often degrades during scale-up due to aggregation phenomena and non-uniform distribution of active sites.
Cost considerations further complicate manufacturing scale-up. Many high-performance ion-exchange polymers rely on expensive fluorinated precursors or specialty monomers. Similarly, catalytic layers may incorporate precious metals or complex metal oxides that significantly impact production economics. Finding cost-effective alternatives without sacrificing performance represents a critical challenge for widespread BPMED adoption.
Environmental and safety concerns also affect manufacturing scalability. Traditional membrane production often involves hazardous solvents and chemicals that pose challenges for worker safety and environmental compliance at industrial scales. Developing greener synthesis routes and safer processing methods is essential for sustainable large-scale production.
Quality control methodologies must evolve alongside production scale-up. Current analytical techniques for characterizing membrane properties are often time-consuming and difficult to implement as in-line monitoring tools. Developing rapid, non-destructive testing methods capable of evaluating key performance parameters during production would significantly enhance manufacturing efficiency.
Addressing these challenges requires interdisciplinary collaboration between materials scientists, chemical engineers, and manufacturing specialists. Recent advances in continuous flow chemistry, automated quality control systems, and environmentally benign synthesis routes offer promising pathways toward overcoming these scalability hurdles, potentially enabling wider commercial deployment of BPMED technology across various industrial applications.